NPS 2390
Tocris Bioscience | Catalog # 4134

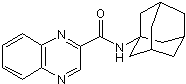
Product Description
NPS 2390 is a group I mGlu antagonist; displays noncompetitive antagonist activity at both mGlu1 and mGlu5 receptors. Thought to act on a site separate from the glutamate binding pocket.
Product Specifications for NPS 2390
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| DMSO | 15.37 | 50 | |
| Ethanol | 15.37 | 50 |
Preparing Stock Solutions for NPS 2390
The following data is based on the product molecular weight 307.39.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.5 mM | 6.51 mL | 32.53 mL | 65.06 mL |
| 2.5 mM | 1.30 mL | 6.51 mL | 13.01 mL |
| 5 mM | 0.65 mL | 3.25 mL | 6.51 mL |
| 25 mM | 0.13 mL | 0.65 mL | 1.30 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 12 publications citing the usage of this product.
- Mabire Synthesis, structure-activity relationship, and receptor pharmacology of a new series of quinoline derivatives acting as selective, noncompetitive mGlu1 antagonists. J.Med.Chem. 2005 PMID: 15771457
- van Wagenen Structure-activity relationship studies of NPS 2390: a potent and selective group I metabotropic glutamate receptor antagonist. Abstr.Soc.Neurosci.
- Lavreysen [3H]R214127: a novel high-affinity radioligand for the mGlu1 receptor reveals a common binding site shared by multiple allosteric antagonists. Mol.Pharmacol. 2003 PMID: 12695537
Product Documents for NPS 2390
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for NPS 2390
For research use only
Citations for NPS 2390
Customer Reviews for NPS 2390 (1)
Have you used NPS 2390?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: RatAssay Type: In VitroCell Line/Tissue: CerebellumVerified Customer | Posted 09/17/2019Compound requires sonication to be dissolved.NPS 2390 was used as a specific blocker of mGluR1 and mGluR5, to isolate pharmacologically response of GABA-A receptors. 10 microM of NPS 2390 in perfusion solution generated a full block of mGluR group I receptor activity, thus leaving a clear GABA-ergic response. Figure: single-channel openings of GABA-A receptor after application of NPS 2390.
There are no reviews that match your criteria.

