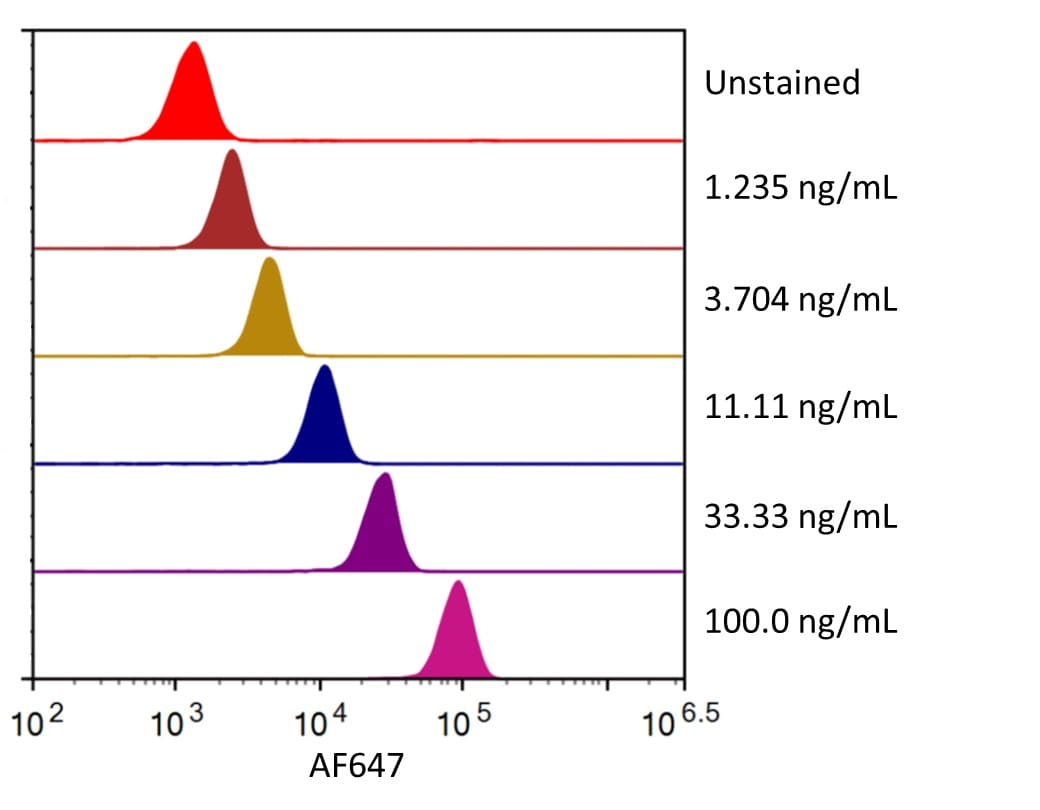
PD-L1, also known as B7-H1, PDL1, is one of the ligands for PD-1 and plays a critical role in the regulation of T cell immunity (1-6). The PD-1:PD-L1 interaction initiates a negative signaling cascade in T cells leading to inhibition of T cell activation (2, 5, 7, 8). PD-L1 provides a molecular stop signal to the adaptive immune system helping to distinguish between self and foreign antigens. PD-L1 also plays a role in the development of immune tolerance by promoting T cell anergy (1, 5) and enhancing regulatory T cell development (8). In addition, PD-L1 favors the development of anti-inflammatory IL-10 and IL-22 producing dendritic cells (7, 9) and inhibits the development of Th17 cells (8). Many cancers exhibit upregulated PD-L1 protein expression, and several cancers with high levels of PD-L1 have been associated with increased tumor aggressiveness and poor prognosis. Using new therapeutics that block the PD-L1:PD-1 interaction has proven successful in the clinic for many cancer types and has sparked great interest in the field of cancer immunotherapy.
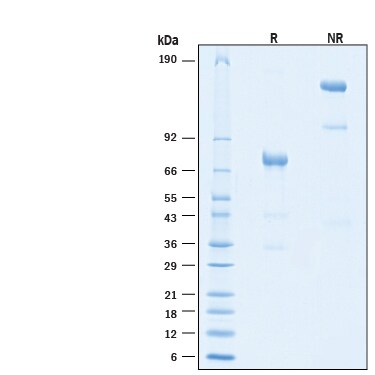
The PD-L1 protein is an approximately 65 kDa transmembrane glycoprotein belonging to the B7 family of immune regulatory molecules (10). Mature human PD-L1 protein consists of a 220 amino acid (aa) extracellular domain (ECD) with two immunoglobulin-like domains, a 21 aa transmembrane segment, and a 31 aa cytoplasmic domain (11). Within the ECD, human PD-L1 shares 73% and 74% aa sequence identity with mouse and rat B7-H1, respectively. Alternative splicing generates additional isoforms that either lack the first Ig-like domain or are truncated within the second Ig-like domain (12). PD-L1 is expressed on inflammatory-activated immune cells including macrophages, T cells, and B cells (10, 13, 14, 16) keratinocytes (9, 11), endothelial and intestinal epithelial cells (2, 9), as well as a variety of carcinomas and melanoma (12, 16).