Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
68 results for "Siglec-E" in Products
Siglec-E Products
Siglecs are I-type (Ig-type) lectins belonging to the Ig superfamily. They are characterized by an N-terminal, Ig-like V-type domain that mediates sialic acid binding, followed by varying numbers of Ig-like C2-type domains (2 to 17), a single transmembrane region, and a cytoplasmic tail. The siglecs can be broadly classified into two subgroups: Siglecs-1, -2, and -4, and a Siglec-3/CD33-related subgroup (Siglecs-3, and -5 through -13 in primates) defined by sequence similarity and clustered ...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Source: | NS0 |
| Accession #: | AAH23280 |
| Applications: | Bioactivity |
Loading...
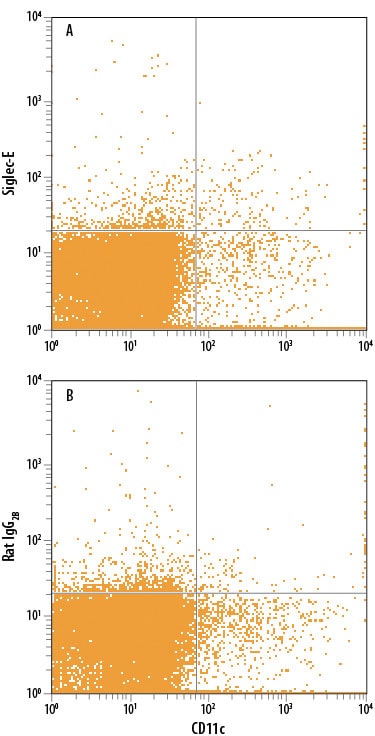
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow, CyTOF-ready |
Loading...
| Reactivity: | Mouse |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Rat IgG2B Monoclonal Clone #750620 |
| Applications: | Flow |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
Loading...


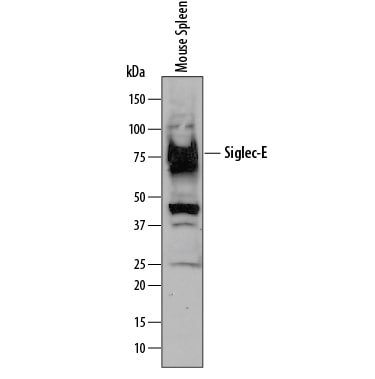
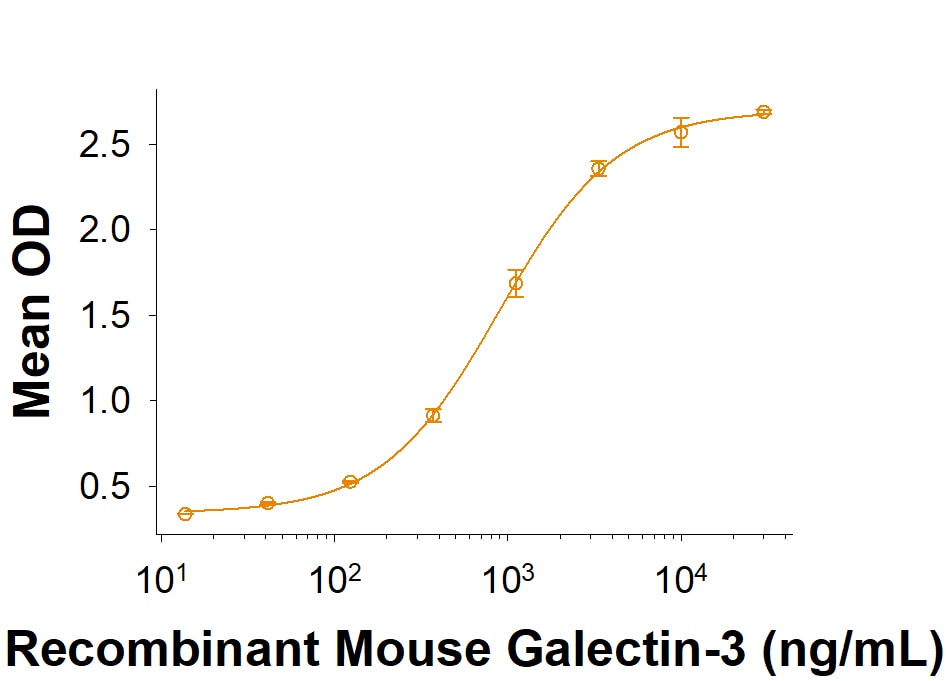
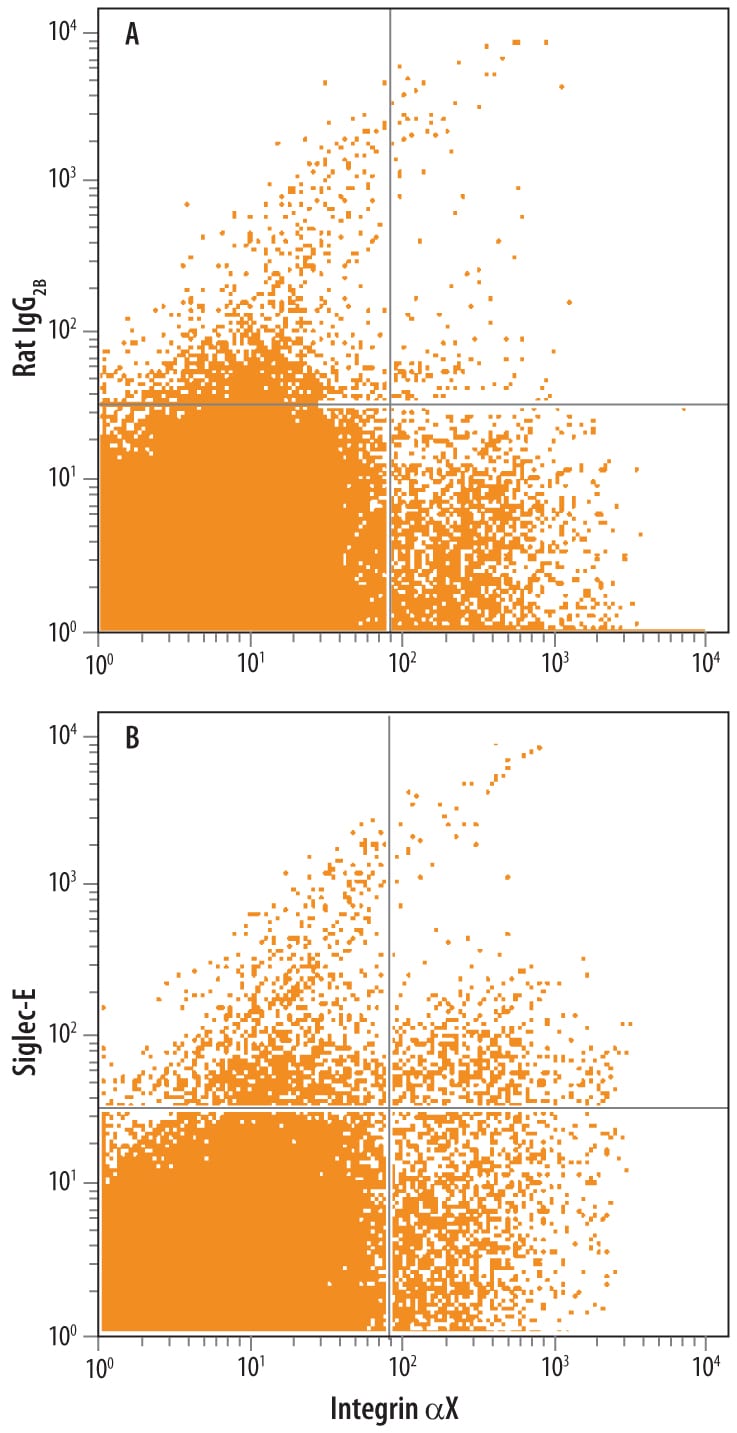

![Product Feature: CoraFluor Probes for TR-FRET Siglec-E Antibody [CoraFluor™ 1]](https://resources.rndsystems.com/images/products/af5806cl1_goat-siglec-e-pab-corafluor-1-8920251633255.png)