Serpin A1 is the archetypal member of the Serpin superfamily of the serine protease inhibitors (1). As one of the most abundant proteinase inhibitors in the circulation, it is synthesized in the liver and secreted into the bloodstream with the major function to protect tissues against neutrophil elastase. A severe serpin A1 deficiency leads to several clinical complications such as pulmonary emphysema, juvenile hepatitis, cirrhosis, and hepatocellular carcinoma (2). The deficiency is caused by point mutations in naturally occurring serpin A1 variants (over 70 are known). For example, the Z variant (Glu342 to Lys) forms intracellular inclusion bodies, is not secreted, and leads to a severe serpin A1 deficiency (3).
Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF
R&D Systems | Catalog # 1268-PI

Loading...
Key Product Details
- R&D Systems NS0-derived Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein (1268-PI)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
NS0
Accession Number
Applications
Inhibition Activity
Loading...
Product Specifications
Source
Mouse myeloma cell line, NS0-derived human Serpin A1/alpha 1-Antitrypsin protein
Glu25-Lys418, with a C-terminal 10-His tag
Glu25-Lys418, with a C-terminal 10-His tag
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<1.0 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Glu25
Predicted Molecular Mass
46 kDa
SDS-PAGE
60 kDa, reducing conditions
Activity
Measured by its ability to inhibit trypsin cleavage of a fluorogenic peptide substrate, Mca-RPKPVE-Nval-WRK(Dnp)-NH2 (Catalog # ES002).
The IC50 value is approximately <5.0 nM, as measured under the described conditions.
The IC50 value is approximately <5.0 nM, as measured under the described conditions.
Reviewed Applications
Read 1 review rated 3 using 1268-PI in the following applications:
Scientific Data Images for Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF
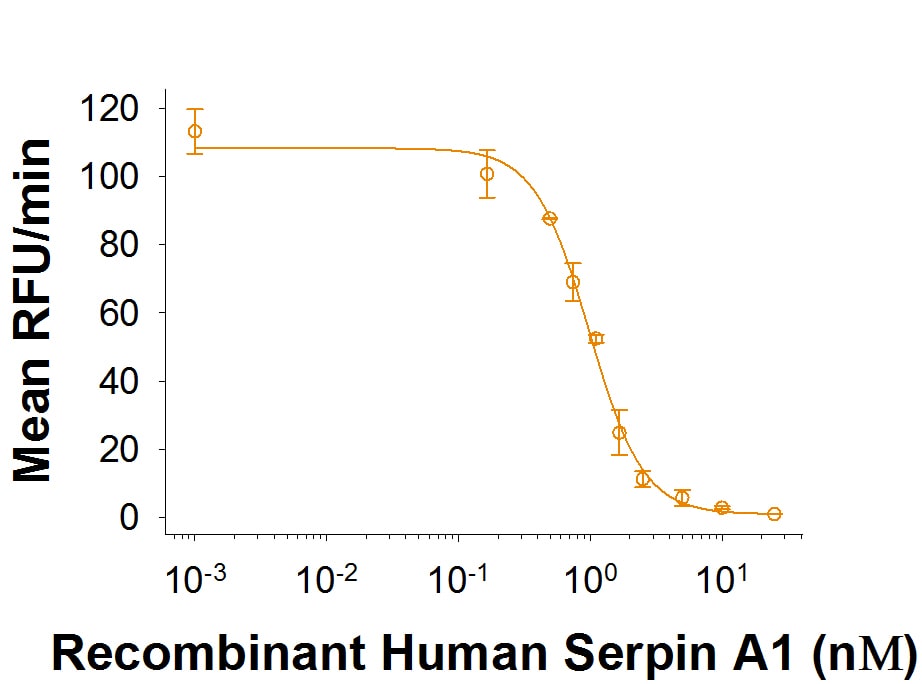
Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein Enzyme Activity
Recombinant Human Serpin A1/alpha-1-Antitrypsin (Catalog # 1268-PI) is measured by its ability to inhibit trypsin cleavage of a fluorogenic peptide substrate, Mca-RPKPVE-Nval-WRK(Dnp)-NH2 (Catalog # ES002).Formulation, Preparation, and Storage
1268-PI
| Formulation | Lyophilized from a 0.2 μm filtered solution in Tris, NaCl and CaCl2 with Trehalose. |
| Reconstitution | Reconstitute at 100 μg/mL in sterile 50 mM Tris, 10 mM CaCl2 and 150 mM NaCl, pH 7.5. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: Serpin A1/alpha 1-Antitrypsin
References
- Silverman, G.A. et al. (2001) J. Biol. Chem. 276:33293.
