Noggin is a secreted homodimeric glycoprotein that is an antagonist of bone morphogenetic proteins (BMPs) (1, 2). Human Noggin cDNA encodes a 232 amino acid (aa) precursor protein; cleavage of a 19 aa signal peptide generates the 213 aa mature protein which contains an N-terminal acidic region, a central basic heparin‑binding segment and a C-terminal cysteine-knot structure (2). Secreted Noggin probably remains close to the cell surface due to its binding of heparin‑containing proteoglycans (3). Noggin is very highly conserved among vertebrates, such that mature human Noggin shares 99%, 99%, 98%, 97% and 89% aa sequence identity with mouse, rat, bovine, equine and chicken Noggin, respectively. Noggin binds some BMPs such as BMP-4 with high affinity and others such as BMP-7 with lower affinity. It antagonizes BMP bioactivities by blocking epitopes on BMPs that are needed for binding to both type I and type II receptors (2, 4). During embryogenesis, Noggin antagonizes specific BMPs at defined times, for example, during neural tube, somite and cardiomyocyte growth and patterning (5-7). During skeletal development, Noggin prevents chondrocyte hyperplasia, thus allowing proper formation of joints (4). Mutations within the cysteine-knot region of human Noggin are linked to multiple types of skeletal dysplasias that result in apical joint fusions (8). Noggin is expressed in defined areas of the adult central nervous system and peripheral tissues such as lung, skeletal muscle and skin (1). During culture of human embryonic stem cells (hESC) or neural stem cells under certain conditions, addition of Noggin to antagonize BMP activity may allow stem cells to proliferate while maintaining their undifferentiated state, or alternatively, to differentiate into dopaminergic neurons (6, 9 - 13). Noggin also appears to maintain adult stem cell populations in-vivo, for example, maintaining neural stem cells within the hippocampus (13).
New
Recombinant Human Noggin Protein, CF
R&D Systems | Catalog # BT-NOG

Loading...
Key Product Details
Source
E. coli
Accession Number
Structure / Form
Disulfide-linked homodimer
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human Noggin protein
Gln28 - Cys232 with an N-terminal Met
Gln28 - Cys232 with an N-terminal Met
Purity
>97%, by SDS-PAGE with quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met-Gln28
Predicted Molecular Mass
23 kDa (monomer)
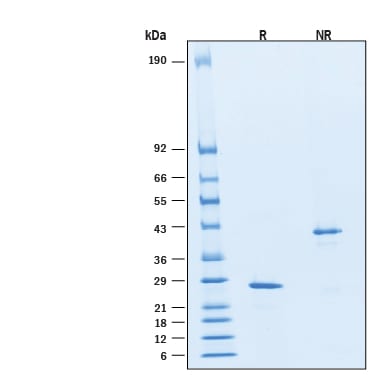
SDS-PAGE
27 kDa, under reducing conditions.
Activity
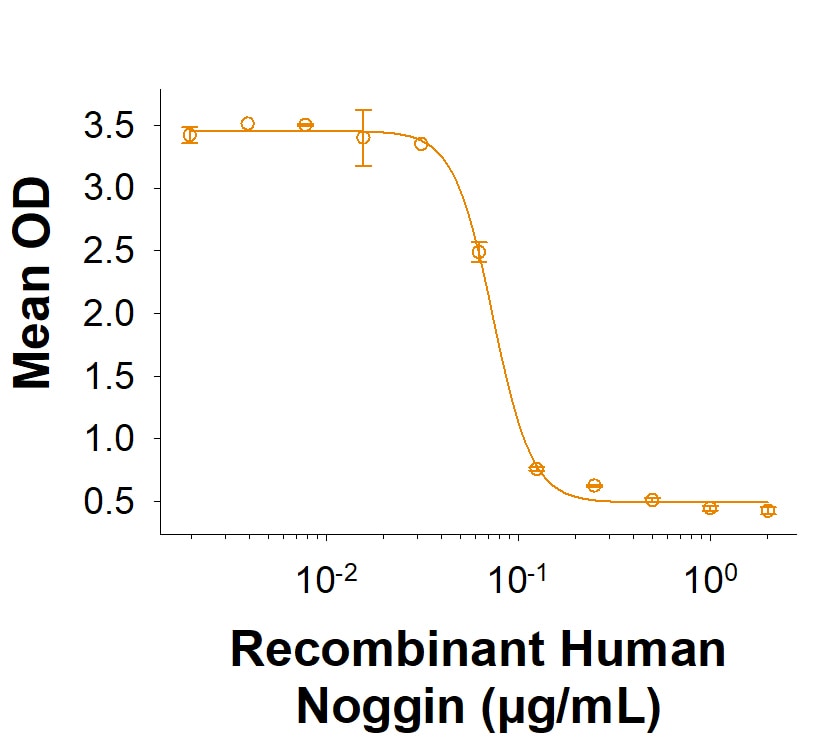
Measured by its ability to inhibit BMP-4-induced alkaline phosphatase production by ATDC5 mouse chondrogenic cells.
