DNMT1 Antibody (60B1220.1) - BSA Free
Novus Biologicals | Catalog # NB100-56519

![Western Blot: DNMT1 Antibody (60B1220.1)BSA Free [NB100-56519] Western Blot: DNMT1 Antibody (60B1220.1)BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Western-Blot-NB100-56519-img0003.jpg)
Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Clonality
Host
Isotype
Scientific Data Images for DNMT1 Antibody (60B1220.1) - BSA Free
Western Blot: DNMT1 Antibody (60B1220.1)BSA Free [NB100-56519]
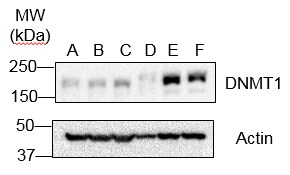
Western Blot: DNMT1 Antibody (60B1220.1) [NB100-56519] - Analysis of Dnmt1 in 2102EP (human embryonic carcinoma) in the A) absence and B) presence of immunizing peptide, recombinant human Dnmt1 protein in the C) absence and D) presence of immunizing peptide, NIH 3T3 (mouse embryonic fribroblast) in the E) absence and F) presence of immunizing peptide, and D3 (mouse embryonic stem cell) in the G) absence and H) presence of immunizing peptide using Dnmt1 antibody. this antibody. 5 ug/ml for testing the human cell line and 0.1 ug/ml was used for testing the recombinant protein and the mouse cell lines. ’s goat anti-mouse Ig HRP secondary antibody and PicoTect ECL substrate solution were used for this test.Immunocytochemistry/ Immunofluorescence: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]
Immunocytochemistry/Immunofluorescence: DNMT1 Antibody (60B1220.1) [NB100-56519] - Dnmt1 antibody was tested in HeLa cells with Dylight 488 (green). Nuclei and alpha-tubulin were counterstained with DAPI (blue) and DyLight 550 (red). Antibody dilution of 1:10 was used. Image objective 40x.Immunohistochemistry-Paraffin: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]
Immunohistochemistry-Paraffin: DNMT1 Antibody (60B1220.1) [NB100-56519] - Tissue section of human hepatocellular carcinoma at 1:50 dilution.Flow Cytometry: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]
Flow Cytometry: DNMT1 Antibody (60B1220.1) [NB100-56519] - An intracellular stain was performed on HeLa cells with DNMT1 [60B1220.1] Antibody NB100-56519AF488 (blue) and a matched isotype control (orange). Cells were fixed with 4% PFA and then permeabilized with 0.1% saponin. Cells were incubated in an antibody dilution of 5 ug/mL for 30 minutes at room temperature. Both antibodies were conjugated to Alexa Fluor 488.Flow Cytometry: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]
Flow Cytometry: DNMT1 Antibody (60B1220.1) [NB100-56519] - Analysis of PE conjugate of NB100-56519. An intracellular stain was performed on HeLa cells with DNMT1 antibody (60B1220.1) NB100-56519PE (blue) and a matched isotype control NBP2-27287PE (orange). Cells were fixed with 4% PFA and then permeablized with 0Simple Western: DNMT1 Antibody (60B1220.1)BSA Free [NB100-56519]
Simple Western: DNMT1 Antibody (60B1220.1) [NB100-56519] - Simple Western lane view shows a specific band for Dnmt1 in 0.5 mg/mL of HeLa lysate. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Immunoprecipitation: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]
DNMT1-Antibody-60B1220-1-Immunoprecipitation-NB100-56519-img0011.jpgWestern Blot: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] -
Western Blot: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] - WA does not alter DNMTs gene & protein expression(A) Effect of WA on DNMT1, DNMT3A & DNMT3B gene expression in MDA-MB-231 & MCF-7 cells normalized to select housekeeping genes & relative to DMSO-treated MCF-7 sample (2-delta delta Ct) as determined by Human Epigenetic Chromatin Modification Enzyme qPCR Array as previously described. Bars represent DNMT1, DNMT3A & DNMT3B relative mRNA (mean ± SEM) expression of at least two independent experiments. (B) Effect of WA on DNMT1, DNMT3A & DNMT3B protein expression in MDA-MB-231 & MCF-7 cells, as compared to to beta -Actin protein levels. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28467815), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Chromatin Immunoprecipitation: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] -
Chromatin Immunoprecipitation: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] - Epigenetic changes & protein binding at Ankrd26 promoter in eAT from mice upon 22 weeks of HFD.ChIP of DNMT1, DNMT3a, DNMT3b (a) & MBD2 (b) binding at Ankrd26 promoter region (−553 bp/−348 bp). (c) MNase for Nuc-2 (−257 bp/−198 bp) & Nuc-1 (−84 bp/−25 bp) occupancy at Ankrd26 promoter. (d) ChIP for acetyl-H4 enrichment at Nuc-2 & Nuc-1. (e) ChIP of RNA Pol II binding at Ankrd26 TSS (+16 bp/+159 bp). (a,b) & (d,e), ChIP enrichment is relative to Input chromatin. (a–e), results are mean ± SD from three independent experiments. **p < 0.01 & ***p < 0.001 vs STD. Image collected & cropped by CiteAb from the following publication (https://www.nature.com/articles/srep43526), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] -
Western Blot: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] - Verification of the expression of the proteins identified.(A) Validation of the SILAC results of selected proteins in immunoblots of protein extracts from the bladder cancer cells analyzed. The results validated the expression levels of proteins identified by the proteomic approach, including differentially & non-differentially expressed candidates. Antibodies displaying a single predominant band at the expected molecular weights were accepted: & alpha -tubulin, was used as the loading control. GSN, Gelsolin; Cul3, Cullin 3; IPO9. Importin 9; EGFR, Epidermal Growth Factor Receptor; NUP133, Nucleoporin 133; HSP70, Heat Shock Protein 70kDa; MCM6, Minichromosome Maintenance Complex Component 6; RCC1, Regulator of Chromosome Condensation 1; BCAS2, Breast Carcinoma Amplified Sequence 2; DNM, Dynamin; NPM, Nucleophosmin; DCTN, Dynactin; CALR, Calreticulin; MAPK, Mitogen-Activated Protein Kinase; DDX21, DEAD (Asp-Glu-Ala-Asp) box polypeptide 21; CDC2: Cell Division Cycle 2; DNMT1, DNA (cytosine-5)-Methyltransferase 1; MSH6, MutS Homolog 6; RAB14, GTPase Rab14; VDAC, Voltage-Dependent Anion Channel; CK18, Cytokeratin 18; CALD, Caldesmon; CD44, CD44 antigen isoform 1 precursor 2; EZR, Ezrin; MSN, Moesin; ANXA2, Annexin A2; CPNE3, Copine 3; FLNA, Filamin A; CAV1, Caveolin 1. Western Blots were scanned & analyzed using alpha -tubulin as normalizing control. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/23308193), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for DNMT1 Antibody (60B1220.1) - BSA Free
Chromatin Immunoprecipitation (ChIP)
Flow (Intracellular)
Flow Cytometry
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry Free-Floating
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Immunoprecipitation
Knockdown Validated
Simple Western
Western Blot
See Simple Western Antibody Database for Simple Western validation: Tested in HeLa lysate 0.5 mg/mL, separated by Size, antibody dilution of 1:100. Separated by Size-Wes, Sally Sue/Peggy Sue.
