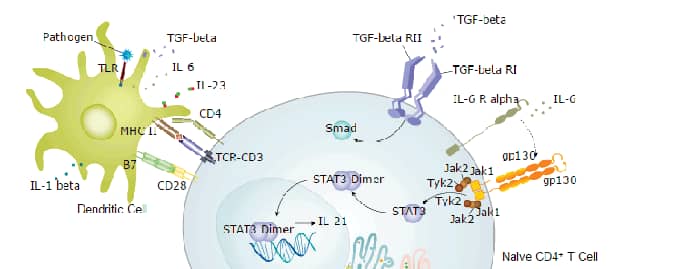
IL-26 was originally cloned from herpesvirus saimiri (HVS)-transformed T cells and named AK155 (1). It is a member of the IL-10 family of class II cytokines that signal via heterodimeric receptor complexes composed of two type I transmembrane receptor subunits (2). The human IL-26 gene has been mapped to chromosome 12q15. It encodes a 171 amino acid polypeptide with a 21 amino acid signal peptide. In addition to HVS-transformed T cells, IL-26 is also expressed in other virus transformed T cell lines, fresh peripheral mononuclear cells, activated NK cells and T cells. A mouse homologue of human IL-26 has not been identified. IL-26 binds with high-affinity to the heterodimeric complex consisting of the ligand-binding IL-20 R alpha and non ligand-binding IL-10 R beta (3). Activation of the receptor complex results in rapid phosphorylation of STAT1 and STAT3. Although the IL-26 receptor complex is highly specific for IL-26 and is not activated by other class II cytokines, the individual subunits of the IL-26 receptor complex are components in receptor complexes for other class II cytokines (1). IL-20 R alpha can form dimers with IL-20 R beta to function as signaling receptors for IL-19, IL-20, and IL-24. IL-10 R beta can complex with IL-10 R alpha, IL-22 R, and IL-28 R alpha to transduce signals for IL-10, IL-22, and the three novel IFNs (IL-28A, IL-28B and IL-29), respectively. The physiological functions of IL-26 remain to be determined. IL-26 was reported to be a homodimer in solution (1).
Human IL‑26/AK155 Biotinylated Antibody
R&D Systems | Catalog # BAF1375

Key Product Details
Species Reactivity
Validated:
Human
Cited:
Human
Applications
Validated:
Western Blot
Cited:
ELISA Development
Label
Biotin
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant human IL-26 (R&D Systems, Catalog # 1375-IL)
Lys22-Gln171
Accession # Q9NPH9
Lys22-Gln171
Accession # Q9NPH9
Specificity
Detects human IL-26 in Western blots. In this format, less than 2% cross-reactivity with recombinant human IL‑10 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Applications for Human IL‑26/AK155 Biotinylated Antibody
Application
Recommended Usage
Western Blot
0.1 µg/mL
Sample: Recombinant Human IL‑26/AK155 Monomer (Catalog # 1375-IL)
Sample: Recombinant Human IL‑26/AK155 Monomer (Catalog # 1375-IL)
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: IL-26/AK155
References
- Knappe. A. et al. (2000) J. Virology 74:3881.
- Renauld, J.-C. (2003) Nature Reviews Immunology 3:667.
- Sheikh, F. et al. (2004) J. Immunol. 172(4):2006.
- Unpublished data (2003) R&D Systems, Inc.
Long Name
Interleukin 26
Alternate Names
AK155, IL26
Entrez Gene IDs
55801 (Human)
Gene Symbol
IL26
UniProt
Additional IL-26/AK155 Products
Product Documents for Human IL‑26/AK155 Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human IL‑26/AK155 Biotinylated Antibody
For research use only
Citations for Human IL‑26/AK155 Biotinylated Antibody
Customer Reviews for Human IL‑26/AK155 Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Human IL‑26/AK155 Biotinylated Antibody and earn rewards!
Have you used Human IL‑26/AK155 Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways