470 results for "S100A8" in 产品
S100A8 Products
S100A8 (also MRP8 and calgranulin A) is a 10 kDa member of the S100 family, EF-hand superfamily of Ca-binding proteins. It is produced by neutrophils and monocytes, and forms Ca2+-dependent heterodimer/heterotetramer complexes (termed calprotectin) with S100A9. It functions both intracellularly and extracellularly, where it binds to RAGE and CD36. Human S100A8 is 93 amino acids (aa) in length. It contains two EF-hand motifs (aa 12 - 47 and 46 - 81) and one high-affinity Ca2+-binding site (aa 59 ...
| Reactivity: | Human |
Build your own Luminex Assay with our Luminex Assay Customization Tool
| Reactivity: | Mouse |
Build your own Luminex Assay with our Luminex Assay Customization Tool
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ICC/IF, Simple Western |
| Reactivity: | Human |
| Assay Range: | 31.2-2000 pg/mL |
| Applications: | ELISA |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #749916 |
| Applications: | IHC, WB |
| Reactivity: | Mouse |
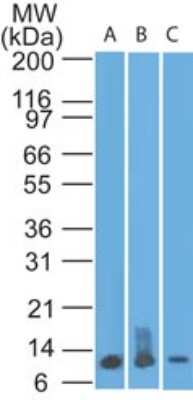
| Details: | Rat IgG2B Monoclonal Clone #335806 |
| Applications: | WB, ICC/IF |
Analyzed by SEC-MALS
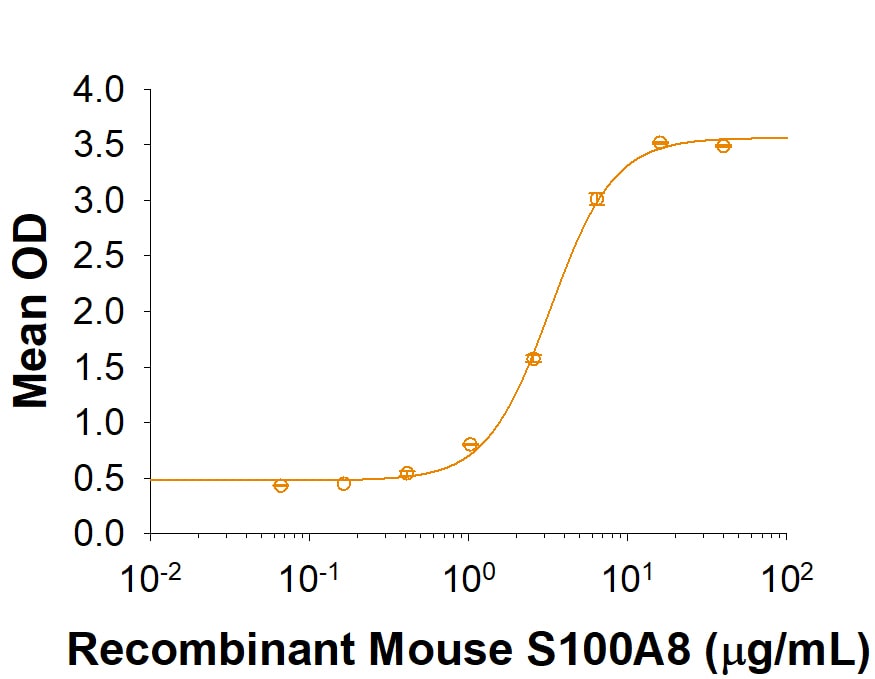
| Reactivity: | Mouse |
| Source: | E. coli |
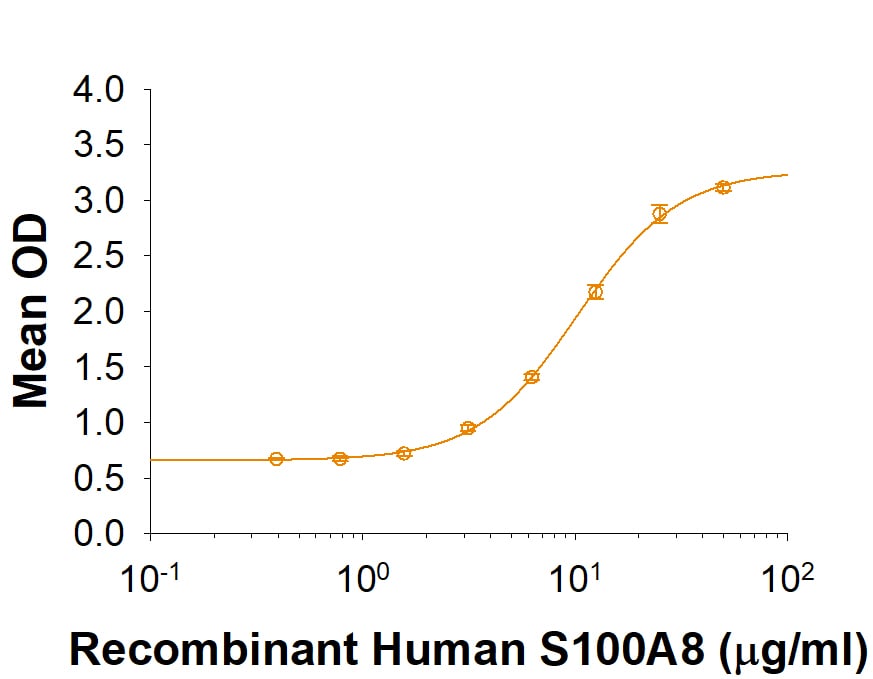
| Applications: | Bioactivity |
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB, Flow, ICC/IF, Simple Western, CyTOF-ready |
