Phospho-CaM Kinase II (T286) Antibody
R&D Systems | Catalog # PPS002

Discontinued Product
PPS002 has been discontinued.
View all CaM Kinase II products.
Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Mouse, Rat, Xenopus
Cited:
Mouse
Applications
Validated:
Western Blot
Cited:
Western Blot, Immunoprecipitation
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Phosphopeptide corresponding to amino acid residues surrounding phospho-T286 of CaM Kinase II
Specificity
Human, mouse, rat, bovine, chicken, Xenopus alpha -CaM Kinase II phosphorylated at T286 in Western blots.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Phospho-CaM Kinase II (T286) Antibody
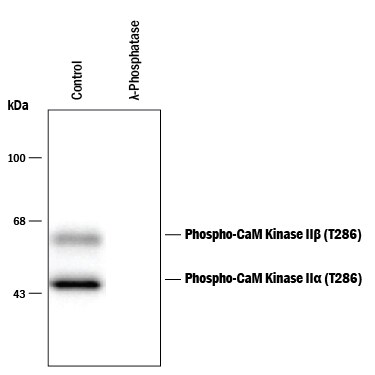
Detection of Phospho-CaM Kinase II (T286) by Western Blot.
Western Blot of rat brain tissue lysate showing specific immunolabeling of the ~50 kDa alpha -CaMKII subunit phosphorylated at T286 and the ~60 kDa beta -CaMKII subunit phosphorylated at T287 (Control). The phosphospecificity of this labeling is demonstrated by treatment with 1200 U of lambda Phosphatase ( lambda -PPase) for 30 minutes before being exposed to the anti-phospho-CaMKII (T286). The immunolabeling is completely eliminated by treatment with lambda -PPase.Applications for Phospho-CaM Kinase II (T286) Antibody
Application
Recommended Usage
Western Blot
1:1000 dilution
Sample: Rat brain tissue
Sample: Rat brain tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Formulation
100 μL in 10 mM HEPES (pH 7.5), 150 mM NaCl, 100 μg/mL BSA and 50% glycerol.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
For long-term storage, ≤ -20° C is recommended. Product is stable at ≤ -20° C for at least 1 year.
Background: CaM Kinase II
References
- Griffith, L.C. (2004) J. Neurosci. 24:8391.
- Hudmon, A. and H. Schulman (2002) Annu. Rev. Biochem. 71:473.
- Lin, C.R. et al. (1987) Proc. Natl. Acad. Sci. USA 84:5962.
- Thiel, G. et al. (1988) Proc. Natl. Acad. Sci. USA 85:6337.
- Elgersma, Y. et al. (2002) Neuron 36:493.
Long Name
Calcium/Calmodulin-dependent Protein Kinase II
Alternate Names
CAMK2, CAMK2B
Additional CaM Kinase II Products
Product Documents for Phospho-CaM Kinase II (T286) Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Phospho-CaM Kinase II (T286) Antibody
For research use only
Related Research Areas
Citations for Phospho-CaM Kinase II (T286) Antibody
Customer Reviews for Phospho-CaM Kinase II (T286) Antibody
There are currently no reviews for this product. Be the first to review Phospho-CaM Kinase II (T286) Antibody and earn rewards!
Have you used Phospho-CaM Kinase II (T286) Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways