Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF
R&D Systems | Catalog # 9258-SE

Loading...
Key Product Details
- R&D Systems NS0-derived Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein (9258-SE)
- Quality control testing to verify active proteins with lot specific assays by in-house scientists
- All R&D Systems proteins are covered with a 100% guarantee
Source
NS0
Accession Number
Structure / Form
Pro domain & mature form
Applications
Bioactivity
Loading...
Product Specifications
Source
Mouse myeloma cell line, NS0-derived mouse Proprotein Convertase 9/PCSK9 protein
Ser156-Gln694 with a C-terminal 10-His tag; Gln35-Gln155
Ser156-Gln694 with a C-terminal 10-His tag; Gln35-Gln155
Purity
>95%, by SDS-PAGE visualized with Silver Staining and quantitative densitometry by Coomassie® Blue Staining.
Endotoxin Level
<0.10 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Ser156 (mature form); No results obtained for pro domain, Gln35 inferred from enzymatic pyroglutamate treatment revealing Asp36
Predicted Molecular Mass
14 kDa (pro domain) and 59 kDa (mature form)
SDS-PAGE
18 kDa (pro domain) and 57-67 kDa (mature form), reducing conditions
Activity
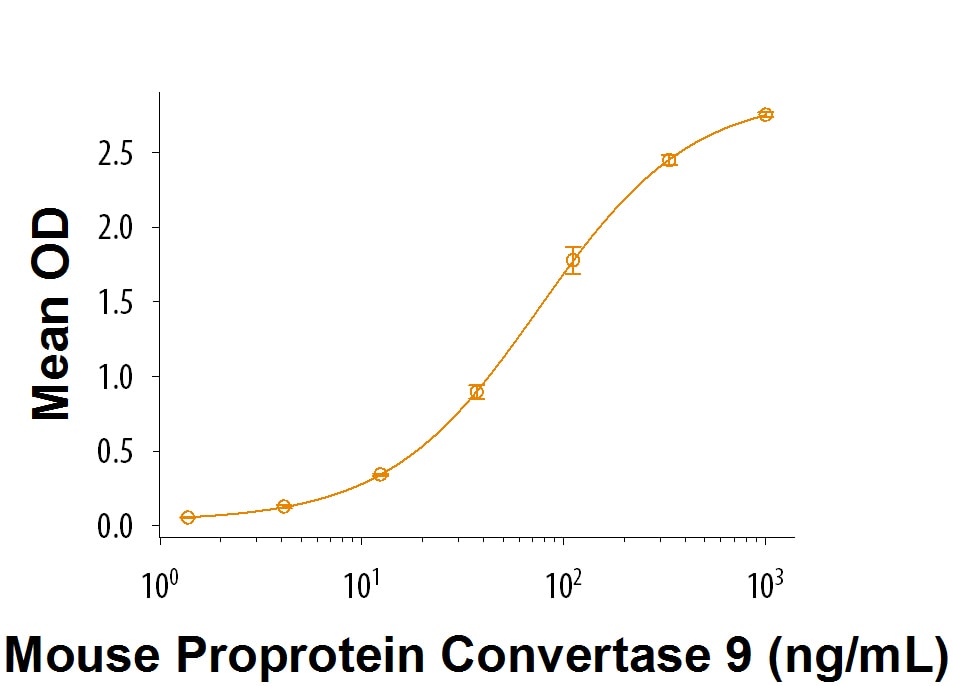
Measured by its binding ability in a functional ELISA.
When Recombinant Mouse LDLR (Catalog # 2255-LD) is coated at 2 µg/mL, Recombinant Mouse Proprotein Convertase 9/PCSK9 (Catalog #
9258-SE) binds with an ED50 = 25-150 ng/mL.
When Recombinant Mouse LDLR (Catalog # 2255-LD) is coated at 2 µg/mL, Recombinant Mouse Proprotein Convertase 9/PCSK9 (Catalog #
9258-SE) binds with an ED50 = 25-150 ng/mL.
Scientific Data Images for Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF
Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein Bioactivity
Recombinant Mouse Proprotein Convertase 9/ PCSK9 Binds to Recombinant Mouse LDL R in an ELISA Binding Assay. When Recombinant Mouse LDL R Recombinant Mouse LDL R (Catalog # 2255-LD) is coated at 2 µg/mL, Recombinant Mouse Proprotein Convertase 9/PCSK9 (Catalog # 9258-SE) binds with an ED50 = 25-150 ng/mL.Formulation, Preparation, and Storage
9258-SE
| Formulation | Supplied as a 0.2 μm filtered solution in Tris, NaCl and Glycerol. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage |
Background: Proprotein Convertase 9/PCSK9
References
- Schulz, R. et al. (2015) Basic Res. Cardiol. 110:4.
- Benjannet, S. et al. (2004) J. Biol. Chem. 279:48865.
- Lagace, T.A. et al. (2006) J. Clin. Invest. 116:2995.
- Grefhorst, A. et al. (2008) J. Lipid Res. 49:1303.
- Zhang, D.W. (2007) J. Biol. Chem. 282:18602.
- DeVay, R.M. et al. (2013) J. Biol. Chem. 288:10805.
- Canuel, M. et al. (2013) PLoS One 8:e64145.
- Poirier, S. et al. (2008) J. Biol. Chem. 283:2363.
- Levy, E. et al. (2013) Atherosclerosis 227:297.
- Le May, C. et al. (2013) Arterioscler. Thromb. Vasc. Biol. 33:1484.
- Roubtsova, A. et al. (2011) Arterioscler. Thromb. Vasc. Biol. 31:785.
- Kosenko, T. et al. (2013) J. Biol. Chem. 288:8279.
- Mayer, G. et al. (2008) J. Biol. Chem. 283:31791.
- Benjannet, S. et al. (2006) J. Biol. Chem. 281:30561.
- Benjannet, S. et al. (2010) J. Biol. Chem. 285:40965.
- Essalmani, R. et al. (2011) J. Biol. Chem. 286:4257.
- Han, B. et al. (2014) J. Lipid Res. 55:1505.
Alternate Names
FH3, FHCL3, HCHOLA3, LDLCQ1, NARC-1, NARC1, PC9, PCSK9
Gene Symbol
PCSK9
UniProt
Additional Proprotein Convertase 9/PCSK9 Products
- All Products for Proprotein Convertase 9/PCSK9
- Proprotein Convertase 9/PCSK9 cDNA Clones
- Proprotein Convertase 9/PCSK9 ELISA Kits
- Proprotein Convertase 9/PCSK9 Lysates
- Proprotein Convertase 9/PCSK9 Primary Antibodies
- Proprotein Convertase 9/PCSK9 Proteins and Enzymes
- Proprotein Convertase 9/PCSK9 Simple Plex
Product Documents for Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF
For research use only
Related Research Areas
Citations for Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF
Customer Reviews for Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF
There are currently no reviews for this product. Be the first to review Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF and earn rewards!
Have you used Recombinant Mouse Proprotein Convertase 9/PCSK9 Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Loading...