Reviewed Applications
Read 4 reviews rated 4.5 using NB100-56519 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: DNMT1
Long Name
Alternate Names
Gene Symbol
UniProt
Additional DNMT1 Products
Product Documents for DNMT1 Antibody (60B1220.1) - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for DNMT1 Antibody (60B1220.1) - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for DNMT1 Antibody (60B1220.1) - BSA Free
Customer Reviews for DNMT1 Antibody (60B1220.1) - BSA Free (4)
Have you used DNMT1 Antibody (60B1220.1) - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: cellSpecies: HumanVerified Customer | Posted 01/30/2019
-
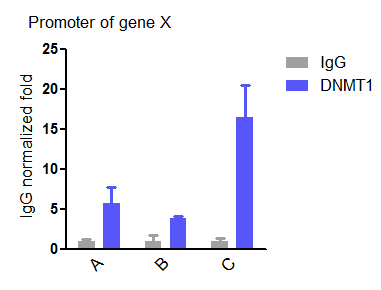
Application: Chromatin ImmunoprecipitationSample Tested: cellSpecies: HumanVerified Customer | Posted 01/30/2019ChIP of DNMT1 at the promoter of a gene showing DNMT1 was more enriched in cell line C than A and BMore than 1×10e6 cells and 2.4 μg antibody are optimal.
-
Application: Western BlotSample Tested: Prostate cancer cell lines, LnCAPSpecies: HumanVerified Customer | Posted 04/14/2016
-
Application: ImmunoprecipitationSample Tested: See PMID 24163373Species: HumanVerified Customer | Posted 12/12/2014
There are no reviews that match your criteria.
Protocols
View specific protocols for DNMT1 Antibody (60B1220.1) - BSA Free (NB100-56519):
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 4% paraformaldehyde to the dish and fix at room temperature for 10 minutes.
2. Remove the paraformaldehyde and wash the cells in PBS.
3. Permeabilize the cells with 0.1% Triton X100 or other suitable detergent for 2 min.
4. Remove the permeabilization buffer and wash three times for 5 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 5 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 5 minutes each.
10. Counter stain DNA with DAPI if required.
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for DNMT1 Antibody (60B1220.1) - BSA Free
-
Q: Could you please tell me the size of your DNMT1 protein along with Expression System? I am also doing mutant DNMT1 protein expression using the truncated part (650-1602=952 aa), using bacterial expression system (E.coli, BL21(De3). But right now I am getting some problem that, my size of protein is little higher than my accurate size? I use Auto-induction method to express the protein.
A: As you can see from our testing data, we see bands at around 185-200 kDa. Full details of the samples can be found on the image captions. Dnmt1,UniProt P26358, is also highly phosphorylated and acetylated which can raise the apparent molecular weight to higher than the predicted molecular weight. This is why you are seeing the change in size compared to what you might expect from your truncated protein.
-
Q: We have recently purchased the above mentioned anti-DNMT1 antibody. However, the product datasheet does not have any exact details on the molecular weight of the antibody and images provided are with very wide range protein marker (near DNMT-1 band 116-200kD).
A: According to Uniprot #P26358 the theoretical molecular weight of human DNMT1 is 183 kDa for isoform 1, ~185 kDa for isoform 2 and 144 kDa for isoform 3. This antibody is designed to detect all 3 of these isoforms.
Please not that this protein is subject to multiple posttranslational modifications which may differ by the species, cell type and disease state and these may affect how the protein runs on WB. In case of the WB image (image 1 of 5) we show several human an mouse cell lines, human recombinant protein, and each blot shows at least 1 band which looks like the predicted molecular weight of the target. I agree that the 116-200 range is rather wide and it is impossible to say that the protein runs exactly at 183kDa, but we are confident this is a specific band as, in addition, it is blocked by the blocking peptide. The entire WB image is displayed to show that there were other protein bands observed in some cell lines, also blocked by the peptide. These may be similar epitopes present at other targets or results of posttranslational processing of the DNMT1 protein in these cell types. Further study would be necessary to determine what exactly are these, but we present this data to make the customers aware that there may be some additional bands on the WB. If you are running a WB and ar enot confident that what you see is your target, we recommend to include some positive (overexpression lysate or treatment that induces the protein expression) or/and negative (KO, siRNA, cell line or tissue known not to express your target protein) controls. -
Q: Could you please tell me the size of your DNMT1 protein along with Expression System? I am also doing mutant DNMT1 protein expression using the truncated part (650-1602=952 aa), using bacterial expression system (E.coli, BL21(De3). But right now I am getting some problem that, my size of protein is little higher than my accurate size? I use Auto-induction method to express the protein.
A: As you can see from our testing data, we see bands at around 185-200 kDa. Full details of the samples can be found on the image captions. Dnmt1,UniProt P26358, is also highly phosphorylated and acetylated which can raise the apparent molecular weight to higher than the predicted molecular weight. This is why you are seeing the change in size compared to what you might expect from your truncated protein.
-
Q: We have recently purchased the above mentioned anti-DNMT1 antibody. However, the product datasheet does not have any exact details on the molecular weight of the antibody and images provided are with very wide range protein marker (near DNMT-1 band 116-200kD).
A: According to Uniprot #P26358 the theoretical molecular weight of human DNMT1 is 183 kDa for isoform 1, ~185 kDa for isoform 2 and 144 kDa for isoform 3. This antibody is designed to detect all 3 of these isoforms.
Please not that this protein is subject to multiple posttranslational modifications which may differ by the species, cell type and disease state and these may affect how the protein runs on WB. In case of the WB image (image 1 of 5) we show several human an mouse cell lines, human recombinant protein, and each blot shows at least 1 band which looks like the predicted molecular weight of the target. I agree that the 116-200 range is rather wide and it is impossible to say that the protein runs exactly at 183kDa, but we are confident this is a specific band as, in addition, it is blocked by the blocking peptide. The entire WB image is displayed to show that there were other protein bands observed in some cell lines, also blocked by the peptide. These may be similar epitopes present at other targets or results of posttranslational processing of the DNMT1 protein in these cell types. Further study would be necessary to determine what exactly are these, but we present this data to make the customers aware that there may be some additional bands on the WB. If you are running a WB and ar enot confident that what you see is your target, we recommend to include some positive (overexpression lysate or treatment that induces the protein expression) or/and negative (KO, siRNA, cell line or tissue known not to express your target protein) controls.

![Immunocytochemistry/ Immunofluorescence: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] Immunocytochemistry/ Immunofluorescence: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Immunocytochemistry-Immunofluorescence-NB100-56519-img0006.jpg)
![Immunohistochemistry-Paraffin: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] Immunohistochemistry-Paraffin: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Immunohistochemistry-Paraffin-NB100-56519-img0007.jpg)
![Flow Cytometry: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] Flow Cytometry: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Flow-Cytometry-NB100-56519-img0010.jpg)
![Flow Cytometry: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] Flow Cytometry: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Flow-Cytometry-NB100-56519-img0009.jpg)
![Simple Western: DNMT1 Antibody (60B1220.1)BSA Free [NB100-56519] Simple Western: DNMT1 Antibody (60B1220.1)BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Simple-Western-NB100-56519-img0008.jpg)
![Immunoprecipitation: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] Immunoprecipitation: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519]](https://resources.rndsystems.com/images/products/DNMT1-Antibody-60B1220-1-Immunoprecipitation-NB100-56519-img0011.jpg)
![Western Blot: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] - DNMT1 Antibody (60B1220.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56519_mouse-monoclonal-dnmt1-antibody-60b1220-1-310202415171327.jpg)
![Chromatin Immunoprecipitation: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] - DNMT1 Antibody (60B1220.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56519_mouse-monoclonal-dnmt1-antibody-60b1220-1-310202415304276.jpg)
![Western Blot: DNMT1 Antibody (60B1220.1) - BSA Free [NB100-56519] - DNMT1 Antibody (60B1220.1) - BSA Free](https://resources.rndsystems.com/images/products/nb100-56519_mouse-monoclonal-dnmt1-antibody-60b1220-1-3102024165721.jpg)