Syndecan-3, also called N-syndecan, is one of four vertebrate syndecans that are principal carriers of heparan sulfate and chondroitin sulfate glycosaminoglycans (GAGs) (1‑3). These type 1 transmembrane proteins show conserved cytoplasmic domains and divergent extracellular domains (1‑3). Human Syndecan-3 is synthesized as a 442 amino acid (aa) core protein with a 44 aa signal sequence, a 343 aa extracellular domain (ECD), a 21 aa transmembrane (TM) region and a 34 aa cytoplasmic tail with a binding site for PDZ domains (1). The ECD of human Syndecan-3 shares 83%, 83%, 92%, 91% and 91% aa identity with of mouse, rat, equine, bovine and canine Syndecan-3, respectively. Splice isoforms of 384 aa and 346 aa, containing either a 28 aa substitution for aa 1‑86 or deletion of aa 1‑96, have been reported (4). Syndecan-3 contains four conserved closely-spaced GAG attachment sites near the N-terminus and unique threonine-rich and mucin-like sequences near the membrane (4). Addition of glycan side chains results in an apparent size of 120‑150 kDa. Non-covalent homodimerization of Syndecan-3 or, potentially, heterodimerization with Syndecan-2 or -4, is dependent on the transmembrane domain (5). A cleavage site near the TM domain allows shedding of soluble ECD; the remainder of the molecule undergoes regulated intramembrane proteolysis (6). Syndecan-3 is expressed in the nervous system and at limb buds during development (1, 2). It is expressed on neuronal axons and Schwann cell perinodal processes, promoting nerve cell migration and synapse formation (7, 8). Roles in memory and body weight regulation have been described (2, 9, 10). Through localization of growth factors such as FGF2, HGF and TGF-beta, it regulates expression of molecules important for differentiation of muscle and bone, such as myogenin, BMP-2 and hedgehog family members (1, 2, 11‑13). In adults, it is upregulated during regeneration, such as following myocardial infarction (14).
Human Syndecan‑3 APC‑conjugated Antibody
R&D Systems | Catalog # FAB3539A

Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Product Specifications
Immunogen
Gln48-Lys383
Accession # O75056
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human Syndecan‑3 APC‑conjugated Antibody
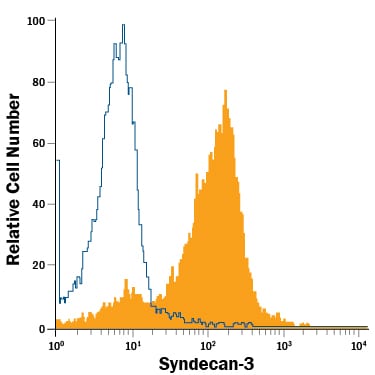
Detection of Syndecan‑3 in A172 Human Cell Line by Flow Cytometry.
A172 human glioblastoma cell line was stained with Goat Anti-Human Syndecan-3 APC-conjugated Antigen Affinity-purified Polyclonal Antibody (Catalog # FAB3539A, filled histogram) or isotype control antibody (Catalog # IC108A, open histogram). View our protocol for Staining Membrane-associated Proteins.Applications for Human Syndecan‑3 APC‑conjugated Antibody
Flow Cytometry
Sample: A172 human glioblastoma cell line
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
Background: Syndecan-3
References
- Tkachenko, E. et al. (2005) Circ. Res. 96:488.
- Reizes, O. et al. (2008) Int. J. Biochem. Cell Biol. 40:28.
- Carey, D.J. et al. (1997) J. Biol. Chem. 272:2873.
- ENTREZ protein Accession # O75056, EAX076736 and EAX07637.
- Dews, I.C. and K.R. MacKenzie (2007) Proc. Natl. Acad. Sci. USA 104:20782.
- Schultz, J.G. et al. (2003) J. Biol. Chem. 278:48651.
- Hienola, A. et al. (2006) J. Cell Biol. 174:569.
- Goutebroze, L. et al. (2003) BMC Neurosci. 4:29.
- Kaksonen, M. et al. (2002) Mol. Cell. Neurosci. 21:158.
- Strader. A.D. et al. (2004) J. Clin. Invest. 114:1354.
- Cornelison, D.D.W. et al. (2004) Genes Dev. 18:2231.
- Fisher, M.C. et al. (2006) Matrix Biol. 25:27.
- Pacifici, M. et al. (2005) J. Bone Miner. Metab. 23:191.
- Finsen, A.V. et al. (2004) Physiol. Genomics 16:301.
Alternate Names
Gene Symbol
UniProt
Additional Syndecan-3 Products
Product Documents for Human Syndecan‑3 APC‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human Syndecan‑3 APC‑conjugated Antibody
For research use only
Related Research Areas
Citations for Human Syndecan‑3 APC‑conjugated Antibody
Customer Reviews for Human Syndecan‑3 APC‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Human Syndecan‑3 APC‑conjugated Antibody and earn rewards!
Have you used Human Syndecan‑3 APC‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars