Immunoassay Solutions
Solutions for discovery, validation and verification workflows.
Explore trusted immunoassay solutions from R&D Systems, ranging from reliable ELISA kits and Luminex® assays to advanced automated platforms like Simple Plex and Simple Western. Built with rigorously tested in-house antibodies and proteins, our assays are designed to deliver accurate, reproducible, and high-quality results every time. Whether you need single-analyte testing, multiplexing, or custom solutions, our platforms offer seamless adaptability to accelerate your biomarker research and discovery. Boost precision, reproducibility, and efficiency with cutting-edge technologies tailored to your workflow and research goals.
Immunoassays at a Glance
ELISA Kits
Single Analyte
Legendary reproducibility has helped make the Quantikine ELISA Kits the most trusted name in ELISA kits. Carefully selected antibody pairs and optimally formulated diluents help ensure accurate and reproducible results from one experiment to the next. Choose from over 1,200 target analytes.
Multiplex Luminex Assays

Multi-analyte
Our Luminex Assays are designed to maximize multiplexing capacity and flexibility while maintaining assay specificity. Personalize your Luminex panel with the largest, mix and match menu on the market. Choose from >490 analytes across multiple species.
Simple Plex Assays

Multi-Analyte and Single Analyte
Simple Plex Assays on the Ella Platform deliver precise, automated immunoassays with ready-to-go cartridges for analysis of up to 8 analytes per sample. Get reproducible results in under 90 minutes with no manual steps, choosing from 380+ analytes across multiple species.
Proteome Profiler Antibody Arrays

Multiplex Arrays
Proteome Profiler Antibody Arrays are multiplex membrane assays for early-stage discovery research, measuring over 100 proteins per sample. Kits include buffers, detection antibodies, and membranes spotted in duplicate with high-quality capture antibodies. Arrays utilize chemiluminescence for detection, and are imaged similar to traditional Western blots.
Simple Western Assays

