EGR2 Antibody - BSA Free
Novus Biologicals | Catalog # NB110-59723

![Western Blot: EGR2 AntibodyBSA Free [NB110-59723] Western Blot: EGR2 AntibodyBSA Free [NB110-59723]](https://resources.rndsystems.com/images/products/EGR2-Antibody-Western-Blot-NB110-59723-img0002.jpg)
Key Product Details
Species Reactivity
Validated:
Human, Mouse, Porcine
Cited:
Mouse, Rat
Applications
Validated:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry/ Immunofluorescence
Cited:
Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Format
BSA Free
Loading...
Product Specifications
Immunogen
A synthetic peptide made to a portion of human EGR2 (within residues 200-300). [Swiss-Prot# P11161]
Reactivity Notes
Immunogen displays the following percentage of sequence identity for non-tested species: mouse (86%), rat (86%), chicken (86%) and Xenopus (86%) proteins.
Localization
Nuclear
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Theoretical MW
50 kDa.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for EGR2 Antibody - BSA Free
Western Blot: EGR2 AntibodyBSA Free [NB110-59723]
Western Blot: EGR2 Antibody [NB110-59723] - Detection of EGR2 in human fetal lung tissue using NB110-59723.Immunocytochemistry/ Immunofluorescence: EGR2 Antibody - BSA Free [NB110-59723]
Immunocytochemistry/Immunofluorescence: EGR2 Antibody [NB110-59723] - MCF7 cells were fixed in 4% paraformaldehyde for 10 minutes and permeabilized in 0.5% Triton X-100 in PBS for 5 minutes. The cells were incubated with anti-EGR2 Antibody NB110-59723 at 2 ug/ml overnight at 4C and detected with an anti-rabbit Dylight 488 (Green) at a 1:1000 dilution for 60 minutes. Nuclei were counterstained with DAPI (Blue). Cells were imaged using a 100X objective and digitally deconvolved.Immunohistochemistry-Paraffin: EGR2 Antibody - BSA Free [NB110-59723]
Immunohistochemistry-Paraffin: EGR2 Antibody [NB110-59723] - Analysis of a FFPE tissue section of mouse brain using 1:200 dilution of EGR2 antibody. The staining was developed using HRP labeled anti-rabbit secondary antibody and DAB reagent, and nuclei of cells were counter-stained with hematoxylin.Immunocytochemistry/ Immunofluorescence: EGR2 Antibody - BSA Free [NB110-59723]
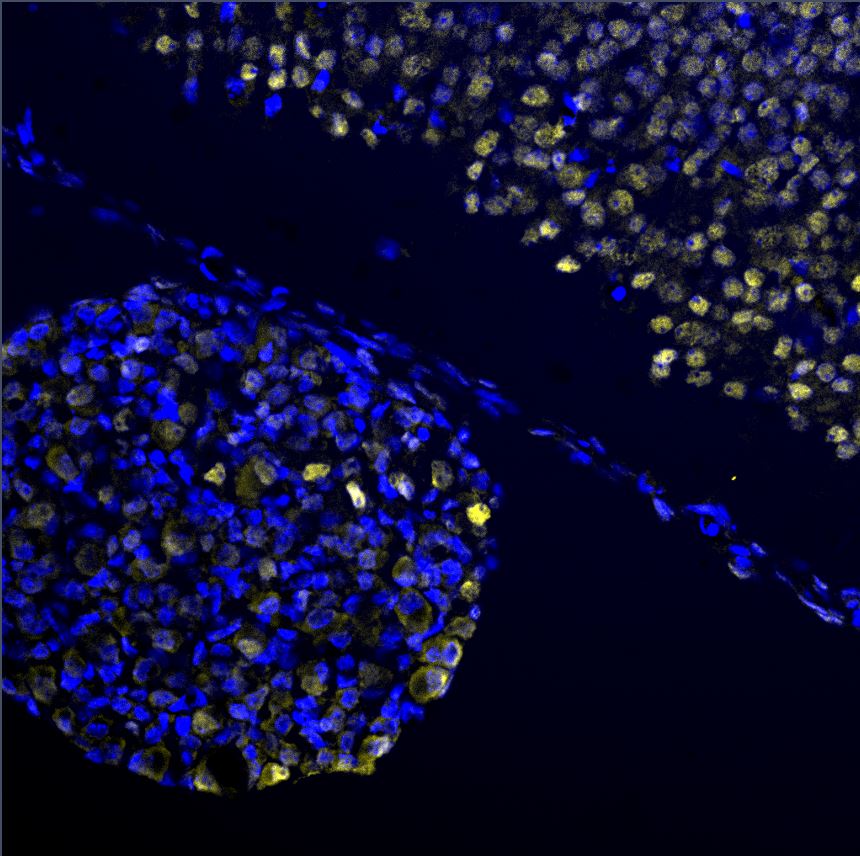
Immunocytochemistry/Immunofluorescence: EGR2 Antibody [NB110-59723] - EGR2 staining on E13.5 dorsal root ganglion and neural tube.Western Blot: EGR2 Antibody - BSA Free [NB110-59723] -
Overexpression of miR-10a-5p decreased cardiomyocyte apoptosis through inhibiting EGR2. (A) The target genes of miR-10a-5p predicted using Starbase, miRWalk, and RAID databases and the up-regulated genes in MI-related dataset GSE23294 and the potential binding sites of miR-10a-5p on EGR2. (B) The luciferase activity measured by dual-luciferase reporter gene assay. (C) Expression of EGR2 in myocardial tissues normalized to GAPDH determined by RT-qPCR (n = 