Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
517 results for "S100P" in Products
S100P Products
S100 family proteins are small (10-12 kDa), acidic proteins that are found exclusively in vertebrates. They belong to a highly conserved group of EF-hand calcium-binding molecules that consists of more than 25 members. They are the only EF-hand proteins that form both homodimeric and heterodimeric complexes. Although structurally related, their expression patterns and functions are usually distinct.
Human S100P is a 22 kDa, homodimeric member of the S100 family of calcium-binding proteins. The ...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #357517 |
| Applications: | KO, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #357514 |
| Applications: | IHC |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #S100P/4386R |
| Applications: | IHC |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #S100P/4386R |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #S100P/7373 |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #S100P/7373 |
| Applications: | IHC |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #116 |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #OTI1A4 |
| Applications: | WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #OTI1A4 |
| Applications: | WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #357517 |
| Applications: | KO, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #357517 |
| Applications: | KO, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #357517 |
| Applications: | KO, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #357517 |
| Applications: | KO, WB, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #357517 |
| Applications: | KO, WB, ICC/IF |
Loading...


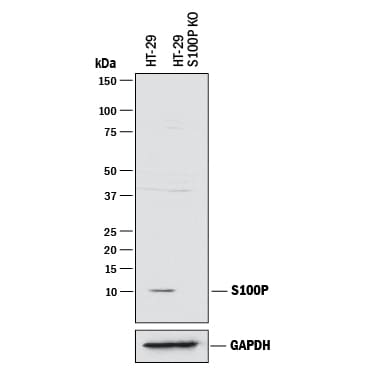
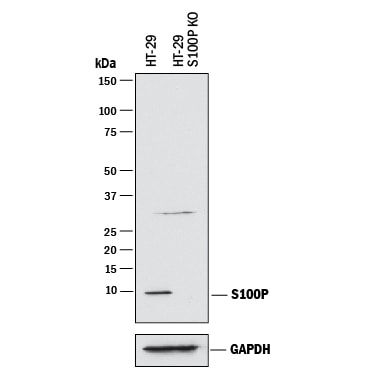
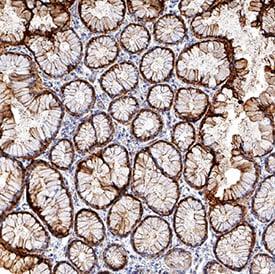

![Immunohistochemistry-Paraffin: S100P Antibody (S100P/4386R) [NBP3-07746] Immunohistochemistry-Paraffin: S100P Antibody (S100P/4386R) [NBP3-07746]](https://resources.rndsystems.com/images/products/S100P-Antibody-S100P-4386R-Immunohistochemistry-Paraffin-NBP3-07746-img0001.jpg)

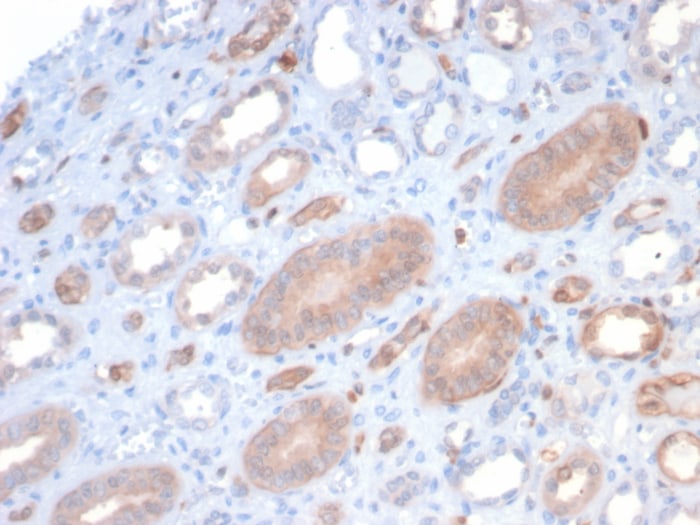
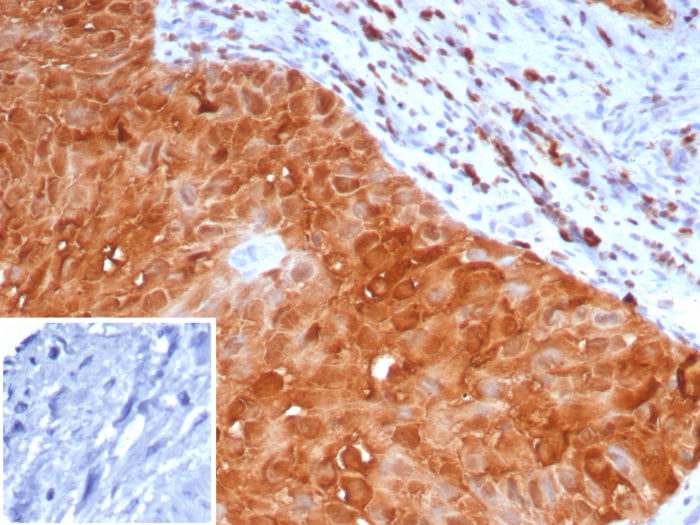
![Immunohistochemistry-Paraffin: S100P Antibody (116) [NBP2-90135] Immunohistochemistry-Paraffin: S100P Antibody (116) [NBP2-90135]](https://resources.rndsystems.com/images/products/S100P-Antibody-116-Immunohistochemistry-Paraffin-NBP2-90135-img0002.jpg)
![Immunohistochemistry-Paraffin: S100P Antibody [NBP2-99514] Immunohistochemistry-Paraffin: S100P Antibody [NBP2-99514]](https://resources.rndsystems.com/images/products/S100P-Antibody-Immunohistochemistry-Paraffin-NBP2-99514-img0003.jpg)
![Immunohistochemistry-Paraffin: S100P Antibody [NBP1-89541] Immunohistochemistry-Paraffin: S100P Antibody [NBP1-89541]](https://resources.rndsystems.com/images/products/S100P-Antibody-Immunohistochemistry-Paraffin-NBP1-89541-img0010.jpg)
![Western Blot: S100P Antibody (OTI1A4) [NBP2-01900] Western Blot: S100P Antibody (OTI1A4) [NBP2-01900]](https://resources.rndsystems.com/images/products/S100P-Antibody-1A4-Western-Blot-NBP2-01900-img0001.jpg)
![Western Blot: S100P Antibody (OTI1A4)Azide and BSA Free [NBP2-73972] Western Blot: S100P Antibody (OTI1A4)Azide and BSA Free [NBP2-73972]](https://resources.rndsystems.com/images/products/S100P-Antibody-OTI1A4-Azide-and-BSA-Free-Western-Blot-NBP2-73972-img0001.jpg)