NMDA (N-Methyl D-Aspartate) receptors are members of the glutamate receptor family of ligand-gated ion channels. The functional NMDA receptor (NMDAR) is a 650-850 kDa heteromultimer of at least two NR2 (NMDAR2) subunits and two NR1 subunits. NR2 subunits determine overall NMDAR characteristics and mediate NMDAR clustering and synaptic localization through cytoplasmic interaction with PSD-95/SAP90 family members. Upon glutamate binding to NR2, and glycine binding to NR1, the NMDA channel is opened, allowing calcium and sodium influx into the cell. There are four genes that code for NR2 subunits (NR2A-D). The NR2B subunit generates a high conductance NMDAR. Human NR2B (also NR3;GRIN2B and GluR epsilon 2) is a 180-200 kDa, 1458 amino acid (aa), three transmembrane (TM) glycoprotein that contains a 531 aa N-terminal extracellular domain (ECD), and a 646 aa cytoplasmic region. At least three tyrosines are known to be phosphorylated by Fyn in the cytoplasmic region. These include Y1252, Y1336 and Y1472. While much is known about Y1472, little information is available about the consequences of Y1252 and Y1336 phosphorylation.
Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
R&D Systems | Catalog # PPS056

Discontinued Product
PPS056 has been discontinued.
View all GRIN2B/NMDAR2B products.
Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Rat, Primate
Cited:
Mouse
Applications
Validated:
Western Blot
Cited:
Western Blot, Immunocytochemistry
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Phosphopeptide corresponding to amino acid residues surrounding the phospho-Y1252 of NMDA R, NR2B Subunit
Specificity
Human, non-human primates, and rat ~180 kDa NMDA Receptor NR2B subunit phosphorylated at Y1252 in Western blots.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
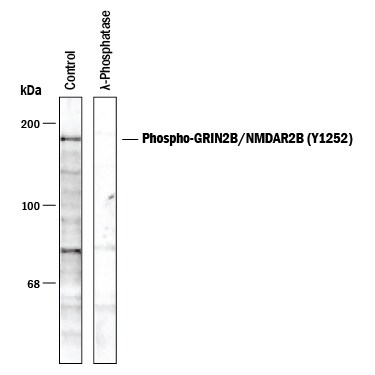
Detection of GRIN2B/NMDAR2B (Y1252) by Western Blot.
Western blot of rat hippocampal lysate showing specific immunolabeling of the approximately 180 kDa NR2B subunit of the NMDAR phosphorylated at Y1252 (Control). The phosphospecificity of this labeling is shown in the second lane ( lambda-phosphatase: lambda PPase). The blot is identical to the control except that it was incubated in lambda PPase (1200 units for 30 minutes) before being exposed to the Anti-Phospho-NMDA NR2B subunit (Y1252). The immunolabeling is completely eliminated by treatment with lambda PPase.Applications for Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
Application
Recommended Usage
Western Blot
1:1000 dilution
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Formulation
100 μL in 10 mM HEPES (pH 7.5), 150 mM NaCl, 100 μg/mL BSA and 50% glycerol.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
For long-term storage, ≤ -20° C is recommended. Product is stable at ≤ -20° C for at least 1 year.
Background: GRIN2B/NMDAR2B
References
- Stephenson, F.A. (2001) Curr. Drug Targets 2:233.
- Cull-candy, S.G. and D.N. Leszkiewicz (2004) Sci. STKE re16 (2004).
- Prybylowski, K. and R.J. Wenthold (2004) J. Biol. Chem. 279:9673.
- Ishii, T. et al. (1993) J. Biol. Chem. 268:2836.
- Hess, S.D. et al. (1996) J. Pharmacol. Exp. Ther. 278:808.
- Nakazawa, T. et al. (2001) J. Biol. Chem. 276:693.
Long Name
Glutamate Receptor, Ionotropic, N-Methyl-D-Aspartate, Subunit 2B
Alternate Names
NMDA R, NR2B Subunit, NMDAR2B, NR2B
Entrez Gene IDs
2904 (Human)
Gene Symbol
GRIN2B
Additional GRIN2B/NMDAR2B Products
Product Documents for Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
For research use only
Citations for Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
Customer Reviews for Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody
There are currently no reviews for this product. Be the first to review Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody and earn rewards!
Have you used Human/Rat/Primate Phospho-GRIN2B/NMDAR2B (Y1252) Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