10). (D) The expression pattern of EGR2 in hypoxic cardiomyocytes normalized to GAPDH determined by RT-qPCR. (E) The expression pattern of EGR2 in cardiomyocytes normalized to GAPDH determined by Western blot analysis. (F), Apoptosis of cardiomyocytes after EGR2 silencing determined by flow cytometry. (G) The expression patterns of EGR2, cleaved-caspase-3, Bax and Bcl-2 in cardiomyocytes after EGR2 silencing normalized to GAPDH determined by Western blot analysis. (H) The caspase-3 activity in hypoxic cardiomyocytes after EGR2 silencing. (I) The ATP content in hypoxic cardiomyocytes after EGR2 silencing. (J) The expression pattern of EGR2 in the hypoxic cardiomyocytes after alteration of EGR2 and/or miR-10a-5p normalized to GAPDH determined by RT-qPCR. (K) The caspase-3 activity in hypoxic cardiomyocytes after alteration of EGR2 and/or miR-10a-5p determined by RT-qPCR. (L) The ATP content in hypoxic cardiomyocytes after alteration of EGR2 and/or miR-10a-5p. * p < 0.05. The above data were all measurement data, and expressed as mean +/- standard deviation. The unpaired t test was adopted for comparison between two groups. One-way ANOVA was adopted for comparison among multiple groups with Tukey’s post hoc test. All data was generated from 3 independent experiments respectively. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/33819189), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: EGR2 Antibody - BSA Free [NB110-59723] -
Expression changes and cellular localization of Stat1 after nerve injury. A (a1) Western blots showing the expression change of Stat1, MAG, Nab2 and Egr2 in nerve injury segment at the indicated time points following a nerve crush, with uninjured nerve used as the control (Ctrl). GAPDH served as the loading control. (a2–6) Histogram quantitatively compare the expression changes of Stat1, MAG, Nab2, and Egr2. ###p < 0.001, one-way ANOVA, *p < 0.05, **p < 0.01, ***p < 0.001 vs control, n = 3 ~ 4 per group. B Immunofluorescence staining of S100 beta (a marker of SCs, red) and Stat1 (green) in injured sciatic nerve showing cellular localization of Stat1: on day 1 of injury, co-localization of Stat1 and S100 beta was almost absent; on day 4 of injury, there was a small amount of Stat1 and S100 beta co-localization; and from day 7 to day 28 of injury, Stat1 was mainly localized in SCs. Scale bars, 100 μm Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/37365519), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for EGR2 Antibody - BSA Free
Application
Recommended Usage
Immunocytochemistry/ Immunofluorescence
1-5 ug/ml
Immunohistochemistry
1:200
Immunohistochemistry-Paraffin
1:200
Western Blot
2 ug/ml
Application Notes
This EGR2 antibody is useful for ICC and Western blot, where a band is seen at ~50 kDa. There is also a strong non-specific band at ~75 kDa. In ICC/IF nuclear staining was observed in Hela cells.
