Best Seller
Recombinant Viral B18R Protein, CF
R&D Systems | Catalog # 8185-BR
Type I IFN Inhibitor

Loading...
Key Product Details
- R&D Systems HEK293-derived Recombinant Viral B18R Protein (8185-BR)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
HEK293
Accession Number
Structure / Form
Disulfide-linked homodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
Human embryonic kidney cell, HEK293-derived viral B18R protein
| MD | Human IgG1 (Pro100-Lys300) |
IEGR | Viral B18R (His20-Glu351) Accession # P25213 |
| N-terminus | C-terminus | ||
Purity
>90%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met-Asp-Pro100
Predicted Molecular Mass
65 kDa (monomer)
SDS-PAGE
80-95 kDa, reducing conditions
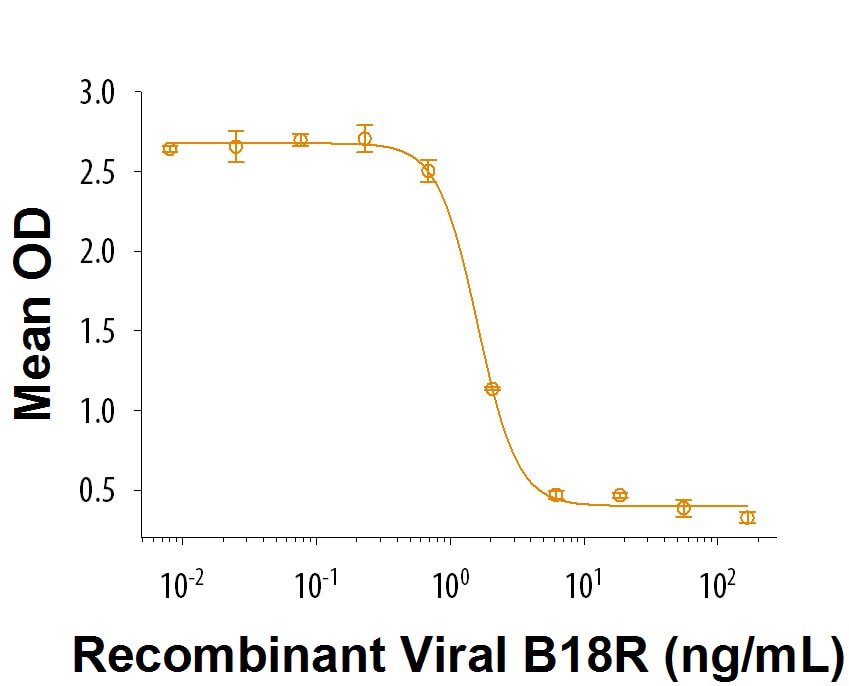
Activity
Measured by its ability to inhibit Type-I IFN-mediated anti-viral activity. Symons, J.A. et al. (1995) Cell 81:551.
The ED50 for this effect, as measured by inhibition of Recombinant Human IFN‑ alpha 2 (Catalog # 11105-1) , is 0.3-1.8 ng/mL.
The ED50 for this effect, as measured by inhibition of Recombinant Human IFN‑ alpha 2 (Catalog # 11105-1) , is 0.3-1.8 ng/mL.
Scientific Data Images for Recombinant Viral B18R Protein, CF
B18R Inhibits Type-I IFN-mediated anti-viral activity.
The viral type I IFN receptor, B18R, inhibits the anti-viral activity of Recombinant Human IFN-alpha 2. The ED50 for this effect is 0.3-1.8 ng/mL.Formulation, Preparation, and Storage
8185-BR
| Formulation | Lyophilized from a 0.2 μm filtered solution in PBS. |
| Reconstitution | Reconstitute at 500 μg/mL in PBS.
Loading...
|
| Shipping | The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: B18R
References
- Smith, G.L. et al. (2013) J. Gen. Virol. 94:2367.
- Perdiguero, B. and M. Esteban (2009) J. Interferon Cytokine Res. 29:581.
- Gomez, C.E. et al. (2012) J. Virol. 86:5026.
- Goebel, S.J. et al. (1990) Virology 179:247.
- Smith, G.L. and Y.S. Chan (1991) J. Gen. Virol. 72:511.
- Alcami, A. et al. (2000) J. Virol. 74:11230.
- Symons, J.A. et al. (1995) Cell 81:551.
- Colamonici, O.R. et al. (1995) J. Biol. Chem. 270:15974.
- Waibler, Z. et al. (2009) J. Virol. 83:1563.
- Warren, L. et al. (2010) Cell Stem Cell. 7:618.
Long Name
Soluble Interferon alpha/beta Receptor B18
Entrez Gene IDs
3707577 (Viral)
Gene Symbol
B19R
UniProt
Additional B18R Products
Product Documents for Recombinant Viral B18R Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Viral B18R Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Viral B18R Protein, CF
Customer Reviews for Recombinant Viral B18R Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Viral B18R Protein, CF and earn rewards!
Have you used Recombinant Viral B18R Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
