Matrix metalloproteinases (MMPs) are a family of zinc and calcium dependent endopeptidases with the combined ability to degrade all the components of the extracellular matrix. MMP-8 (neutrophil collagenase) is expressed in neutrophils, where it is stored in specific granules. MMP-8 release from the neutrophils is stimulated by various factors such as interleukins 1 and 8, TNF-alpha and GM-CSF. MMP-8 is capable of cleaving types I, II and III triple-helical collagen, gelatin peptides, fibronectin, proteoglycans, aggrecan, serpins, beta -casein and peptides such as angiotensin and substance P. In addition to its function in phagocytosis, MMP‑8 has a high capacity for infiltrating connective tissue, and is implicated in the breakdown of the extracellular matrix in diseases such as rheumatoid arthritis. Structurally, MMP-8 consists of several domains: a pro-domain that is cleaved upon activation, a catalytic domain containing the zinc-binding site, a short hinge region and a hemopexin-like domain. MMP-8 is heavily glycosylated.
Recombinant Human MMP-8 Protein, CF
R&D Systems | Catalog # 908-MP


Key Product Details
- R&D Systems NS0-derived Recombinant Human MMP-8 Protein (908-MP)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
Accession Number
Structure / Form
Applications
Product Specifications
Source
Phe21-Gly467
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The specific activity is >250 pmol/min/µg, as measured under the described conditions.
Reviewed Applications
Read 3 reviews rated 5 using 908-MP in the following applications:
Formulation, Preparation, and Storage
908-MP
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl and CaCl2. |
| Shipping | The product is shipped with dry ice or equivalent. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: MMP-8
Long Name
Alternate Names
Gene Symbol
UniProt
Additional MMP-8 Products
Product Documents for Recombinant Human MMP-8 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human MMP-8 Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Human MMP-8 Protein, CF
Customer Reviews for Recombinant Human MMP-8 Protein, CF (3)
Have you used Recombinant Human MMP-8 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
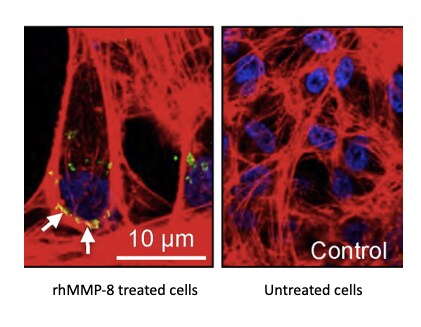
Application: In vitro bioactivity in cell cultureVerified Customer | Posted 03/04/2023rhMMP-8 was used to induce pro-inflammatory responses in ARPE-19 human cells. After the induction, the present of pro-angiogenic proteins, a proteolytic product derived from the pro-inflammatory status, was quantified by confocal imaging.
-
Application: Enzymatic activity in vitroVerified Customer | Posted 09/07/2019
-
Application: In vitro bioactivity in cell cultureVerified Customer | Posted 09/30/2018
There are no reviews that match your criteria.
Protocols
View specific protocols for Recombinant Human MMP-8 Protein, CF (908-MP):
- Assay Buffer: 50 mM Tris, 10 mM CaCl2, 150 mM NaCl, 0.05% (w/v) Brij-35, pH 7.5 (TCNB)
- Recombinant Human MMP‑8 (rhMMP-8) (Catalog # 908-MP)
- p-aminophenylmercuric acetate (APMA) (Sigma, Catalog # A-9563), 100 mM stock in DMSO
- Substrate: MCA-Pro-Leu-Gly-Leu-DPA-Ala-Arg-NH2 (Catalog # ES001), 2 mM stock in DMSO
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent
- Activate rhMMP-8 at 100 µg/mL with 1 mM APMA in Assay Buffer.
- Incubate reaction at 37 °C for 1 hour.
- Dilute activated rhMMP-8 to 1.0 ng/µL in Assay Buffer.
- Dilute Substrate to 20 µM in Assay Buffer.
- In a plate load 50 µL of 1.0 ng/µL rhMMP-8, and start the reaction by adding 50 µL of 20 µM Substrate to wells. Include a Substrate Blank containing 50 µL Assay Buffer and 50 µL of 20 µM Substrate.
- Read at excitation and emission wavelengths of 320 nm and 405 nm, respectively, in kinetic mode for 5 minutes.