Reviewed Applications
Read 1 review rated 4 using NB110-59723 in the following applications:
Formulation, Preparation, and Storage
Purification
Immunogen affinity purified
Formulation
PBS
Format
BSA Free
Preservative
0.05% Sodium Azide
Concentration
1.0 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at 4C short term. Aliquot and store at -20C long term. Avoid freeze-thaw cycles.
Background: EGR2
Alternate Names
AT591, CMT1D, CMT4E, early growth response 2, early growth response 2 (Krox-20 homolog, Drosophila), early growth response protein 2, EGR-2, FLJ14547, Krox-20 homolog, Drosophila, KROX-20, Drosophila, homolog (early growth response-2), KROX20DKFZp686J1957, Zinc finger protein Krox-20
Gene Symbol
EGR2
Additional EGR2 Products
Product Documents for EGR2 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for EGR2 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for EGR2 Antibody - BSA Free
Customer Reviews for EGR2 Antibody - BSA Free (1)
4 out of 5
1 Customer Rating
Have you used EGR2 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunohistochemistry-ParaffinSample Tested: Embryonic neural tube and Embryonic dorsal root ganglionSpecies: MouseVerified Customer | Posted 02/24/2017EGR2 staining on E13.5 dorsal root ganglion and neural tube
There are no reviews that match your criteria.
Protocols
View specific protocols for EGR2 Antibody - BSA Free (NB110-59723):
EGR2 Antibody:
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and add 10% formalin to the dish. Fix at room temperature for 30 minutes.
2. Remove the formalin and add ice cold methanol. Incubate for 5-10 minutes.
3. Remove methanol and add washing solution (i.e. PBS). Be sure to not let the specimen dry out. Wash three times for 10 minutes.
4. To block nonspecific antibody binding incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
5. Add primary antibody at appropriate dilution and incubate at room temperature from 2 hours to overnight at room temperature.
6. Remove primary antibody and replace with washing solution. Wash three times for 10 minutes.
7. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
8. Remove antibody and replace with wash solution, then wash for 10 minutes. Add Hoechst 33258 to wash solution at 1:25,0000 and incubate for 10 minutes. Wash a third time for 10 minutes.
9. Cells can be viewed directly after washing. The plates can also be stored in PBS containing Azide covered in Parafilm (TM). Cells can also be cover-slipped using Fluoromount, with appropriate sealing.
*The above information is only intended as a guide. The researcher should determine what protocol best meets their needs. Please follow safe laboratory procedures.
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and add 10% formalin to the dish. Fix at room temperature for 30 minutes.
2. Remove the formalin and add ice cold methanol. Incubate for 5-10 minutes.
3. Remove methanol and add washing solution (i.e. PBS). Be sure to not let the specimen dry out. Wash three times for 10 minutes.
4. To block nonspecific antibody binding incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
5. Add primary antibody at appropriate dilution and incubate at room temperature from 2 hours to overnight at room temperature.
6. Remove primary antibody and replace with washing solution. Wash three times for 10 minutes.
7. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
8. Remove antibody and replace with wash solution, then wash for 10 minutes. Add Hoechst 33258 to wash solution at 1:25,0000 and incubate for 10 minutes. Wash a third time for 10 minutes.
9. Cells can be viewed directly after washing. The plates can also be stored in PBS containing Azide covered in Parafilm (TM). Cells can also be cover-slipped using Fluoromount, with appropriate sealing.
*The above information is only intended as a guide. The researcher should determine what protocol best meets their needs. Please follow safe laboratory procedures.
EGR2 Antibody:
Western Blot Protocol
1. Perform SDS-PAGE (4-12%) on samples to be analyzed, loading 40 ug of total protein of human fetal lung per lane.
2. Transfer proteins to Nitrocellulose according to the instructions provided by the manufacturer of the transfer
apparatus.
3. Rinse membrane with dH2O and then stain the blot using Ponceau S for 1-2 minutes to access the transfer of proteins onto the nitrocellulose membrane. Rinse the blot in water to remove excess stain and mark the lane locations and locations of molecular weight markers using a pencil.
