Siglecs (sialic acid binding Ig-like lectins) are I-type (Ig-type) lectins belonging to the Ig superfamily. They are characterized by an N‑terminal Ig-like V-type domain which mediates sialic acid binding, followed by varying numbers of Ig-like C2-type domains (1, 2). Eleven human Siglecs have been cloned and characterized. They are sialoadhesin/CD169/Siglec-1, CD22/Siglec-2, CD33/Siglec-3, Myelin-Associated Glycoprotein (MAG/Siglec-4a) and Siglecs 5 to 11 (1‑3). To date, no Siglec has been shown to recognized any cell surface ligand other than sialic acids, suggesting that interactions with glycans containing this carbohydrate are important in mediating the biological functions of Siglecs. Siglecs 5 to 11 share a high degree of sequence similarity with CD33/Siglec-3 both in their extracellular and intracellular regions. They are collectively referred to as CD33-related Siglecs. One remarkable feature of the CD33-related Siglecs is their differential expression pattern within the hematopoietic system (1, 2). This fact, together with the presence of two conserved immunoreceptor tyrosine-based inhibition motifs (ITIMs) in their cytoplasma tails, suggests that CD33-related Siglecs are involved in the regulation of cellular activation within the immune system.
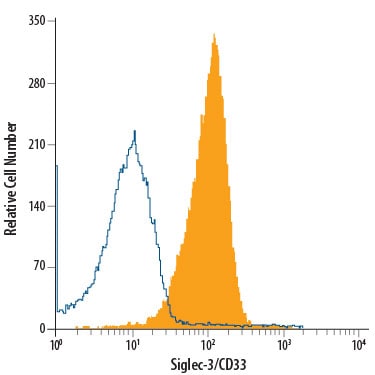
Human Siglec-3 is alternatively known as myeloid cell surface antigen CD33 and GP67. Human Siglec-3 cDNA encodes a 364 amino acid (aa) polypeptide with a hydrophobic signal peptide, an N-terminal Ig-like V-type domain, one Ig-like C2-type domains, a transmembrane region and a cytoplasmic tail (1, 4). Siglec-3 expression is restricted to cells of myelomonocytic lineage (2). It binds sialic acid preferring alpha 2,3- linkage over alpha 2,6- linkage (5). Studies indicated that Siglec-3 recruits SHP-1 and SHP-2 to its ITIMs (6, 7). When co-crosslinking with Fc gamma R1, Siglec-3 inhibits tyrosine phosphorylation and calcium mobilization, suggesting Siglec-3 can mediate inhibitory signals (7).