Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
R&D Systems | Catalog # PPS039

Discontinued Product
PPS039 has been discontinued.
View all Tryptophan Hydroxylase 1/TPH-1 products.
Key Product Details
Species Reactivity
Human, Mouse, Rat, Bovine, Canine, Chicken, Primate, Rabbit, Xenopus, Zebrafish
Applications
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Phosphopeptide corresponding to amino acid residues surrounding the phospho-S58 of Tryptophan Hydroxylase 1/TPH-1
Specificity
This antibody is specific for the ~55 kDa TPH phosphorylated at S58 in Western blots.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
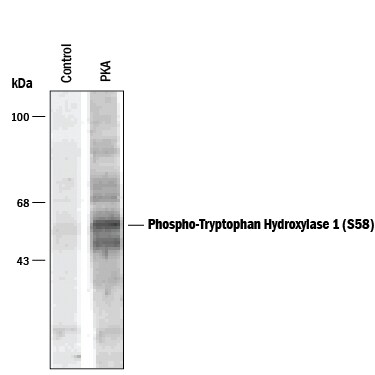
Detection of Phospho-Tryptophan Hydroxylase (S58) by Western Blot.
Western blot of recombinant rabbit tryptophan hydroxylase in a crude bacterial lysate incubated in the absence (Control) and presence of cAMP-dependent protein kinase (PKA).Applications for Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
Application
Recommended Usage
Western Blot
1:1000 dilution
Sample: Crude bacterial lysate
Sample: Crude bacterial lysate
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Formulation
100 μL in 10 mM HEPES (pH 7.5), 150 mM NaCl, 100 μg/mL BSA and 50% glycerol.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
For long-term storage, ≤‑20° C is recommended. Product is stable at ≤ ‑20° C for at least 1 year.
Background: Tryptophan Hydroxylase 1/TPH-1
References
- Boularand, S. et al. (1990) Nucleic Acids Res. 18:4257.
- Wang, G-A. et al. (1998) J. Neurochem. 71:1769.
- Kumer, S.C. et al. (1997) J. Neurochem. 69:1738.
- Wang, L. et al. (2002) Biochemistry 41:12569.
- Banik, U. et al. (1997) J. Biol. Chem. 272:26219.
Alternate Names
TPH-1, TPRH
Gene Symbol
TPH1
Additional Tryptophan Hydroxylase 1/TPH-1 Products
Product Documents for Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
For research use only
Citations for Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
Customer Reviews for Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody
There are currently no reviews for this product. Be the first to review Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody and earn rewards!
Have you used Phospho-Tryptophan Hydroxylase 1/TPH‑1 (S58) Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
