230 results for "DDR1" in 产品
DDR1 Products
DDR1, also known as CAK, CD167a, RTK6, and TrkE, is a 120-140 kDa type I transmembrane glycoprotein that belongs to the discoidin-like domain containing subfamily of receptor tyrosine kinases and serve as receptors for collagen. DDR1 is expressed on epithelial tissues, activated monocytes and neutrophils, and in several cancers. Compared to isoform DDR1b, DDR1a lacks 37 aa’s that include a Shc-interacting NPxY motif in the cytoplasmic juxtamembrane region. Two additional kinase ...
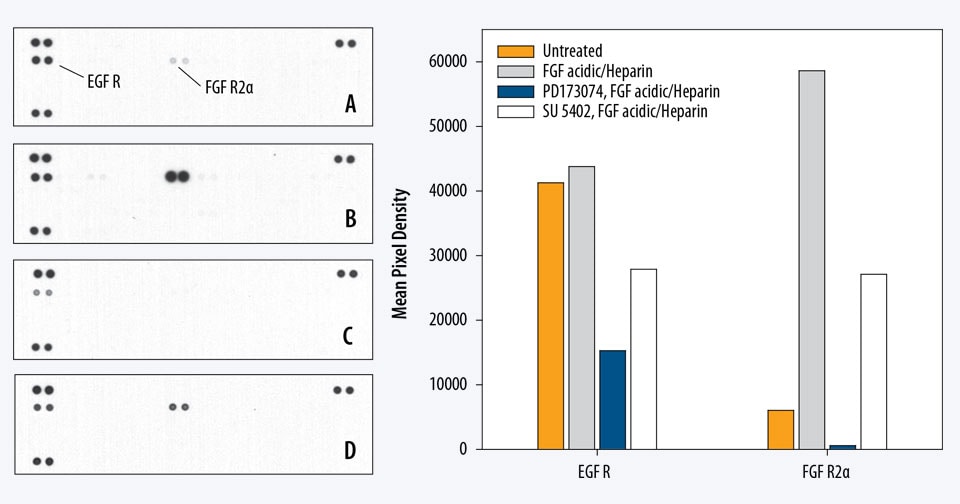
Contains 4 membranes - each spotted in duplicate with 49 different RTK antibodies
| Reactivity: | Human |
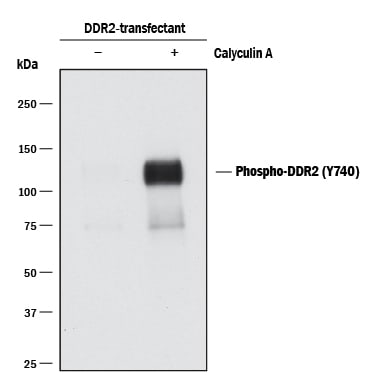
Recombinant Monoclonal Antibody.
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #1119D |
| Applications: | WB, Simple Western |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB, Simple Western |
| Reactivity: | Human |
| Source: | NS0 |
| Accession #: | Q5ST11 |
| Applications: | Binding Activity |
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
Potent and selective v-Abl tyrosine kinase inhibitor; also inhibits PDGFR and c-kit
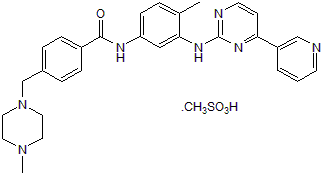
| Alternate Names: | Gleevec,CGP 57148B |
| Chemical Name: | 4-[(4-Methyl-1-piperazinyl)methyl]-N-[4-methyl-3-[[4-(3-pyridinyl)-2-pyrimidinyl]amino]phenyl]benzamide methanesulfonate |
| Purity: | ≥98% |
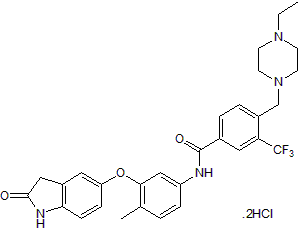
Selective DDR1 inhibitor
| Chemical Name: | N-[3-[(2,3-Dihydro-2-oxo-1H-indol-5-yl)oxy]-4-methylphenyl]-4-[(4-ethyl-1-piperazinyl)methyl]-3-(trifluoromethyl)benzamide dihydrochloride |
| Purity: | ≥98% |
| Reactivity: | Mouse |
| Source: | NS0 |
| Accession #: | Q03146 |
| Applications: | Bioactivity |
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #290420 |
| Applications: | IP |
| Reactivity: | Human |
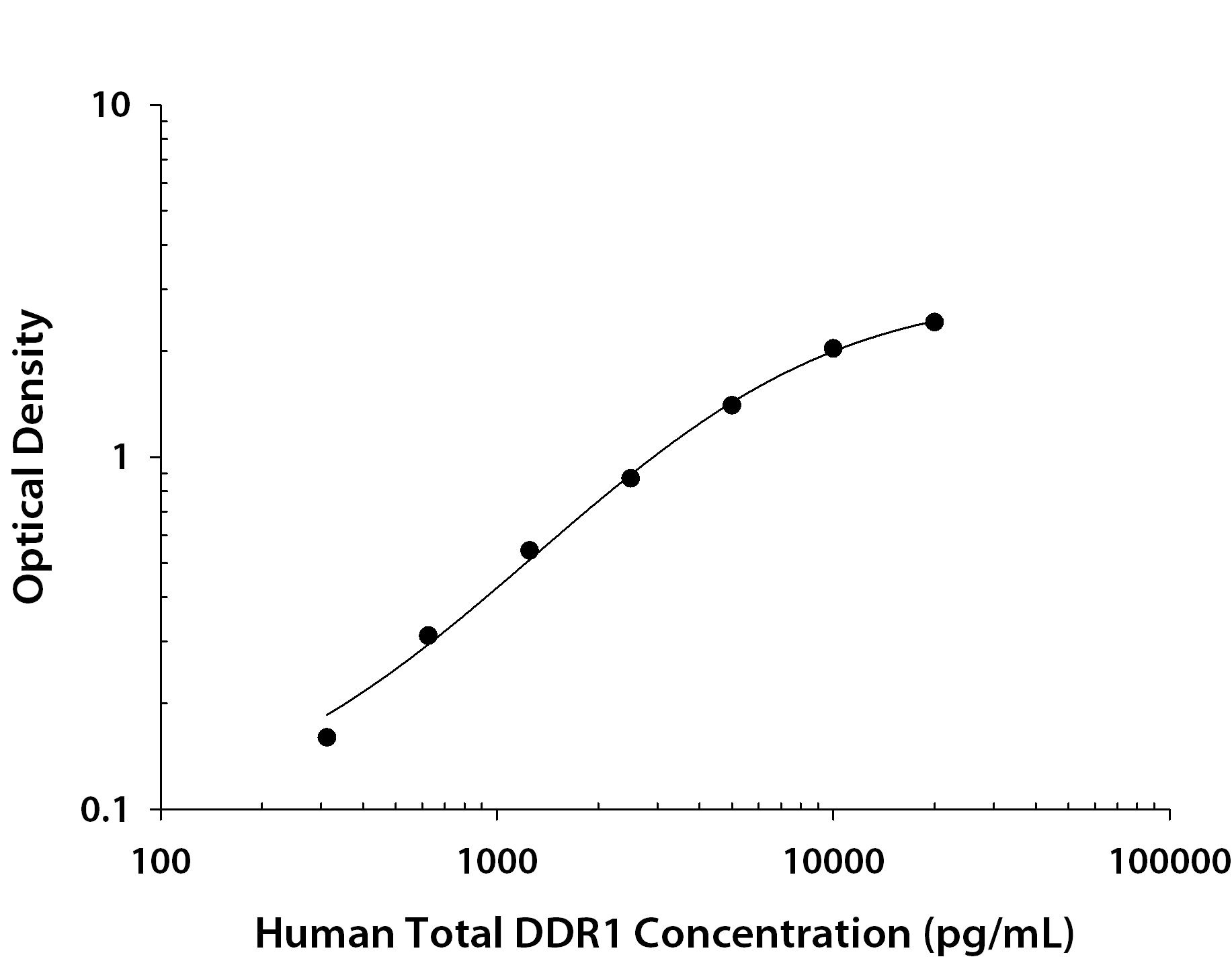
| Assay Range: | 312.5-20000 pg/mL |
| Applications: | ELISA |
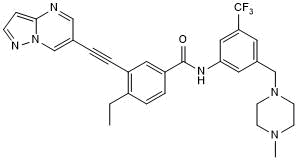
Potent and selective DDR1 inhibitor; orally bioavailable
| Chemical Name: | 4-Ethyl-N-[3-[(4-methyl-1-piperazinyl)methyl]-5-(trifluoromethyl)phenyl]-3-(2-pyrazolo[1,5-a]pyrimidin-6-ylethynyl)benzamide |
| Purity: | ≥98% |
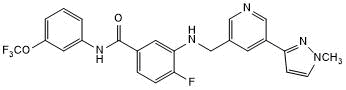
Potent and selective discoidin domain receptor 1/2 inhibitor
| Chemical Name: | 4-Fluoro-3-[[[5-(1-methyl-1H-pyrazol-3-yl)-3-pyridinyl]methyl]amino]-N-[3-(trifluoromethoxy)phenyl]benzamide |
| Purity: | ≥98% |
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #4F2C12 |
| Applications: | WB, ELISA, Flow |
| Reactivity: | Human |
| Applications: | ELISA |
| Reactivity: | Human, Porcine, Bat, Bovine, Canine, +3 More |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB |





![Western Blot: DDR1 Antibody [NBP1-33134] Western Blot: DDR1 Antibody [NBP1-33134]](https://resources.rndsystems.com/images/products/DDR1-Antibody-Western-Blot-NBP1-33134-img0004.jpg)

![Western Blot: DDR1 Antibody (4F2C12)BSA Free [NBP2-61692] Western Blot: DDR1 Antibody (4F2C12)BSA Free [NBP2-61692]](https://resources.rndsystems.com/images/products/DDR1-Antibody-4F2C12-Western-Blot-NBP2-61692-img0003.jpg)
![Immunohistochemistry-Paraffin: DDR1 Antibody - BSA Free [NB110-39011] Immunohistochemistry-Paraffin: DDR1 Antibody - BSA Free [NB110-39011]](https://resources.rndsystems.com/images/products/DDR1-Antibody---BSA-Free-Immunohistochemistry-Paraffin-NB110-39011-img0002.jpg)
![Western Blot: DDR1 Antibody [NBP2-16123] Western Blot: DDR1 Antibody [NBP2-16123]](https://resources.rndsystems.com/images/products/DDR1-Antibody-Western-Blot-NBP2-16123-img0004.jpg)