- Calculate specific activity:
|
Specific Activity (pmol/min/µg) = |
Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Derived using calibration standard MCA-Pro-Leu-OH (Bachem, Catalog # M-1975).
Per Well:
- rhMMP-8: 0.050 µg
- Substrate: 10 µM
FAQs for Recombinant Human MMP-8 Protein, CF
-
Q: Can the enzyme be stored after activation, or do I need to use it immediately after activation?
A: We recommend only activating the amount of enzyme needed for your assay, and recommend activating the enzyme immediately prior to use. Any unactivated enzyme should be stored in aliquots at either the stock concentration at which the enzyme was supplied, or the reconstitution concentration, according to the product datasheet.
-
Q: Does this MMP enzyme need to be activated to work?
A: Yes, this enzyme requires activation prior to use.
-
Q: If I use this enzyme at a higher concentration, do I need to change the concentration of APMA to activate it?
A: We have only optimized activation conditions for one particular concentration of this MMP enzyme as part of our regular QC testing for enzymatic activity. Activating the enzyme at any different concentration would have to be optimized by the end user.
-
Q: What is the activity of this enzyme in units/µg?
A: We supply this enzyme as a mass and calculate its activity relative to mass (pmol/min/µg). We have not calibrated this enzyme to an international standard unit, so we are unable to provide a conversion to units/µg.
-
Q: Can the enzyme be stored after activation, or do I need to use it immediately after activation?
A: We recommend only activating the amount of enzyme needed for your assay, and recommend activating the enzyme immediately prior to use. Any unactivated enzyme should be stored in aliquots at either the stock concentration at which the enzyme was supplied, or the reconstitution concentration, according to the product datasheet.
-
Q: Does this MMP enzyme need to be activated to work?
A: Yes, this enzyme requires activation prior to use.
-
Q: If I use this enzyme at a higher concentration, do I need to change the concentration of APMA to activate it?
A: We have only optimized activation conditions for one particular concentration of this MMP enzyme as part of our regular QC testing for enzymatic activity. Activating the enzyme at any different concentration would have to be optimized by the end user.
-
Q: What is the activity of this enzyme in units/µg?
A: We supply this enzyme as a mass and calculate its activity relative to mass (pmol/min/µg). We have not calibrated this enzyme to an international standard unit, so we are unable to provide a conversion to units/µg.
-
Q: Can the enzyme be stored after activation, or do I need to use it immediately after activation?
A: We recommend only activating the amount of enzyme needed for your assay, and recommend activating the enzyme immediately prior to use. Any unactivated enzyme should be stored in aliquots at either the stock concentration at which the enzyme was supplied, or the reconstitution concentration, according to the product datasheet.
-
Q: Does this MMP enzyme need to be activated to work?
A: Yes, this enzyme requires activation prior to use.
-
Q: If I use this enzyme at a higher concentration, do I need to change the concentration of APMA to activate it?
A: We have only optimized activation conditions for one particular concentration of this MMP enzyme as part of our regular QC testing for enzymatic activity. Activating the enzyme at any different concentration would have to be optimized by the end user.
-
Q: What is the activity of this enzyme in units/µg?
A: We supply this enzyme as a mass and calculate its activity relative to mass (pmol/min/µg). We have not calibrated this enzyme to an international standard unit, so we are unable to provide a conversion to units/µg.
-
Q: Can the enzyme be stored after activation, or do I need to use it immediately after activation?
A: We recommend only activating the amount of enzyme needed for your assay, and recommend activating the enzyme immediately prior to use. Any unactivated enzyme should be stored in aliquots at either the stock concentration at which the enzyme was supplied, or the reconstitution concentration, according to the product datasheet.
-
Q: Does this MMP enzyme need to be activated to work?
A: Yes, this enzyme requires activation prior to use.
-
Q: If I use this enzyme at a higher concentration, do I need to change the concentration of APMA to activate it?
A: We have only optimized activation conditions for one particular concentration of this MMP enzyme as part of our regular QC testing for enzymatic activity. Activating the enzyme at any different concentration would have to be optimized by the end user.
-
Q: What is the activity of this enzyme in units/µg?
A: We supply this enzyme as a mass and calculate its activity relative to mass (pmol/min/µg). We have not calibrated this enzyme to an international standard unit, so we are unable to provide a conversion to units/µg.