| Reactivity: | Mouse |
| Assay Range: | 31.2-2000 pg/mL |
| Applications: | ELISA |
| Reactivity: | Mouse |
| Details: | Rat IgG2b Lambda Monoclonal Clone #63N13G5 |
| Applications: | IHC, WB, Flow |
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #48M7C7 |
| Applications: | IHC, WB, Flow, ICC/IF |
Recombinant Monoclonal Antibody.
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #1099F |
| Applications: | IHC, Dual ISH-IHC, ICC/IF |
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | P05109 |
| Applications: | Bioactivity |
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #48M7C7 |
| Applications: | IHC, WB, Flow |
| Reactivity: | Mouse |
| Details: | Rat IgG Lambda Monoclonal Clone #63N13G5 |
| Applications: | IHC, WB, Flow |
| Reactivity: | Mouse |
| Source: | E. coli |
| Accession #: | P27005 |
| Applications: | Bioactivity |
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF |
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #48M7F9 |
| Applications: | IHC, WB |
| Reactivity: | Human, Mouse |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #48M7F9 |
| Applications: | IHC, WB |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #900027 |
| Applications: | IHC, ELISA, Flow, CyTOF-ready |
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #900028 |
| Applications: | ELISA, Flow, CyTOF-ready |
| Reactivity: | Human |