The ED50 for this effect is 0.0200-0.240 μg/mL
The ED50 for this effect is 0.0200-0.240 μg/mL
Scientific Data Images for Recombinant Human Noggin Protein, CF
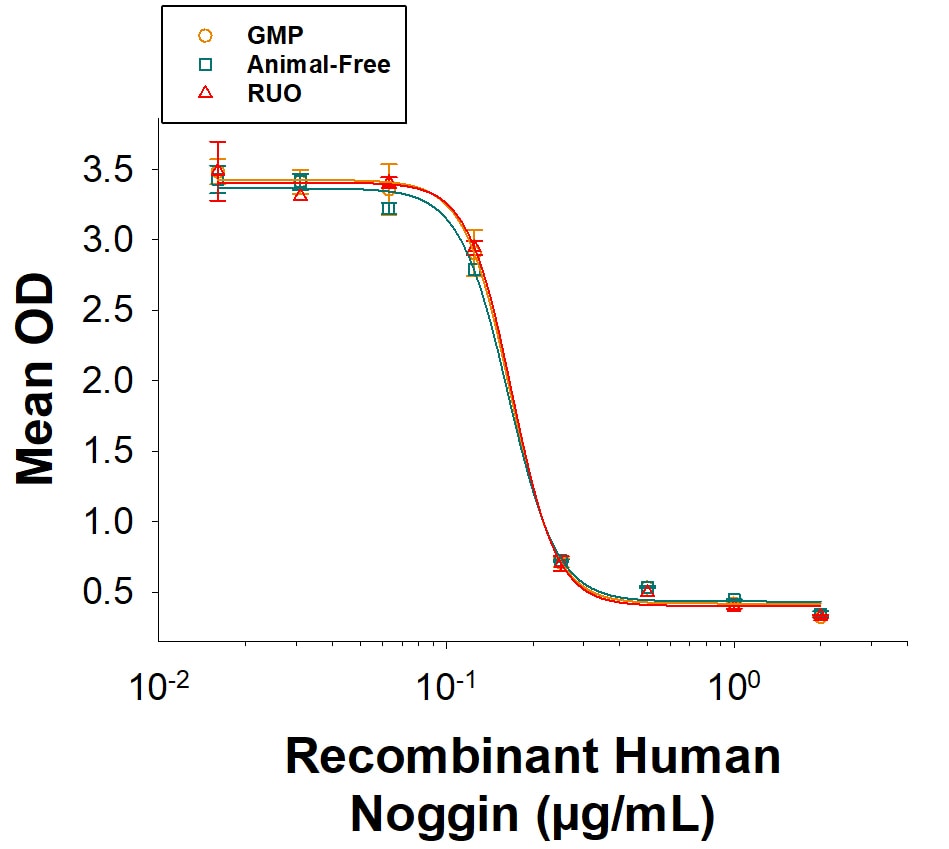
Equivalent Bioactivity of GMP,Animal-Free, and RUO gradesof Recombinant HumanNoggin.
Equivalent bioactivity of GMP (BT-NOG-GMP), Animal-Free (BT-NOG-AFL) and RUO (Catalog # BT-NOG) grades of Recombinant Human Noggin as measured by their ability to inhibit BMP-4-induced alkaline phosphatase production by ATDC5 mouse chondrogenic cells (orange, green, red, respectively).Recombinant Human Noggin Protein Bioactivity.
Recombinant Human Noggin Protein (Catalog # BT-NOG) inhibits Recombinant Human BMP-4 (314-BP) induced alkaline phosphatase production in the ATDC5 mouse chondrogenic cell line. The ED50 for this effect is 0.0200-0.240 µg/mL.Recombinant Human Noggin Protein SDS-PAGE.
2 μg/lane of Recombinant Human Noggin Protein (Catalog # BT-NOG) was resolved with SDS-PAGE under reducing (R) and non-reducing (NR) conditions and visualized by Coomassie® Blue staining, showing bands at 27 kDa and 43 kDa, respectively.Formulation, Preparation, and Storage
BT-NOG
| Formulation | Lyophilized from a 0.2 μm filtered solution in Sodium Acetate with Trehalose. |
| Reconstitution | Reconstitute at 500 μg/mL in water. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Noggin
References
- Valenzuela, D.M. et al. (1995) J. Neurosci. 15:6077.
- Groppe, J. et al. (2002) Nature 420:636.
- Paine-Saunders, S et al. (2002) J. Biol. Chem. 277:2089.
- Brunet, L. J. et al. (1998) Science 280:1455.
- McMahon, J. A. et al. (1998) Genes Dev. 12:1438.
- Itsykson, P. et al. (2005) Mol. Cell. Neurosci. 30:24.
- Yuasa, S. et al. (2005) Nat. Biotechnol. 23:607.
- Gong, Y. et al. (1999) Nat. Genet. 21:302.
- Xu, R.-H. et al. (2005) Nat. Methods 2:185.
- Wang, G. et al. (2005) Biochem. Biophys. Res. Commun. 330:934.
- Chaturvedi, G. et al. (2009) Cell Prolif. 42:425.
- Chiba, S. et al. (2008) Stem Cells 26:2810.
- Bonaguidi, M.A. et al. (2008) J. Neurosci. 28:9194.
Alternate Names
NOG, SYM1, SYNS1, SYNS1A
Gene Symbol
NOG
UniProt
Additional Noggin Products
Product Documents for Recombinant Human Noggin Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Noggin Protein, CF
For research use only
Related Research Areas
Customer Reviews for Recombinant Human Noggin Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Human Noggin Protein, CF and earn rewards!
Have you used Recombinant Human Noggin Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review