EMMPRIN is expressed in areas of tissue remodeling, including tumors, endometrium, placenta, skin, and regions undergoing angiogenesis (1, 2, 6-9). It is also expressed in cells with high metabolic activity, such as lymphoblasts, macrophages and tumor cells (2, 10). On cells with elevated metabolic rates, EMMPRIN is often co-expressed with the amino acid transporter CD98h (11). EMMPRIN also interacts with caveolin-1 (via its C2-like domain), and this reduces the level of EMMPRIN glycosylation and subsequent EMMPRIN multimerization and activity (12). EMMPRIN’s TM sequence contains a Glu and a Pro which are important for intracellular interactions with cyclophilins (CyP) (3, 13, 14). CyPA (cyclosporin A receptor) and CyP60 interactions with the TM segment promote leukocyte inflammatory chemotaxis and surface expression of EMMPRIN, respectively (13, 14). An active 22 kDa fragment can be shed from tumor cells by MT1-MMP (1). Tumor cells can also release active, full-length EMMPRIN in microvesicles (15, 16). Functionally, EMMPRIN is known to induce urokinase-type plasminogen activator (uPA), VEGF, hyaluronan, and multiple MMPs (1, 2, 7, 8, 9). Human EMMPRIN (269 aa) shows 58%, 58%, 62%, and 52% aa identity with mouse, rat, bovine, and chicken EMMPRIN, respectively. It also shows 25% and 38% aa identity with the related proteins, embigin and neuroplastin (SDR-1), respectively (4).
Human EMMPRIN/CD147 Biotinylated Antibody
R&D Systems | Catalog # BAF972

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot
Cited:
ELISA Development, Proximity Ligation Assay, Functional Assay, Proximity Ligation Assay (PLA)
Label
Biotin
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant human EMMPRIN/CD147
Thr25-His205
Accession # Q54A51
Thr25-His205
Accession # Q54A51
Specificity
Detects human EMMPRIN/CD147 in Western blots. In Western blots, approximately 5% cross‑reactivity with recombinant mouse EMMPRIN is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human EMMPRIN/CD147 Biotinylated Antibody
Detection of Human EMMPRIN/CD147 by Immunohistochemistry
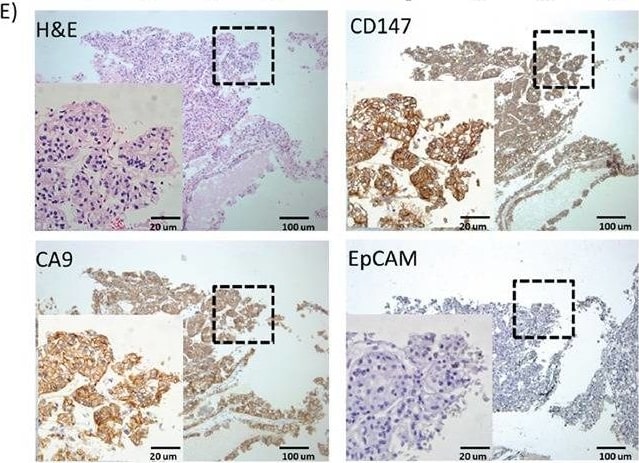
IHC analysis of EpCAM, CA9 and CD147 expression in RCC patient cohortA. Positive expression percentages of EpCAM, CA9 and CD147 in RCC patient samples of different clinical stages by IHC analysis (I, n=29; II, n=19; III, n=16; IV, n=6). B. IHC scores of EpCAM, CA9 and CD147 in RCC samples of (A). *** p<0.001. C. Positive expression percentages of CA9+ and/or CD147+ in EpCAM+ (I, n=8; II, n=3; III, n=1; IV, n=1) or EpCAM- (I, n=21; II, n=16; III, n=15; IV, n=5) RCC patient samples of different clinical stages by IHC analysis. D. IHC scores of EpCAM, CA9 and CD147 in RCC samples of (C). E. H&E and IHC staining of EpCAM, CA9 and CD147 in representative EpCAM- RCC patient tissues. The selected enlarged areas are indicated in dashed rectangles. Original magnification, 400× (enlarged) and 100× (original); scale bars: 20 μm (enlarged) and 100 μm (original). Image collected and cropped by CiteAb from the following publication (https://www.oncotarget.com/lookup/doi/10.18632/oncotarget.10979), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human EMMPRIN/CD147 Biotinylated Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Human EMMPRIN/CD147 Fc Chimera (Catalog # 972-EMN)
Sample: Recombinant Human EMMPRIN/CD147 Fc Chimera (Catalog # 972-EMN)
Reviewed Applications
Read 1 review rated 4 using BAF972 in the following applications:
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: EMMPRIN/CD147
References
- Gabison, E.E. et al. (2005) Biochimie 87:361.
- Yurchenko, V. et al. (2006) Immunology 117:301.
- Kasinrerk, W. et al. (1992) J. Immunol. 149:847.
- Miyauchi, T. et al. (1991) J. Biochem. 110:770.
- Hanna, S.M. et al. (2003) BMC Biochem. 4:17.
- Riethdorf, S. et al. (2006) Int. J. Cancer 119:1800.
- Braundmeier, A.G. et al. (2006) J. Clin. Endocrinol. Metab. 91:2358.
- Tang, Y. et al. (2006) Mol. Cancer Res. 4:371.
- Quemener, C. et al. (2007) Cancer Res. 67:9.
- Wilson, M.C. et al. (2005) J. Biol. Chem. 280:27213.
- Xu, D. and M.E. Hemler (2005) Mol. Cell. Proteomics 4:1061.
- Tang, W. et al. (2004) Mol. Biol. Cell 15:4043.
- Arora, K. et al. (2005) J. Immunol. 175:517.
- Pushkarsky, T. et al. (2005) J. Biol. Chem. 280:27866.
- Egawa, N. et al. (2006) J. Biol. Chem. 281:37576.
- Sidhu, S.S. et al. (2004) Oncogene 23:956.
Long Name
Extracellular Matrix Metalloproteinase Inducer
Alternate Names
Basigin, BSG, CD147
Gene Symbol
BSG
UniProt
Additional EMMPRIN/CD147 Products
Product Documents for Human EMMPRIN/CD147 Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human EMMPRIN/CD147 Biotinylated Antibody
For research use only
Citations for Human EMMPRIN/CD147 Biotinylated Antibody
Customer Reviews for Human EMMPRIN/CD147 Biotinylated Antibody (1)
4 out of 5
1 Customer Rating
Have you used Human EMMPRIN/CD147 Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: ELISASample Tested: See PMID: 23598007Species: HumanVerified Customer | Posted 01/15/2015
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
