MCT8/SLC16A2 Antibody - BSA Free
Novus Biologicals | Catalog # NBP2-57308

![Immunocytochemistry/ Immunofluorescence: MCT8/SLC16A2 Antibody [NBP2-57308] Immunocytochemistry/ Immunofluorescence: MCT8/SLC16A2 Antibody [NBP2-57308]](https://resources.rndsystems.com/images/products/MCT8-SLC16A2-Antibody-Immunocytochemistry-Immunofluorescence-NBP2-57308-img0001.jpg)
Loading...
Key Product Details
Species Reactivity
Validated:
Human, Mouse
Cited:
Human
Predicted:
Rat (90%). Backed by our 100% Guarantee.
Applications
Validated:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunocytochemistry/ Immunofluorescence
Cited:
Immunohistochemistry, IF/IHC
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Format
BSA Free
Loading...
Product Specifications
Immunogen
This antibody was developed against a recombinant protein corresponding to the following amino acid sequence: LMHQRMFKKEQRDSSKDKMLAPDPDPNGELLPGSPNPEEPI
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for MCT8/SLC16A2 Antibody - BSA Free
Immunocytochemistry/ Immunofluorescence: MCT8/SLC16A2 Antibody [NBP2-57308]
Immunocytochemistry/Immunofluorescence: MCT8/SLC16A2 Antibody [NBP2-57308] - Staining of human cell line U-251 MG shows localization to plasma membrane. Antibody staining is shown in green.Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]
Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] - Staining of human skeletal muscle shows no positivity in striated muscle fibers as expected.Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]
Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] - Staining of mouse embryo E11 shows strong membranous positivity in the developing circumventricular organ.Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]
Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] - Staining of human cerebral cortex shows moderate positivity in neuropil.Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]
Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] - Staining of human kidney shows moderate membranous positivity in cells in tubules.Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]
Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] - Staining of human liver shows moderate to strong membranous positivity in hepatocytes.Immunocytochemistry/ Immunofluorescence: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 and OATP1C1 in the cerebral barriers of the human and macaque motor cortex. Representative brightfield (A,E,I,M) and fluorescence confocal microscope (B–D,F–H,J–L,N–T) photomicrographs showing immunostaining for MCT8 (A–H,Q–T) and OATP1C1 (I–P) in blood vessels of human and macaque motor cortex. (A,E) show strong MCT8 staining in the endothelial layer of small vessels and capillaries (white arrowheads) in humans and macaques, respectively. (B–D,F–H) show colocalization of MCT8 (green) with the endothelial markers UEA-I (red) and ENG (red). (I,M) show weak OATP1C1 staining in small vessels (white arrowheads) and venules (black arrowheads). The red arrowhead in M points to a glial cell positive for OATP1C1 with processes surrounding the capillary. (J–L,N–P) show colocalization of OATP1C1 (green) with UEA-I (red) and endoglin (red) in a few blood vessels of the human and macaque motor cortex, respectively. White arrowheads point to vessels. In both human and macaque brain tissues, MCT8-immunopositive capillaries are much more abundant than OATP1C1-immunopositive capillaries, which can only be seen occasionally. Moreover, (Q–T) show MCT8-expressing pericytes (white arrowheads) in the human motor cortex that in addition to express MCT8 (green), also show the pericyte biomarker PDGFR-beta (pink), but not the endothelial marker UEA-I (red). Counterstaining with DAPI (blue) shows nuclei of all cells. ENG: endoglin, UEA-I: Ulex Europaeus Agglutinin-I. PDGFR-beta : platelet-derived growth factor receptor beta. Scale bar = 100 μm (A,E,I,M), 25 μm (B–D), 50 μm (J–L), 120 μm (F–H,N–P), and 12 μm (Q–T). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 and OATP1C1 in the cerebral barriers of the human and macaque motor cortex. Representative brightfield (A,E,I,M) and fluorescence confocal microscope (B–D,F–H,J–L,N–T) photomicrographs showing immunostaining for MCT8 (A–H,Q–T) and OATP1C1 (I–P) in blood vessels of human and macaque motor cortex. (A,E) show strong MCT8 staining in the endothelial layer of small vessels and capillaries (white arrowheads) in humans and macaques, respectively. (B–D,F–H) show colocalization of MCT8 (green) with the endothelial markers UEA-I (red) and ENG (red). (I,M) show weak OATP1C1 staining in small vessels (white arrowheads) and venules (black arrowheads). The red arrowhead in M points to a glial cell positive for OATP1C1 with processes surrounding the