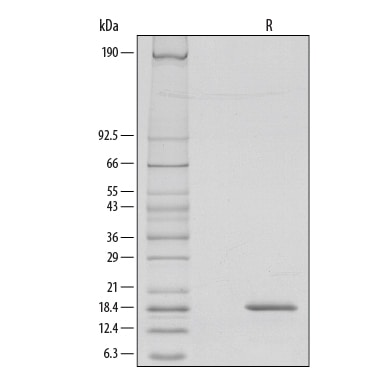
In humans, two distinct cDNA clones for G-CSF, encoding 207 and 204 amino acid precursor proteins, have been isolated. Both proteins have a 30 amino acid signal peptide and have identical amino acid sequences except for a three amino acid insertion (deletion) at the 35th amino acid residue from the N-terminus of the mature protein. Human G-CSF is 73% identical at the amino acid level to murine G-CSF and the two proteins show species cross-reactivity.
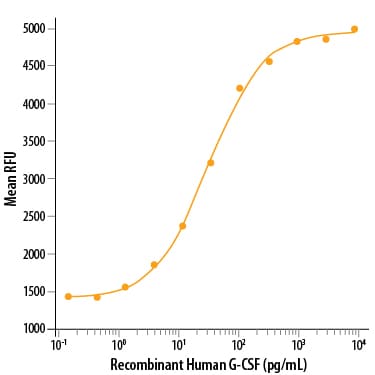
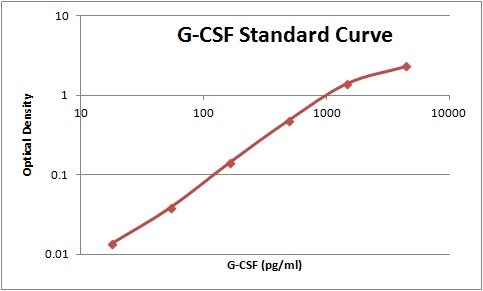
In vitro, G-CSF stimulates growth, differentiation and functions of cells from the neutrophil lineage. It also has blast cell growth factor activity and can synergize with IL-3 to shorten the Go period of early hematopoietic progenitors. Consistent with its in vitro functions, G-CSF has been found to play important roles in defense against infection, in inflammation and repair, and in the maintenance of steady state hematopoiesis.