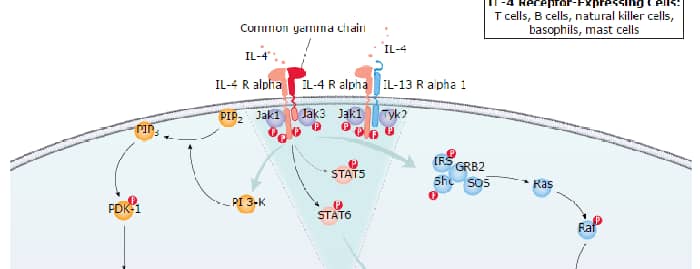
Two type 1 membrane proteins belonging to the hematopoietin receptor family have been cloned and shown to bind IL-13 with differing affinities. The lower affinity IL-13 binding protein, previously designated IL-13 R alpha, or NR4, is now referred to as IL-13 R alpha 1. The high-affinity IL-13 binding protein, previously also designated IL-13 R or IL-13 R alpha ', is now referred to as IL-13 R alpha 2.
The human IL-13 R alpha 1 was originally cloned based on sequence homology to the mouse IL-13 R alpha 1. The IL-13 R alpha 1 cDNA encodes a 427 amino acid (aa) precursor protein with a putative 21 aa signal peptide, a 324 aa extracellular domain, a 23 aa transmembrane region and a 59 aa cytoplasmic tail. Human and mouse IL-13 R alpha 1 share 76% aa sequence identity. The extracellular domain of IL-13 R alpha 1 is also closely related to that of IL-13 R alpha 2. IL‑13 R alpha 1 has been shown to combine with the IL‑4 R alpha to form a high-affinity receptor complex capable of transducing an IL-13-dependent proliferative signal. The role of IL-13 R alpha 2 in IL-13 signaling remains to be elucidated.