capillary. (J–L,N–P) show colocalization of OATP1C1 (green) with UEA-I (red) and endoglin (red) in a few blood vessels of the human and macaque motor cortex, respectively. White arrowheads point to vessels. In both human and macaque brain tissues, MCT8-immunopositive capillaries are much more abundant than OATP1C1-immunopositive capillaries, which can only be seen occasionally. Moreover, (Q–T) show MCT8-expressing pericytes (white arrowheads) in the human motor cortex that in addition to express MCT8 (green), also show the pericyte biomarker PDGFR-beta (pink), but not the endothelial marker UEA-I (red). Counterstaining with DAPI (blue) shows nuclei of all cells. ENG: endoglin, UEA-I: Ulex Europaeus Agglutinin-I. PDGFR-beta : platelet-derived growth factor receptor beta. Scale bar = 100 μm (A,E,I,M), 25 μm (B–D), 50 μm (J–L), 120 μm (F–H,N–P), and 12 μm (Q–T). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 in pyramidal neurons of the human and macaque motor cortex. Representative brightfield photomicrographs show immunostaining for MCT8 in layers III (A), V (B) and VI (C) of the human motor cortex, and layers II (G), III (H), and V (I) of the macaque motor cortex. White arrowheads point to pyramidal neurons with immunopositive signal in the soma, apical and basal dendrites. Black arrowheads point to smaller cells with the morphology of interneurons. (D–F) (human) and (J–L) (macaque) show confocal microscope images from double-stained sections for MCT8 (green) and the pyramidal neuron markers SMI-32 (red) for 200 kDa neurofilament protein or neurogranin/RC3 (red), respectively. White arrowheads point to pyramidal neurons. Counterstaining with DAPI (blue) shows nuclei of all cells. Note that in humans, the MCT8 signal is located mainly in the pyramidal cell membrane, while in macaques it is located in the membrane and the cytoplasm. Scale bar = 50 μm (A), 120 μm (B,I), 25 μm (C–F), 60 μm (G), 62.5 μm (H), and 43.5 μm (J–L). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 in pyramidal neurons of the human and macaque motor cortex. Representative brightfield photomicrographs show immunostaining for MCT8 in layers III (A), V (B) and VI (C) of the human motor cortex, and layers II (G), III (H), and V (I) of the macaque motor cortex. White arrowheads point to pyramidal neurons with immunopositive signal in the soma, apical and basal dendrites. Black arrowheads point to smaller cells with the morphology of interneurons. (D–F) (human) and (J–L) (macaque) show confocal microscope images from double-stained sections for MCT8 (green) and the pyramidal neuron markers SMI-32 (red) for 200 kDa neurofilament protein or neurogranin/RC3 (red), respectively. White arrowheads point to pyramidal neurons. Counterstaining with DAPI (blue) shows nuclei of all cells. Note that in humans, the MCT8 signal is located mainly in the pyramidal cell membrane, while in macaques it is located in the membrane and the cytoplasm. Scale bar = 50 μm (A), 120 μm (B,I), 25 μm (C–F), 60 μm (G), 62.5 μm (H), and 43.5 μm (J–L). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 and OATP1C1 in human and macaque motor cortex astrocytes. Representative brightfield (A–E,I) photomicrographs of sections immunostained for MCT8 (A,C) and OATP1C1 (B,D,E,I) in cortical layer I (A–D) and subjacent white matter (E,I). Confocal microscope images of human (F–H) and macaque (J–O) sections double-labeled for OATP1C1 (green) and GFAP (red). Counterstaining with DAPI (blue) shows nuclei of all cells. Note that there is full colocalization of both markers. White and red arrowheads point to astrocytes. Blue arrowheads point to a blood vessel. GFAP: glial fibrillary acidic protein. Scale bar = 50 μm (A–D,J–L), 110 μm (E,I), 25 μm (F–H), and 78 μm (M–O). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 and OATP1C1 in the cerebral barriers of the human and macaque motor cortex. Representative brightfield (A,E,I,M) and fluorescence confocal microscope (B–D,F–H,J–L,N–T) photomicrographs showing immunostaining for MCT8 (A–H,Q–T) and OATP1C1 (I–P) in blood vessels of human and macaque motor cortex. (A,E) show strong MCT8 staining in the endothelial layer of small vessels and capillaries (white arrowheads) in humans and macaques, respectively. (B–D,F–H) show colocalization of MCT8 (green) with the endothelial markers UEA-I (red) and ENG (red). (I,M) show weak OATP1C1 staining in small vessels (white arrowheads) and venules (black arrowheads). The red arrowhead in M points to a glial cell positive for OATP1C1 with processes surrounding the capillary. (J–L,N–P) show colocalization of OATP1C1 (green) with UEA-I (red) and endoglin (red) in a few blood vessels of the human and macaque motor cortex, respectively. White arrowheads point to vessels. In both human and macaque brain tissues, MCT8-immunopositive capillaries are much more abundant than OATP1C1-immunopositive capillaries, which can only be seen occasionally. Moreover, (Q–T) show MCT8-expressing pericytes (white arrowheads) in the human motor cortex that in addition to express MCT8 (green), also show the pericyte biomarker PDGFR-beta (pink), but not the endothelial marker UEA-I (red). Counterstaining with DAPI (blue) shows nuclei of all cells. ENG: endoglin, UEA-I: Ulex Europaeus Agglutinin-I. PDGFR-beta : platelet-derived growth factor receptor beta. Scale bar = 100 μm (A,E,I,M), 25 μm (B–D), 50 μm (J–L), 120 μm (F–H,N–P), and 12 μm (Q–T). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] -
