EphB6, also known as Mep (1), is a member of the Eph receptor family which binds members of the ephrin ligand family. There are two classes of receptors, designated A and B. Both the A and B class receptors have an extracellular region consisting of a globular domain, a cysteine-rich domain, and two fibronectin type III domains. This is followed by the transmembrane region and cytoplasmic region. The cytoplasmic region contains a juxtamembrane motif with two tyrosine residues, which are the major autophosphorylation sites, a kinase domain, and a conserved sterile alpha motif (SAM) in the carboxy tail which contains one conserved tyrosine residue. Activation of kinase activity occurs after ligand recognition and binding. However, it has been found that EphB6 contains substitutions within the kinase domain which results in EphB6 having no kinase activity (4). The ligands which bind EphB6 are unknown (2, 3). However, we have observed that the ephrin-B1 and ephrin-B2 ligands can bind the immobilized receptor in an ELISA-type assay. The extracellular domains of human and mouse EphB6 share 92% amino acid identity. Only membrane-bound or Fc-clustered ligands are capable of activating the receptor in vitro. While soluble monomeric ligands bind the receptor, they do not induce receptor autophosphorylation and activation (2). In vivo, the ligands and receptors display reciprocal expression (3). It has been found that nearly all receptors and ligands are expressed in developing and adult neural tissue (3). The Eph/ephrin families also appear to play a role in angiogenesis (3).

Key Product Details
Species Reactivity
Mouse
Applications
Western Blot
Label
Biotin
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
Mouse myeloma cell line NS0-derived recombinant mouse EphB6
Leu33-Ser587
Accession # O08644
Leu33-Ser587
Accession # O08644
Specificity
Detects mouse EphB6 in Western blots. In Western blots, less than 1% cross-reactivity with recombinant human (rh) EphA1, recombinant mouse (rm) EphA3, rmEphA4, rmEphrin-B1, rmEphA6, rmEphA7, recombinant rat EphA5, rmEphrin-B2, rmEphB1, rmEphB2, rmEphB3, rmEphB4, rrEphB1, rmEphA8, rmEphA2, and rhEphrin-B3 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Mouse EphB6 Biotinylated Antibody
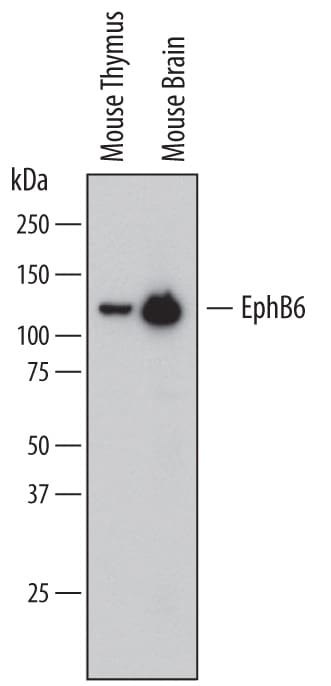
Detection of Mouse EphB6 by Western Blot.
Western blot shows lysates of mouse thymus tissue and mouse brain tissue. PVDF membrane was probed with 1 µg/mL of Goat Anti-Mouse EphB6 Biotinylated Antigen Affinity-purified Polyclonal Antibody (Catalog # BAF611) followed by Streptavidin-HRP (Catalog # DY998). A specific band was detected for EphB6 at approximately 120 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.Applications for Mouse EphB6 Biotinylated Antibody
Application
Recommended Usage
Western Blot
1 µg/mL
Sample: Mouse thymus tissue and mouse brain tissue
Sample: Mouse thymus tissue and mouse brain tissue
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS.
Loading...
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with BSA as a carrier protein.
Shipping
The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: EphB6
References
- Eph Nomenclature Committee [letter] (1997) Cell 90:403.
- Flanagan, J.G. and P. Vanderhaeghen (1998) Annu. Rev. Neurosci. 21:309.
- Pasquale, E.B. (1997) Curr. Opin. Cell Biol. 9:608.
- Gurniak, C.B. and L.J. Berg (1996) Oncogene 13:777.
Long Name
Eph Receptor B6
Alternate Names
Hep, Mep
Gene Symbol
EPHB6
UniProt
Additional EphB6 Products
Product Documents for Mouse EphB6 Biotinylated Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Mouse EphB6 Biotinylated Antibody
For research use only
Customer Reviews for Mouse EphB6 Biotinylated Antibody
There are currently no reviews for this product. Be the first to review Mouse EphB6 Biotinylated Antibody and earn rewards!
Have you used Mouse EphB6 Biotinylated Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Cellular Response to Hypoxia Protocols
- R&D Systems Quality Control Western Blot Protocol
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