- Barbour, K.W. et al. (2002) Genomics 80:515.
- Lomas, D.A. et al. (2002) Biochem. Soc. Trans. 30:89.
Alternate Names
A1AT, alpha 1-Antitrypsin, alpha 1-Proteinase Inhibitor
Entrez Gene IDs
5265 (Human)
Gene Symbol
SERPINA1
UniProt
Additional Serpin A1/alpha 1-Antitrypsin Products
Product Documents for Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF
Customer Reviews for Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF (1)
3 out of 5
1 Customer Rating
Have you used Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunoassay StandardVerified Customer | Posted 12/20/2017
There are no reviews that match your criteria.
Protocols
View specific protocols for Recombinant Human Serpin A1/alpha-1-Antitrypsin Protein, CF (1268-PI):
Materials
- Assay Buffer: 50 mM Tris, 10 mM CaCl2, 150 mM NaCl, 0.05% (w/v) Brij-35, pH 7.5 (TCNB)
- Recombinant Human Serpin A1/ alpha 1‑Antitrypsin (rhSerpin A1) (Catalog # 1268-PI)
- Trypsin (Sigma, Catalog # T1426)
- Substrate: MCA-Arg-Pro-Lys-Pro-Val-Glu-Nval-Trp-Arg-Lys(DNP)-NH2 (Catalog # ES002), 2 mM stock in DMSO
- F16 Black Maxisorp Plate (Nunc, Catalog # 475515)
- Fluorescent Plate Reader (Model: SpectraMax Gemini EM by Molecular Devices) or equivalent.
- Dilute Trypsin to 0.25 µg/mL in Assay Buffer.
- Prepare a curve of rhSerpin A1 (MW: 45,667 Da) in Assay Buffer. Make the following serial dilutions:
200 nM, 100 nM, 50 nM, 33.3 nM, 22.2 nM, 14.8 nM, 9.88 nM, and 3.29 nM. - Combine 25 µL of 0.25 µg/mL Trypsin with 25 µL of rhSerpin A1 serial curve dilutions. Include two controls of 25 µL Assay Buffer with 25 µL of 0.25 µg/mL Trypsin.
- Incubate at room temperature for 30 minutes.
- After incubation, add 200 µL of Assay Buffer to each serial curve dilution.
- Dilute Substrate to 20 µM in Assay Buffer.
- In a plate, load 50 µL of the diluted rhSerpin A1 curve, and start the reaction by adding 50 µL of 20 µM Substrate to wells.
- Read at excitation and emission wavelengths of 320 nm and 405 nm (top read), respectively, in kinetic mode for 5 minutes.
- Derive the 50% inhibition concentration (IC50) value for hSerpin A1 by plotting RFU/min (or specific activity) vs. concentration with 4-PL fitting.
- The specific activity for Trypsin at each point may be determined using the following formula (if needed):
|
Specific Activity (pmol/min/µg) = |
Adjusted Vmax* (RFU/min) x Conversion Factor** (pmol/RFU) |
| amount of enzyme (µg) |
*Adjusted for Substrate Blank
**Derived using calibration standard MCA-Pro-Leu-OH (Bachem, Catalog # M-1975).
Per Well:
- Trypsin: 0.00125 µg
- rhSerpin A1: 10, 5, 2.5, 1.665, 1.11, 0.74, 0.494, 0.165 and 0 nM
- Substrate: 10 µM
Loading...
Associated Pathways

