Growth differentiation factor-9 (GDF-9) is a member of the transforming growth factor-beta (TGF-beta ) superfamily, and is an oocyte secreted paracrine factor essential for mammalian ovarian folliculogenesis (1‑2). Mouse GDF-9 is synthesized as a 441 amino acid (aa) prepropeptide that contains a 29 aa signal sequence, a 277 aa propeptide, and a 135 aa mature chain. Residues 340‑441 constitute a TGF-beta like domain. In addition, there is one potential site of N-linked glycosylation in the mature chain. Unlike other members of the TGF-beta superfamily, GDF‑9 lacks the conserved cysteine residue that is believed to form the sole disulfide linkage between subunits in other family members (3). Mature mouse GDF-9 shares 90% aa sequence identity with mature human GDF 9. The protein is expressed throughout the development of the maturing follicle (2). GDF-9 functions as a paracrine factor in the regulation of granulosa cell proliferation and differentiation, and is essential for fertility (2, 4). Studies on GDF-9 null mice have demonstrated arrested follicular development at the primary follicle stage (5). Mouse GDF-9 induces Smad2 phosphorylation and inhibin production in rat diethylstilbestrol treated granulosa cells (6) and in human granulosa-luteal cells (7). The downstream signaling actions of GDF 9 are mediated by the type I receptor, activin receptor-like kinase 5 (ALK5), initiating the subsequent activation of Smad2 and Smad3 (2, 8). GDF 9 uses the BMP type II receptor (BMPRII) as its other signaling receptor (2, 9).
Human/Mouse/Hamster GDF‑9 Antibody
R&D Systems | Catalog # AF739

Discontinued Product
AF739 has been discontinued.
View all GDF-9 products.
Key Product Details
Species Reactivity
Validated:
Human, Mouse, Hamster
Cited:
Human, Mouse
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen
Label
Unconjugated
Antibody Source
Polyclonal Goat IgG
Loading...
Product Specifications
Immunogen
E. coli-derived recombinant mouse GDF-9
Specificity
Detects mouse GDF‑9 in direct ELISAs and human, mouse and hamster GDF-9 in Western blots. In direct ELISAs, less than 1% cross‑reactivity with recombinant mouse (rm) GDF-1, rmGDF-5, rmGDF-6, rmGDF-8, and rmGDF-15 is observed.
Clonality
Polyclonal
Host
Goat
Isotype
IgG
Scientific Data Images for Human/Mouse/Hamster GDF‑9 Antibody
Detection of Human/Hamster GDF‑9 by Western Blot.
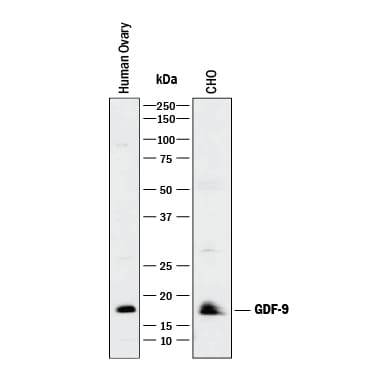
Western blot shows lysates of human ovary tissue and CHO Chinese hamster ovary cell line. PVDF membrane was probed with 1 µg/mL of Goat Anti-Human/Mouse/Hamster GDF-9 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF739) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (HAF017). A specific band was detected for GDF-9 at approximately 18 kDa (as indicated). This experiment was conducted under reducing conditions and using Immunoblot Buffer Group 1.GDF‑9 in Mouse Ovary.
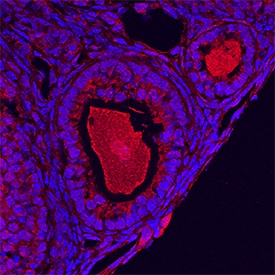
GDF-9 was detected in perfusion fixed frozen sections of mouse ovary using Goat Anti-Human/Mouse/Hamster GDF-9 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF739) at 5 µg/mL overnight at 4 °C. Tissue was stained using the NorthernLights™ 557-conjugated Anti-Goat IgG Secondary Antibody (red; NL001) and counterstained with DAPI (blue). Specific staining was localized to developing oocytes. View our protocol for Fluorescent IHC Staining of Frozen Tissue Sections.Detection of Mouse GDF‑9 by Western Blot.