Expression of MCT8 and OATP1C1 in human and macaque motor cortex astrocytes. Representative brightfield (A–E,I) photomicrographs of sections immunostained for MCT8 (A,C) and OATP1C1 (B,D,E,I) in cortical layer I (A–D) and subjacent white matter (E,I). Confocal microscope images of human (F–H) and macaque (J–O) sections double-labeled for OATP1C1 (green) and GFAP (red). Counterstaining with DAPI (blue) shows nuclei of all cells. Note that there is full colocalization of both markers. White and red arrowheads point to astrocytes. Blue arrowheads point to a blood vessel. GFAP: glial fibrillary acidic protein. Scale bar = 50 μm (A–D,J–L), 110 μm (E,I), 25 μm (F–H), and 78 μm (M–O). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/36834621), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for MCT8/SLC16A2 Antibody - BSA Free
Application
Recommended Usage
Immunocytochemistry/ Immunofluorescence
0.25-2 ug/ml
Immunohistochemistry
1:500 - 1:1000
Immunohistochemistry-Paraffin
1:500 - 1:1000
Application Notes
ICC/IF Fixation Permeabilization: Use PFA/Triton X-100. IHC-P Retrieval method: HIER pH6.
Formulation, Preparation, and Storage
Purification
Affinity purified
Formulation
PBS (pH 7.2) and 40% Glycerol
Format
BSA Free
Preservative
0.02% Sodium Azide
Concentration
Concentrations vary lot to lot. See vial label for concentration. If unlisted please contact technical services.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles.
Background: MCT8/SLC16A2
Long Name
Monocarboxylic Acid Transporter 8 (Solute Carrier Family 16 Member 1)
Alternate Names
AHDS, DXS128, MRX22, SLC16A2, XPCT
Gene Symbol
SLC16A2
Additional MCT8/SLC16A2 Products
Product Documents for MCT8/SLC16A2 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for MCT8/SLC16A2 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for MCT8/SLC16A2 Antibody - BSA Free
Customer Reviews for MCT8/SLC16A2 Antibody - BSA Free
There are currently no reviews for this product. Be the first to review MCT8/SLC16A2 Antibody - BSA Free and earn rewards!
Have you used MCT8/SLC16A2 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways
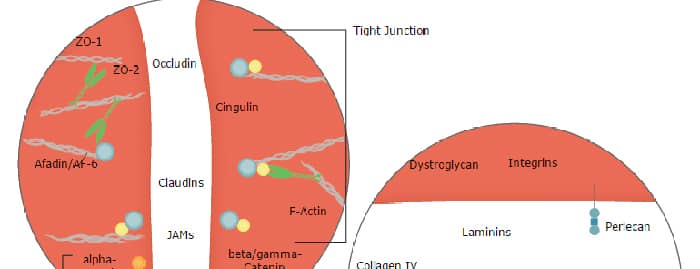
 Blood-Brain Barrier Pathway: Anatomy
Blood-Brain Barrier Pathway: Anatomy

![Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]](https://resources.rndsystems.com/images/products/MCT8-SLC16A2-Antibody-Immunohistochemistry-Paraffin-NBP2-57308-img0018.jpg)
![Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]](https://resources.rndsystems.com/images/products/MCT8-SLC16A2-Antibody-Immunohistochemistry-Paraffin-NBP2-57308-img0014.jpg)
![Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]](https://resources.rndsystems.com/images/products/MCT8-SLC16A2-Antibody-Immunohistochemistry-Paraffin-NBP2-57308-img0015.jpg)
![Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]](https://resources.rndsystems.com/images/products/MCT8-SLC16A2-Antibody-Immunohistochemistry-Paraffin-NBP2-57308-img0016.jpg)
![Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308] Immunohistochemistry-Paraffin: MCT8/SLC16A2 Antibody [NBP2-57308]](https://resources.rndsystems.com/images/products/MCT8-SLC16A2-Antibody-Immunohistochemistry-Paraffin-NBP2-57308-img0017.jpg)
![Immunocytochemistry/ Immunofluorescence: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunocytochemistry-immunofluorescence-13220261935485.jpg)
![Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunohistochemistry-132202619222914.jpg)
![Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunohistochemistry-13220261941185.jpg)
![Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunohistochemistry-132202619434117.jpg)
![Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunohistochemistry-132202619351214.jpg)
![Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunohistochemistry-132202619333620.jpg)
![Immunohistochemistry: MCT8/SLC16A2 Antibody - BSA Free [NBP2-57308] - MCT8/SLC16A2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nbp2-57308_rabbit-polyclonal-mct8-slc16a2-antibody-immunohistochemistry-13220261920722.jpg)