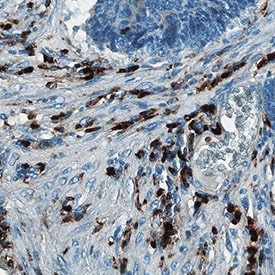
| Details: | Mouse IgG1 Monoclonal Clone #CL11171 |
| Applications: | IHC |
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #CL11171 |
| Applications: | IHC |
| Reactivity: | Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ICC/IF |
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #203 |
| Applications: | WB, IP |




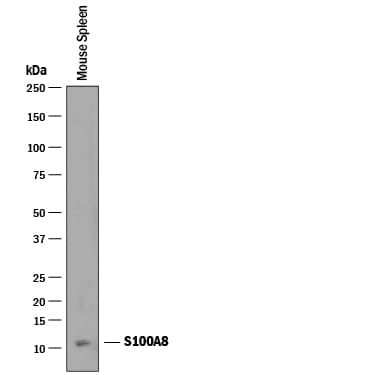

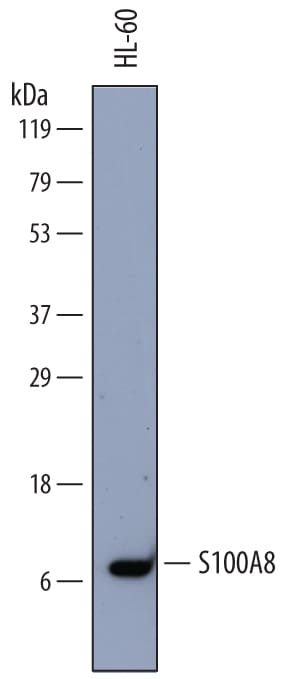

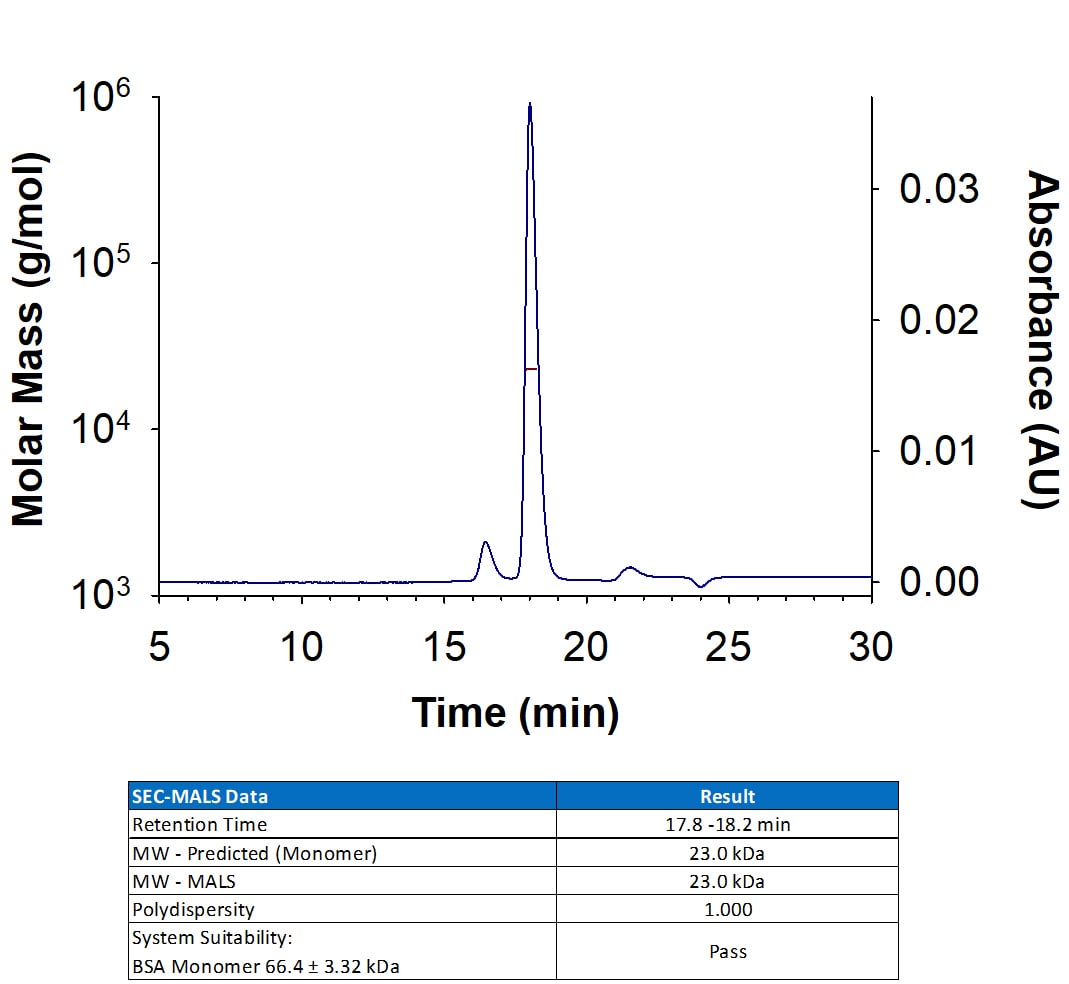



![Western Blot: S100A8 Antibody (63N13G5)BSA Free [NBP2-27067] Western Blot: S100A8 Antibody (63N13G5)BSA Free [NBP2-27067]](https://resources.rndsystems.com/images/products/S100A8-Antibody-63N13G5-Western-Blot-NBP2-27067-img0006.jpg)
![Western Blot: S100A8 Antibody (48M7C7)Azide Free [NBP2-25269] Western Blot: S100A8 Antibody (48M7C7)Azide Free [NBP2-25269]](https://resources.rndsystems.com/images/products/S100A8-A9-Antibody-48M7C7-Azide-Free-Western-Blot-NBP2-25269-img0001.jpg)
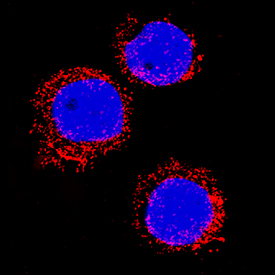
![Western Blot: S100A8 Antibody (48M7C7) [NBP2-25256] Western Blot: S100A8 Antibody (48M7C7) [NBP2-25256]](https://resources.rndsystems.com/images/products/S100A8-A9-Antibody-48M7C7-Western-Blot-NBP2-25256-img0001.jpg)
![Western Blot: S100A8 Antibody (63N13G5)Azide Free [NBP2-25273] Western Blot: S100A8 Antibody (63N13G5)Azide Free [NBP2-25273]](https://resources.rndsystems.com/images/products/S100A8-Antibody-63N13G5-Azide-Free-Western-Blot-NBP2-25273-img0001.jpg)
