Syndecan-2, previously known as fibroglycan or heparan sulfate proteoglycan, is a member of the syndecan family of Type 1 transmembrane proteins capable of carrying heparan sulfate (HS) and chondroitin sulfate glycosaminoglycans. The four vertebrate syndecans show conserved cytoplasmic domains and divergent extracellular portions (except for GAG attachment sites). Among the Syndecans, Syndecan-2 is most similar to Syndecan-4 (1‑3). Human Syndecan-2 is synthesized as a 201 amino acid (aa) core protein with an 18 aa signal sequence, a 126 aa extracellular domain (ECD), a 25 aa transmembrane region and a 32 aa cytoplasmic tail (4). The human ECD of Syndecan-2 contains three closely-spaced consensus Ser-Gly sequences for the attachment of HS side chains. It shares 76%, 73%, 87%, 78% and 63% aa identity with the ECD of mouse, rat, bovine, canine and chicken Syndecan-2, respectively. The cytoplasmic tail has both serine and tyrosine phosphorylation sites. Addition of 20‑80 disaccharides per side chain adds considerably to the size of the 22 kDa core protein. Non-covalent homodimerization of Syndecan-2 is dependent on the transmembrane domain (5). Syndecan-2 is expressed in cells of mesenchymal origin, neuronal and epithelial cells, and is the predominant syndecan expressed during embryonic development. Expression is upregulated in several cancer cell lines (6). After induction in macrophages by inflammatory mediators, Syndecan-2 selectively binds FGFbasic, VEGF and EGF (7). Syndecan-2 expressed on human primary osteoblasts binds GM-CSF and may function as a co‑receptor (8). Activated endothelial cell Syndecan-2 specifically binds IL-8 and may participate in promoting neutrophil extravasation by forming a chemotactic IL-8 gradient (9). Typically, cytokine, chemokine and extracellular matrix protein binding occurs through interaction with HS side chains, but the Syndecan-2 extracellular domain can bind TGF-beta directly via protein-protein interaction (10).
Human Syndecan-2/CD362 PE‑conjugated Antibody
R&D Systems | Catalog # FAB2965P

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Glu19-Gly144
Accession # AAH49836.1
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Syndecan-2/CD362 PE‑conjugated Antibody
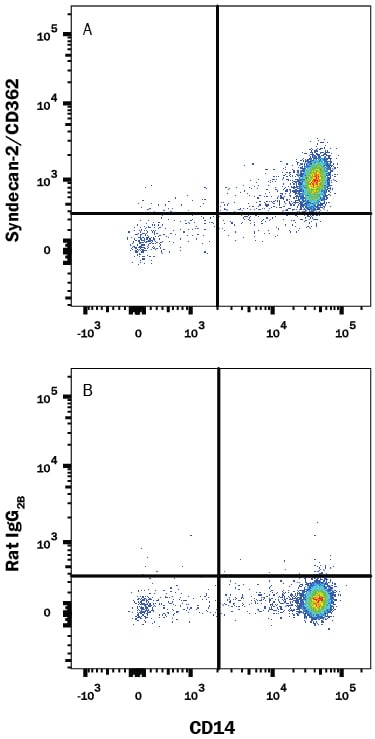
Detection of Syndecan-2/CD362 in Human Blood Monocytes by Flow Cytometry.
Human peripheral blood monocytes were stained with Mouse Anti-Human CD14 APC-conjugated Monoclonal Antibody (Catalog # FAB3832A) and either (A) Rat Anti-Human Syndecan-2/CD362 PE-conjugated Monoclonal Antibody (Catalog # FAB2965P) or (B) Rat IgG2BPhycoerythrin Isotype Control (Catalog # IC013P). View our protocol for Staining Membrane-associated Proteins.Applications for Human Syndecan-2/CD362 PE‑conjugated Antibody
Flow Cytometry
Sample: Human peripheral blood monocytes
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, 2 to 8 °C as supplied.
Background: Syndecan-2/CD362
References
- Tkachenko, E. et al. (2005) Circ. Res. 96:488.
- Oh, E.-S, and J. R. Couchman (2004) Mol. Cells 17:181.
- Essner, J. J. et al. (2006) Int. J. Biochem. Cell Biol. 38:152.
- Marynen, P. et al. (1989) J. Biol. Chem. 264:7017.
- Choi, S. et al. (2005) J. Biol. Chem. 280:42573.
- Park, H. et al. (2002) J. Biol. Chem. 277:29730.
- Clasper, S. et al. (1999) J. Biol. Chem. 274:24113.
- Modrowski, D. et al. (2000) J. Biol. Chem. 275:9178.
- Halden, Y. et al. (2004) Biochem. J. 377:533.
- Chen, L. et al. (2004) J. Biol. Chem. 279:15715.
Alternate Names
Gene Symbol
UniProt
Additional Syndecan-2/CD362 Products
Product Documents for Human Syndecan-2/CD362 PE‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Syndecan-2/CD362 PE‑conjugated Antibody
For research use only
Citations for Human Syndecan-2/CD362 PE‑conjugated Antibody
Customer Reviews for Human Syndecan-2/CD362 PE‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Human Syndecan-2/CD362 PE‑conjugated Antibody and earn rewards!
Have you used Human Syndecan-2/CD362 PE‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Associated Pathways
 VEGF - VEGF R2 Signaling Pathways
VEGF - VEGF R2 Signaling Pathways
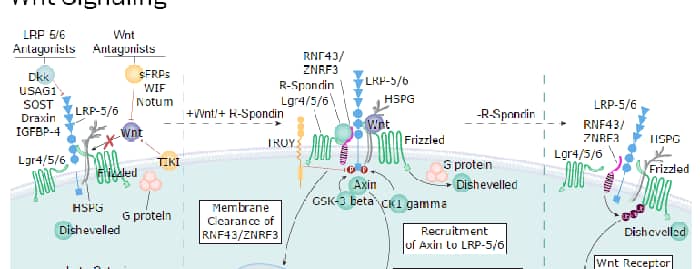
 Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling
Wnt Signaling Pathways: beta-Catenin-dependent Wnt Signaling