4. Rinse the blot in TBS for approximately 5 minutes.
5. Block the membrane using 5% non-fat dry milk + 1% BSA in TBS, 1 hour at room temperature.
6. Rinse the membrane in dH2O and then wash the membrane in wash buffer [TBS + 0.1% Tween] 3 times for 10 minutes each.
7. Dilute the rabbit anti-EGR2 primary antibody (NB 110-59723) in blocking buffer and incubate 2 hours at room temperature.
8. Rinse the membrane in dH2O and then wash the membrane in wash buffer [TBS + 0.1% Tween] 3 times for 10 minutes each.
9. Apply the diluted rabbit-IgG HRP-conjugated secondary antibody in blocking buffer (as per manufacturers
instructions) and incubate 1 hour at room temperature.
10. Wash the blot in wash buffer [TBS + 0.1% Tween] 3 times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturers instructions (Pierce. ECL).
Note: Tween-20 can be added to the blocking or antibody diultion buffer at a final concentration of 0.05-0.2%, provided it does not interfere with antibody-antigen binding.
Western Blot Protocol
1. Perform SDS-PAGE (4-12%) on samples to be analyzed, loading 40 ug of total protein of human fetal lung per lane.
2. Transfer proteins to Nitrocellulose according to the instructions provided by the manufacturer of the transfer
apparatus.
3. Rinse membrane with dH2O and then stain the blot using Ponceau S for 1-2 minutes to access the transfer of proteins onto the nitrocellulose membrane. Rinse the blot in water to remove excess stain and mark the lane locations and locations of molecular weight markers using a pencil.
4. Rinse the blot in TBS for approximately 5 minutes.
5. Block the membrane using 5% non-fat dry milk + 1% BSA in TBS, 1 hour at room temperature.
6. Rinse the membrane in dH2O and then wash the membrane in wash buffer [TBS + 0.1% Tween] 3 times for 10 minutes each.
7. Dilute the rabbit anti-EGR2 primary antibody (NB 110-59723) in blocking buffer and incubate 2 hours at room temperature.
8. Rinse the membrane in dH2O and then wash the membrane in wash buffer [TBS + 0.1% Tween] 3 times for 10 minutes each.
9. Apply the diluted rabbit-IgG HRP-conjugated secondary antibody in blocking buffer (as per manufacturers
instructions) and incubate 1 hour at room temperature.
10. Wash the blot in wash buffer [TBS + 0.1% Tween] 3 times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturers instructions (Pierce. ECL).
Note: Tween-20 can be added to the blocking or antibody diultion buffer at a final concentration of 0.05-0.2%, provided it does not interfere with antibody-antigen binding.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

![Immunocytochemistry/ Immunofluorescence: EGR2 Antibody - BSA Free [NB110-59723] Immunocytochemistry/ Immunofluorescence: EGR2 Antibody - BSA Free [NB110-59723]](https://resources.rndsystems.com/images/products/EGR2-Antibody-Immunocytochemistry-Immunofluorescence-NB110-59723-img0007.jpg)
![Immunohistochemistry-Paraffin: EGR2 Antibody - BSA Free [NB110-59723] Immunohistochemistry-Paraffin: EGR2 Antibody - BSA Free [NB110-59723]](https://resources.rndsystems.com/images/products/EGR2-Antibody-Immunohistochemistry-Paraffin-NB110-59723-img0008.jpg)
![Immunocytochemistry/ Immunofluorescence: EGR2 Antibody - BSA Free [NB110-59723] Immunocytochemistry/ Immunofluorescence: EGR2 Antibody - BSA Free [NB110-59723]](https://resources.rndsystems.com/images/products/EGR2-Antibody-Immunofluorescence-NB110-59723-img0006.jpg)
![Western Blot: EGR2 Antibody - BSA Free [NB110-59723] - EGR2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb110-59723_rabbit-polyclonal-egr2-antibody-western-blot-132202615262624.jpg)
![Western Blot: EGR2 Antibody - BSA Free [NB110-59723] - EGR2 Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb110-59723_rabbit-polyclonal-egr2-antibody-western-blot-132202617571020.jpg)