![Immunocytochemistry/ Immunofluorescence: S100A8 Antibody - BSA Free [NBP1-90314] S100A8 Antibody - BSA Free Immunocytochemistry/ Immunofluorescence: S100A8 Antibody - BSA Free [NBP1-90314]](https://resources.rndsystems.com/images/products/nbp1-90314_rabbit-polyclonal-s100a8-antibody-29520251675316.jpg)
![Western Blot: S100A8 Antibody (48M7F9) [NBP2-25257] Western Blot: S100A8 Antibody (48M7F9) [NBP2-25257]](https://resources.rndsystems.com/images/products/S100A8-A9-Antibody-48M7F9-Western-Blot-NBP2-25257-img0001.jpg)
![Immunohistochemistry-Paraffin: S100A8 Antibody (CL11171) [NBP3-07985] Immunohistochemistry-Paraffin: S100A8 Antibody (CL11171) [NBP3-07985]](https://resources.rndsystems.com/images/products/S100A8-Antibody-CL11171-Immunohistochemistry-Paraffin-NBP3-07985-img0004.jpg)
![Immunohistochemistry-Paraffin: S100A8 Antibody [NBP3-43984] S100A8 Antibody (CL11171) - Azide and BSA Free Immunohistochemistry-Paraffin: S100A8 Antibody [NBP3-43984]](https://resources.rndsystems.com/images/products/nbp3-43984_mouse-s100a8-mab-cl11171-azide-and-bsa-free-282202512163679.jpg)
![Western Blot: S100A8 Antibody (203) [NBP2-89806] Western Blot: S100A8 Antibody (203) [NBP2-89806]](https://resources.rndsystems.com/images/products/S100A8-Antibody-203-Western-Blot-NBP2-89806-img0001.jpg)