The Transferrin Receptor (TfR) also known as TfR-1 and CD71 is a type 2 transmembrane glycoprotein expressed on erythroid progenitors, muscle cells and proliferating cells as a 188 kDa disulfide-linked homodimer of 95 kDa monomers (1-4). As the major mediator of cellular iron uptake, it binds and internalizes diferric transferrin, allowing iron release at the low pH of the endosome (2, 5). The human TfR cDNA encodes 760 amino acids (aa) including a 67 aa N-terminal intracellular domain, a 21 aa transmembrane domain, and a 672 aa extracellular domain (ECD) with helical, peptidase (nonfunctional), and ligand binding domains, including an RGD potential integrin binding site (5). Human TfR ECD shares 75-80% aa identity with mouse, rat, feline, canine, equine, porcine and bovine TfR. A 679 aa alternately spliced form begins at aa 82 and is presumably secreted, while in an 804 aa form, 44 aa are inserted at aa 518 within the peptidase region (6). Most soluble TfR (sTfR) arises from trypsin proteolysis at aa 100, producing the circulating form of TfR (3). sTfR concentration in plasma or serum is proportional to total TfR and can be increased by iron deficiency (3). Erythroid progenitors, which use iron for hemoglobin synthesis, normally account for the bulk of total body TfR production (3). Since rapidly growing cells require iron to replicate DNA, cancer cells can express up to 5-fold more TfR than quiescent cells in the surrounding tissue (2, 4). Antibody targeting of TfR can inhibit tumor cell proliferation and induce apoptosis (2, 4). The hereditary hemochromatosis protein HFE competes with diferric transferrin for binding to TfR, and targets TfR for degradation rather than recycling (2, 5). TfR has been reported to have ferritin-independent functions in T cell development, immunological synapse formation and Galectin-3-mediated cell death, and to be a cell entry receptor for New World hemorrhagic fever arenaviruses (2, 4, 7).
Human TfR (Transferrin R) APC‑conjugated Antibody
R&D Systems | Catalog # FAB2474A

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Product Specifications
Immunogen
Specificity
Clonality
Host
Isotype
Scientific Data Images for Human TfR (Transferrin R) APC‑conjugated Antibody
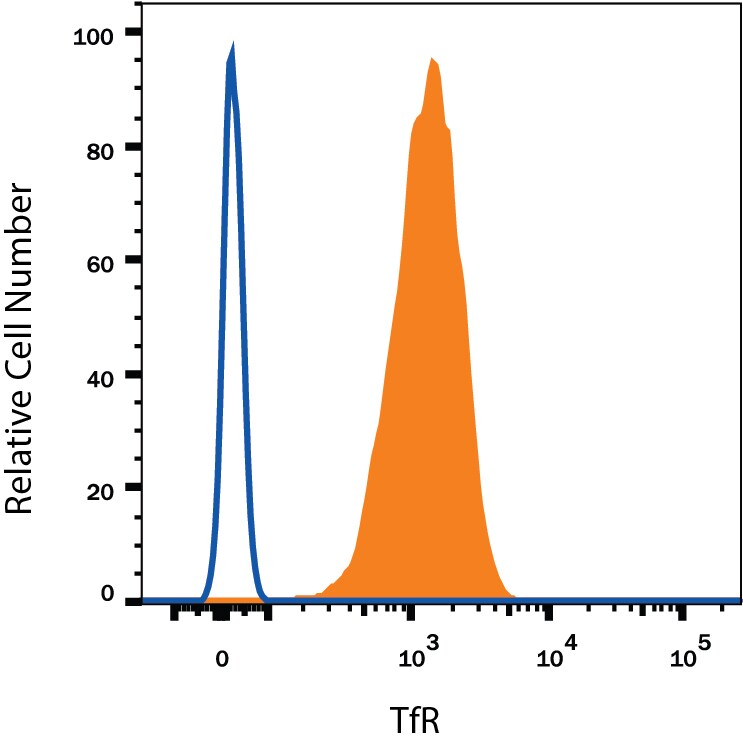
Detection of TfR (Transferrin R) in U937 Human Cell Line by Flow Cytometry.
U937 human histiocytic lymphoma cell line was stained with Mouse Anti-Human TfR (Transferrin R) APC-conjugated Monoclonal Antibody (Catalog # FAB2474A, filled histogram) or isotype control antibody (Catalog # IC002A, open histogram). View our protocol for Staining Membrane-associated Proteins.Applications for Human TfR (Transferrin R) APC‑conjugated Antibody
Flow Cytometry
Sample: U937 human histiocytic lymphoma cell line
Spectra Viewer
Plan Your Experiments
Use our spectra viewer to interactively plan your experiments, assessing multiplexing options. View the excitation and emission spectra for our fluorescent dye range and other commonly used dyes.
Spectra ViewerFlow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Shipping
Stability & Storage
- 12 months from date of receipt, 2 to 8 °C as supplied.
Background: TfR (Transferrin R)
References
- Schneider, C. et al. (1984) Nature 311:675.
- Daniels, T.R. et al. (2006) Clin. Immunol. 121:144.
- Skikne, B.S. (2008) Am. J. Hematol. 83:872.
- Macedo, M.F. and M. deSousa (2008) Inflamm. Allergy Drug Targets 7:41.
- Aisen, P. (2004) Int. J. Biochem. Cell Biol. 36:2137.
- Entrez protein Accession # EAW53671, EAW53672.
- Radoshitzky, S.R. et al. (2007) Nature 446:92.
Long Name
Alternate Names
Gene Symbol
Additional TfR (Transferrin R) Products
Product Documents for Human TfR (Transferrin R) APC‑conjugated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human TfR (Transferrin R) APC‑conjugated Antibody
For research use only
Citations for Human TfR (Transferrin R) APC‑conjugated Antibody
Customer Reviews for Human TfR (Transferrin R) APC‑conjugated Antibody
There are currently no reviews for this product. Be the first to review Human TfR (Transferrin R) APC‑conjugated Antibody and earn rewards!
Have you used Human TfR (Transferrin R) APC‑conjugated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Liperfluo
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- View all Protocols, Troubleshooting, Illustrated assays and Webinars