Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
267 results for "LAP (TGF-beta 1)" in 产品
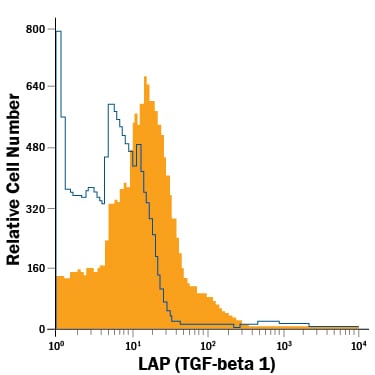
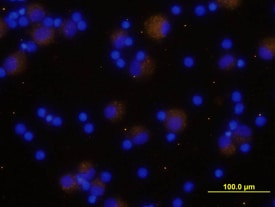
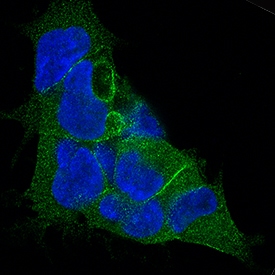
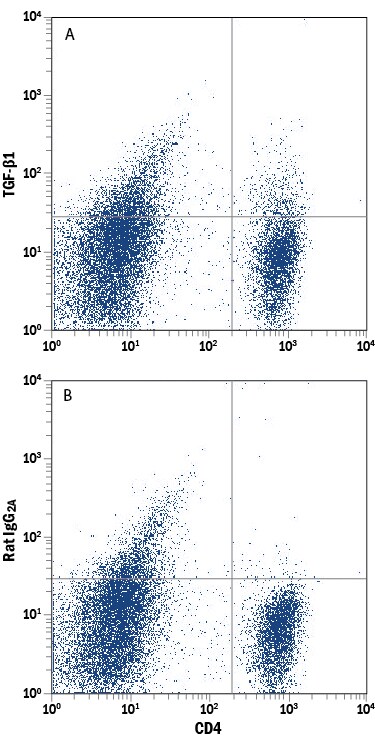
LAP (TGF-beta 1) Products
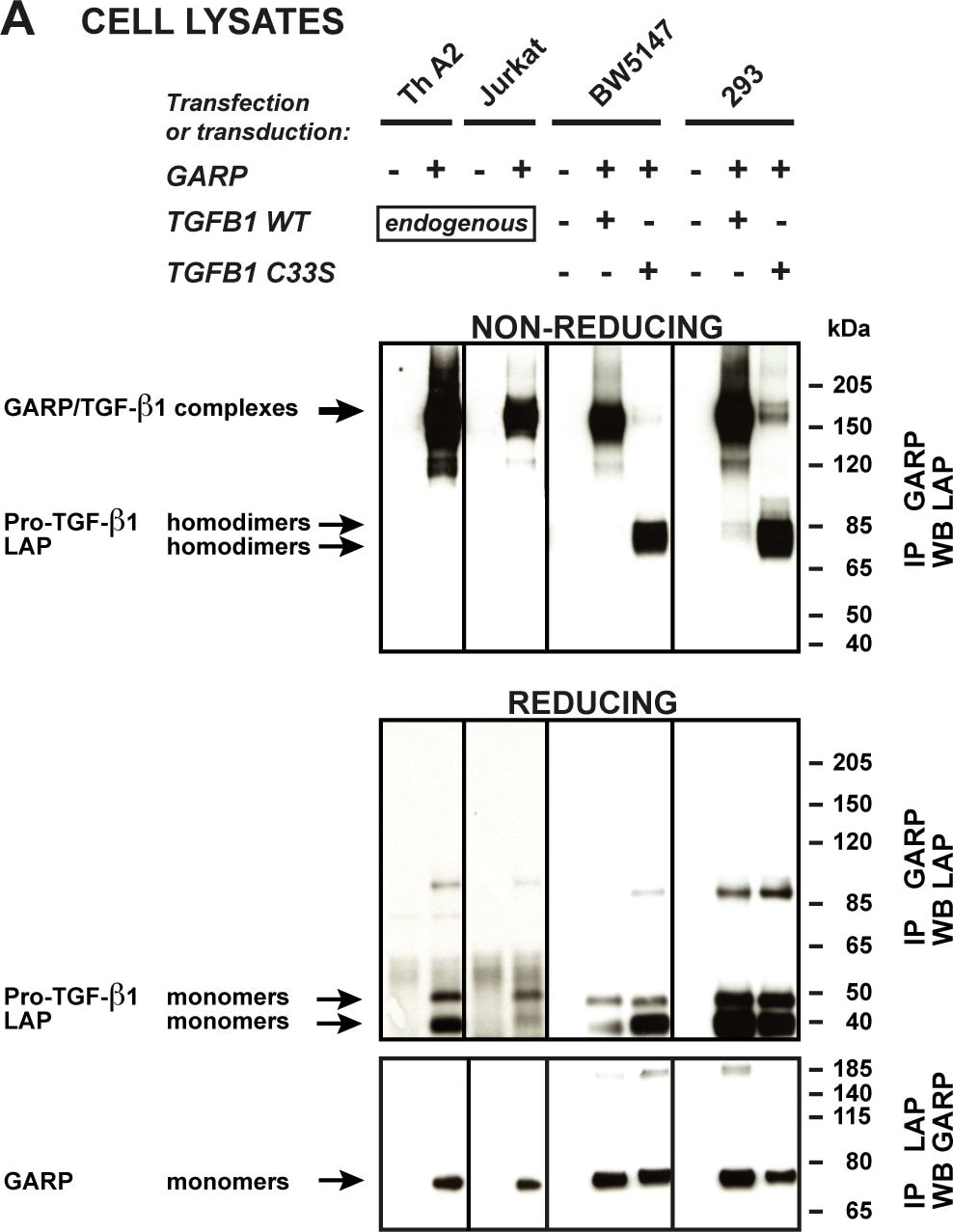
TGF-beta 1 is expressed as a proprotein that is cleaved within the trans-Golgi to yield a latency-associated peptide (LAP) and the mature TGF-beta 1. Disulfide-linked homodimers of LAP and TGF-beta 1 remain noncovalently associated after secretion, forming the small latent TGF-beta 1 complex. Purified LAP can also associate with active TGF-beta and neutralize TGF-beta activity. Covalent linkage of LAP to one of three latent TGF-beta binding proteins (LTBPs) creates a large latent complex that ...
| Reactivity: | Human |
| Source: | Sf 21 (baculovirus) |
| Accession #: | P01137.2 |
| Applications: | Bioactivity |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF, Simple Western |
Loading...
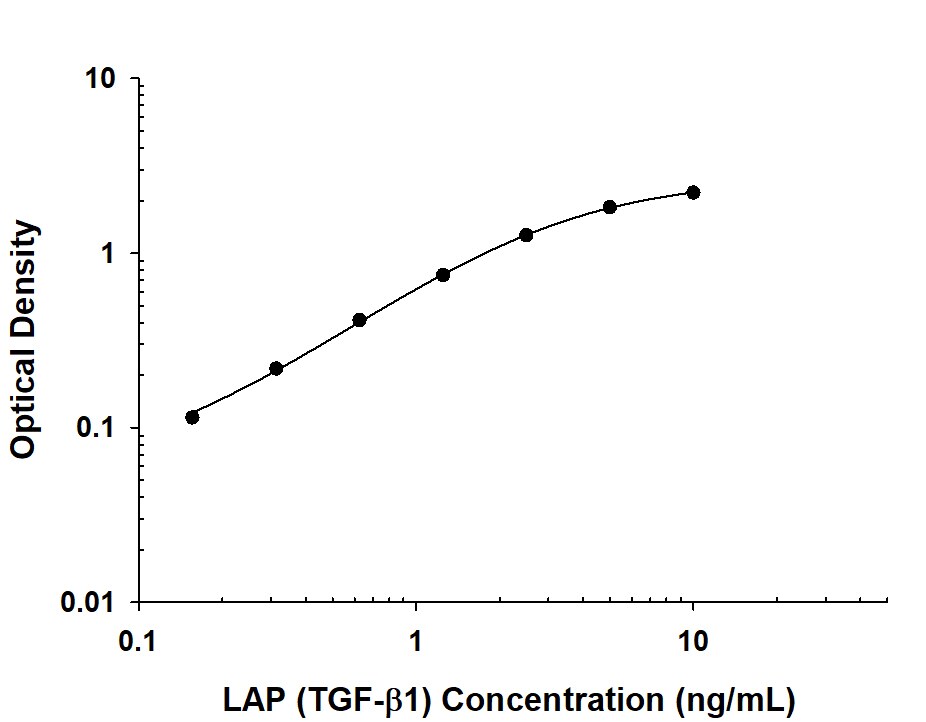
| Reactivity: | Human |
| Sensitivity: | 3.4 pg/mL |
| Assay Range: | 23.4-1500 pg/mL (Cell Culture Supernates, Cell Lysates, Serum, Platelet-poor EDTA Plasma, Platelet-poor Heparin Plasma, Urine) |
| Applications: | ELISA |
Loading...
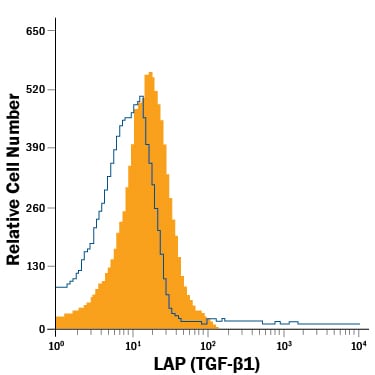
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #7F6 |
| Applications: | IHC, WB, ELISA, Flow |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB, B/N, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #27235 |
| Applications: | B/N, ICC/IF |
Loading...
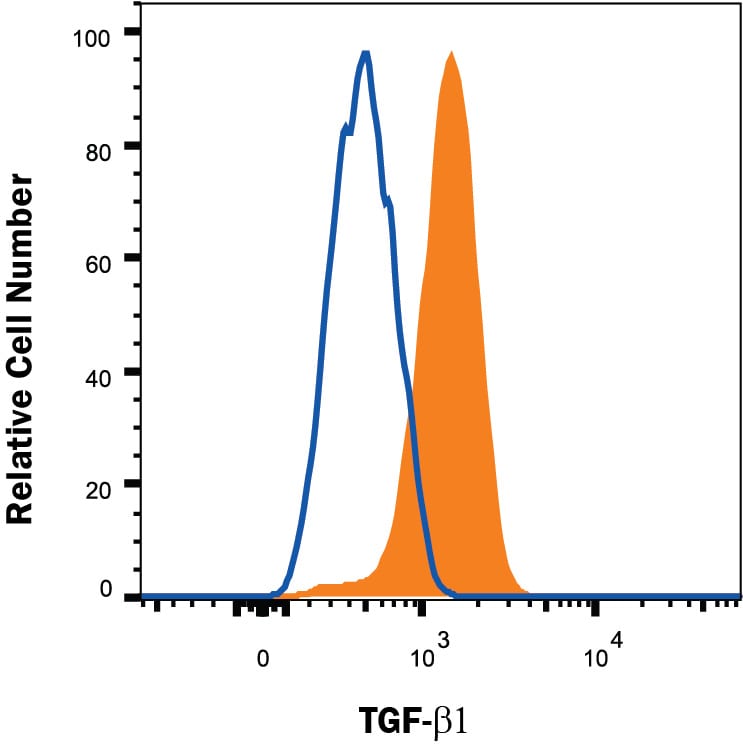
| Reactivity: | Mouse |
| Details: | Rat IgG2A Monoclonal Clone #860206 |
| Applications: | Flow, CyTOF-ready |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #27232 |
| Applications: | Flow, CyTOF-ready |
Loading...
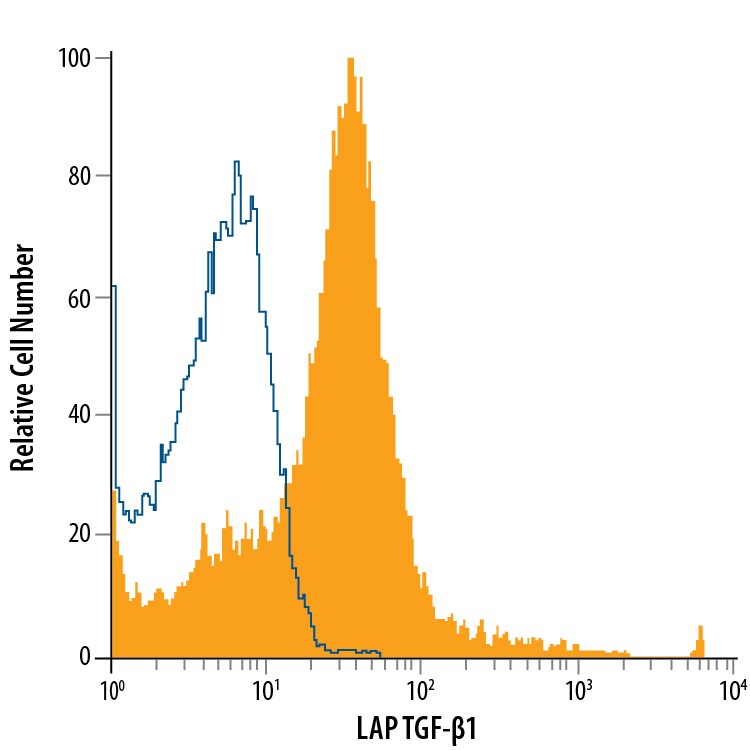
| Reactivity: | Human, Mouse, Rat |
| Details: | Mouse IgG1 Monoclonal Clone #7F6 |
| Applications: | IHC, WB, ELISA, Flow, CyTOF-ready |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #27232 |
| Applications: | Flow |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #27240 |
| Applications: | ELISA(Det) |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #1015022 |
| Applications: | ELISA, Flow, ICC/IF, CyTOF-ready |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #9005 |
| Applications: | ELISA(Cap) |
Loading...
| Reactivity: | Human |
| Assay Range: | 62.5-4000 pg/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #1015016 |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #27232 |
| Applications: | Flow |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #27232 |
| Applications: | Flow, ICC/IF |
Loading...
For use with catalog number DLAP00
| Reactivity: | Human |
| Applications: | ELISA, Ctrl |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, B/N, ICC/IF |
Loading...






![Western Blot: LAP (TGF-beta 1) Antibody (7F6)BSA Free [NBP2-22114] Western Blot: LAP (TGF-beta 1) Antibody (7F6)BSA Free [NBP2-22114]](https://resources.rndsystems.com/images/products/TGF-beta-1-Antibody-7F6-Western-Blot-NBP2-22114-img0002.jpg)
![Western Blot: LAP (TGF-beta 1) Antibody (7F6)Azide and BSA Free [NBP2-80827] Western Blot: LAP (TGF-beta 1) Antibody (7F6)Azide and BSA Free [NBP2-80827]](https://resources.rndsystems.com/images/products/LAP-TGF-beta-1-Antibody-7F6-Azide-and-BSA-Free-Western-Blot-NBP2-80827-img0002.jpg)