Single or Multiplex Western Assays
Automated capillary western systems powered by Simple Western technology provide absolute quantitation to measure endogenous protein expression in cell or tissue lysates. Process up to 96 samples in 3 hours with only 3 µL of lysate samples.
Which Immunoassay is Right For Me?
| Assay | Benefits | Number of Analytes | Format | Sample Volume | Throughput | Assay Time | Instrument |
|---|---|---|---|---|---|---|---|
| DuoSet ELISA Kits | Largest menu, economical, flexible | 1 | Matched antibody pair sandwich immunoassay | 10-200 µL | 96-well plate | 20 hrs | Plate reader |
| Quantikine ELISA Kits | Most published, gold standard | 1 | Matched antibody pair sandwich immunoassay | 10-200 µL | 96-well plate | 1.5-5 hrs | Plate reader |
| Luminex Assays | Large multiplex panels, customization | Up to 50 | Bead-based (multiplex microspheres) | 50 µL | 96- or 384-well plate | 4 hrs | Luminex system |
| Simple Plex Assays | Hands-free, rapid and reproducible quantification | Up to 8 | Microfluidic cartridge-based | 2.5-25 µL | 16-,32, and 72-sample cartridge | 1.5 hrs | Ella |
| Proteome Profiler Arrays | Largest screening capabilities | Up to 119 | Membrane array | 50- 500 µL | 1 sample per membrane | 20 hrs | Imaging system |
| Simple Western | Open platform, size and quantitation information | Up to 8 | Capillary western | 3 µL | Up to 100 capillaries | 3 hrs | Abby, Jess, Leo |
Finding the Right Immunoassay from Discovery to Validation
Multiplex Assays
Discovery
- Analyze up to 119 proteins simultaneously
- Quickly assess complex signaling responses
Validation
High Performance Luminex Assays
- Rigorously validated for accuracy, and precision
- Flexible panels available
Validated Assays
- Automated detection of up to 8 analytes
- Rigorously validated for accuracy, precision and reproducibility
- Results in 90 minutes
Single Analyte Assays
Discovery
- Adaptable to multiple platforms
- Over 1,200 kits available covering 16 species
Verification & Validation
- Exhaustively validated for superior performance and reproducibility
Validated Assays
- Rigorously validated for accuracy, precision and reproducibility
- Hands-free, Results in 90 minutes
Move Between Immunoassay Platforms
We offer a full catalog of immunoassay solutions so you can find the best platform for your needs. When your needs change, you can also move between platforms with confidence and consistency in your results. High-Performance Luminex Assays and Simple Plex Cartridges are both correlated to the gold standard Quantikine ELISA. Whether you are starting with a multiplex assay or single analyte, we have your current and future needs covered.
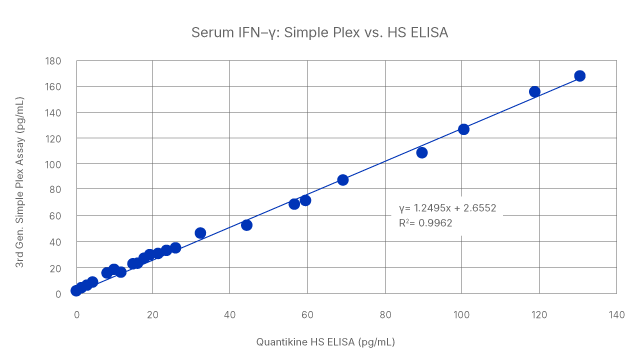
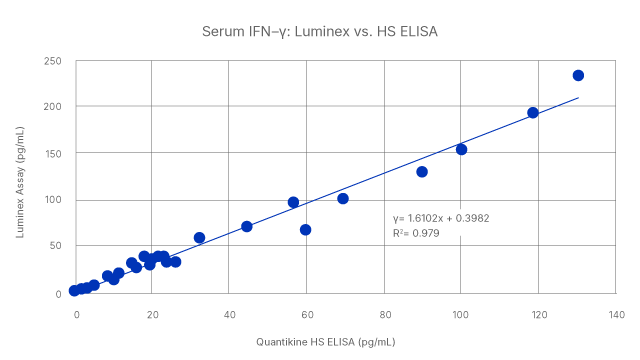
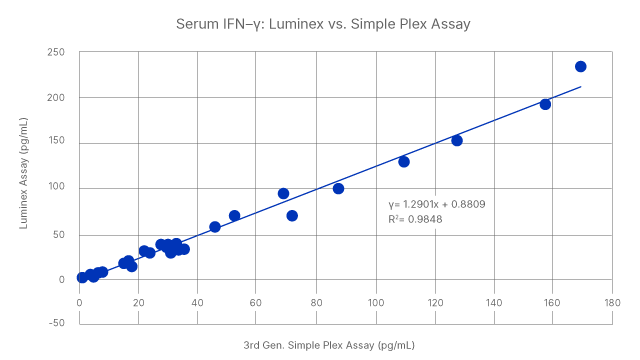
Seamless correlation across platforms: Human serum levels of IFN-gamma was measured using High Sensitivity Quantikine ELISA, High-Performance Luminex Assay, and Simple Plex assays. The results show a tight correlation across the three types of immunoassays, with R2 values of >0.979.
R&D Systems Immunoassay Solutions

Learn about the different available immunoassay platforms, benefits, and the list of analytes.
Assay Insider

Assay Insider, your go-to newsletter for staying up-to-date on the latest developments in immunoassays from R&D Systems. Each issue features industry news, new resources, tips and tricks, customer stories, new and featured products.
ELISA Guide
A clear, comprehensive guide to ELISAs. Learn about different ELISA formats, controls, best practices, and troubleshooting.
Luminex Assay Guide

The Luminex Assays User Guide offers insights on choosing the right multiplex assay and ensuring consistent, reliable performance.
Simple Plex Assay Guide

Simple Plex assays are hands-free, single or multi-analyte with no cross-reactivity, and reproducible even across multiple labs.
