MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
Novus Biologicals | Catalog # MAB91871

Key Product Details
Species Reactivity
Applications
Label
Antibody Source
Format
Product Specifications
Immunogen
Modification
Clonality
Host
Isotype
Description
Scientific Data Images for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
Applications for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
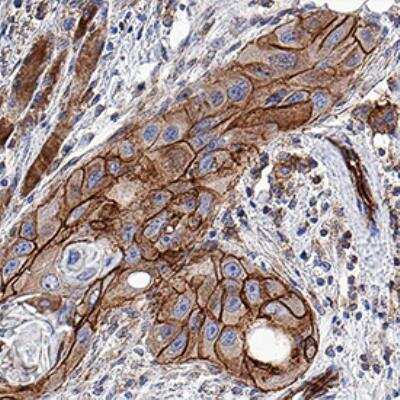
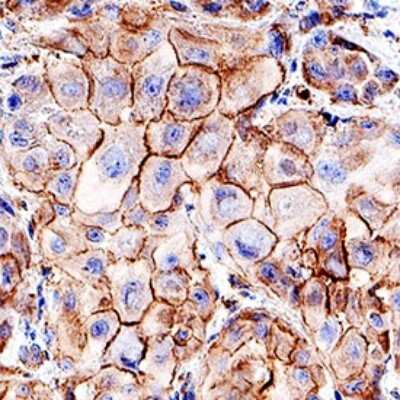
Immunohistochemistry
Immunohistochemistry-Paraffin
Reviewed Applications
Read 2 reviews rated 4.5 using MAB91871 in the following applications:
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: MLKL
Long Name
Alternate Names
Gene Symbol
Additional MLKL Products
Product Documents for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
Customer Reviews for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free (2)
Have you used MLKL [p Thr357] Antibody (954724) - Azide and BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Simple WesternSample Tested: HT-29 human colon adenocarcinoma cell line and MC-38Species: Human and MouseVerified Customer | Posted 10/16/2020pMLKL AbBio-Techne ResponseThis review was submitted through the legacy Novus Innovators Program, reflecting a new species or application tested on a primary antibody.
-
Application: Immunohistochemistry-ParaffinSample Tested: FFPESpecies: HumanVerified Customer | Posted 10/09/202020X
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for MLKL [p Thr357] Antibody (954724) - Azide and BSA Free
-
Q: hello! we're working with MAB91871. the IF staining is looking pretty good, but occasionally we are finding bright specular artifacts. From the secondary controls we have narrow it down to being due to the presence of the primary - do y'all have any handling data on this product and how to minimize aggregates?
A: Here are some general suggestions to try and reduce aggregation background signal in your organoid IF staining with MAB91871.
- This is a purified antibody with no BSA, so it ought not to be an issue with buffer but its possible it's could be sticky.
Do you use 0.05-0.1 % Triton X 100? - You might try adding NaCl to the blocking buffer/antibody diluent so that the final concentration is between 0.15 M and 0.6 M NaCl. The best NaCl concentration to use will have to be determined empirically, but this should help reduce ionic interactions and keep the AB soluble.
- If it's a blocking issue, you could of course increase the blocking buffer composition and/or concentration.
- It's possible that high primary AB concentration is leading to non-specific binding and background staining, so you might try reducing the final concentration of the primary antibody used.
- This is a purified antibody with no BSA, so it ought not to be an issue with buffer but its possible it's could be sticky.

![MLKL [p Thr357] Antibody (954724) - Azide and BSA Free MAB91871](https://resources.rndsystems.com/images/reviews/review_mab91871_54221_0_0_0_0_0.png)
![MLKL [p Thr357] Antibody (954724) - Azide and BSA Free MAB91871](https://resources.rndsystems.com/images/reviews/review_mab91871_53971_0_0.png)