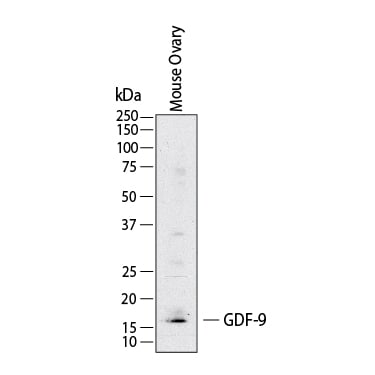
Western Blot shows lysates of mouse ovary. PVDF membrane was probed with 1 µg/ml of Goat Anti-Human/Mouse/Hamster GDF‑9 Antigen Affinity-purified Polyclonal Antibody (Catalog # AF739) followed by HRP-conjugated Anti-Goat IgG Secondary Antibody (Catalog # HAF017). A specific band was detected for GDF‑9 at approximately 17 kDa (as indicated). This experiment was conducted under reducing conditions and using Western Blot Buffer Group 1.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
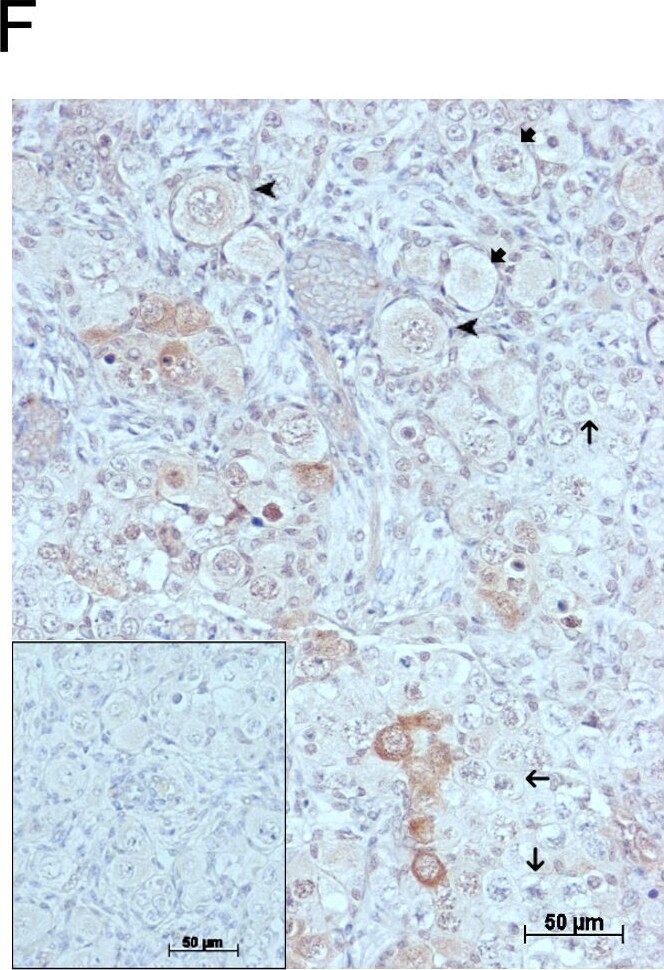
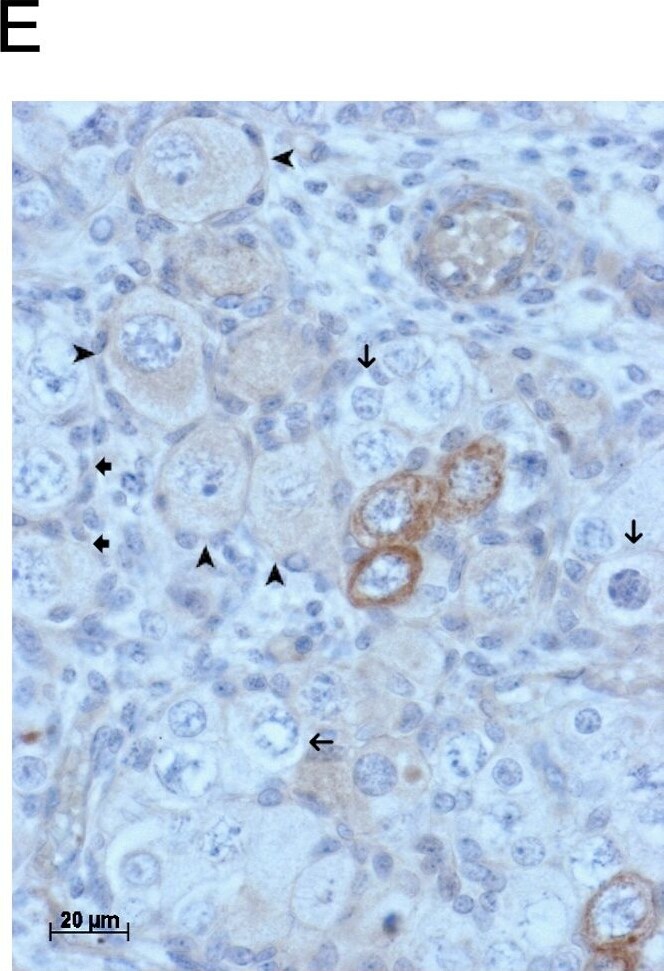
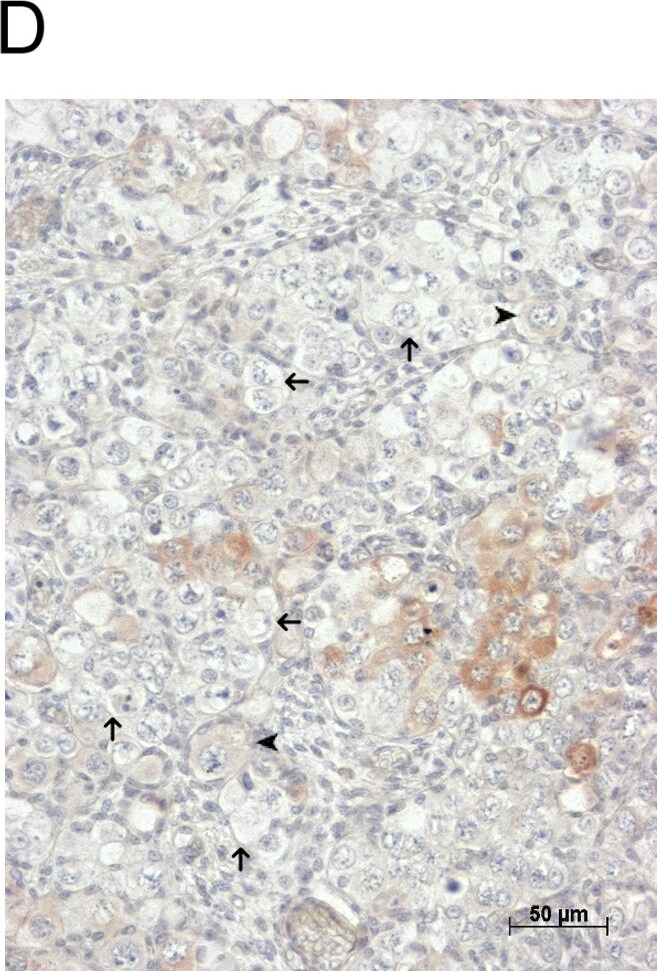
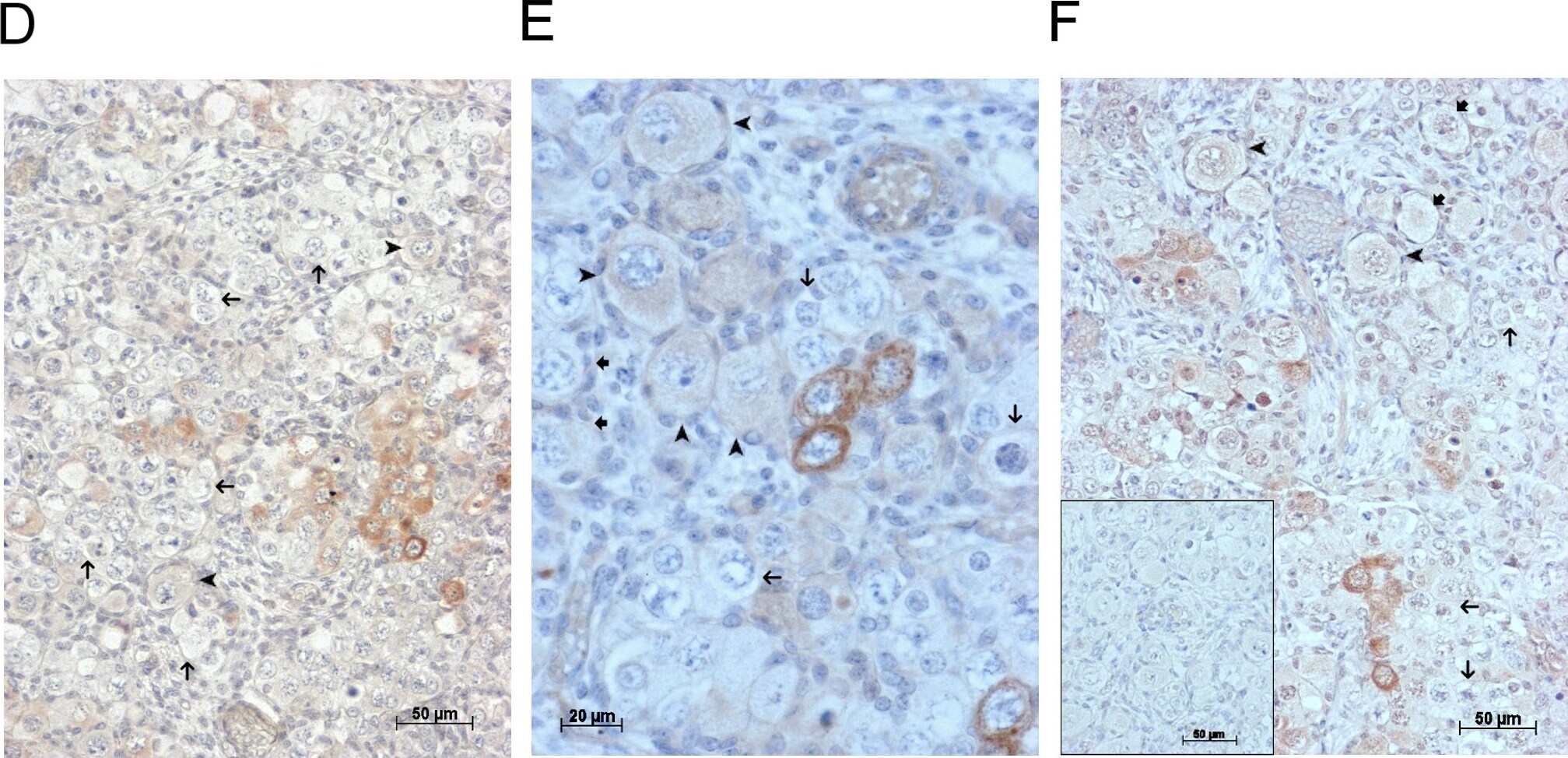
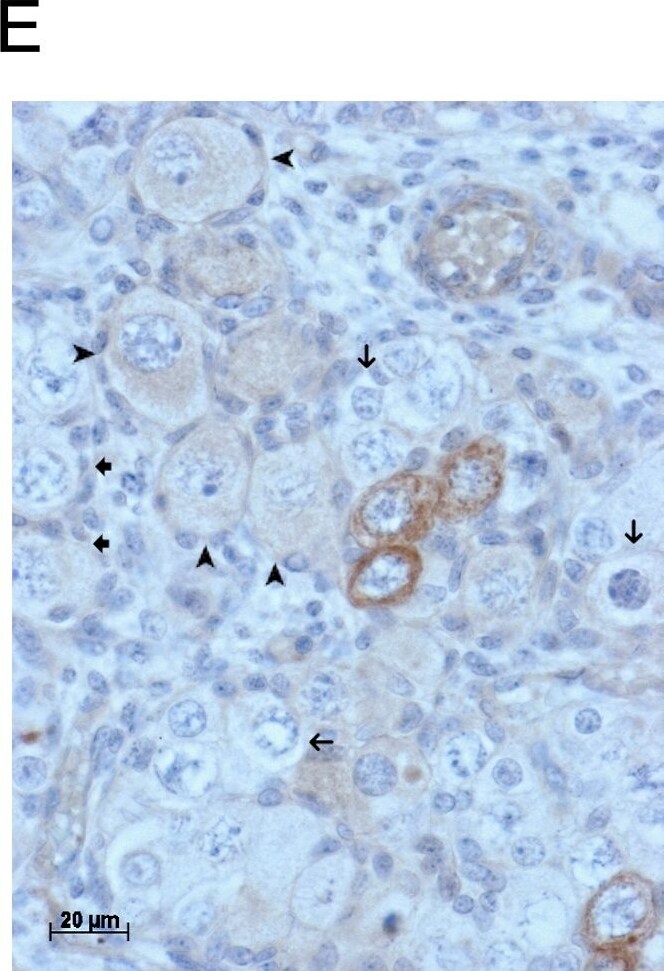
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunocytochemistry/ Immunofluorescence
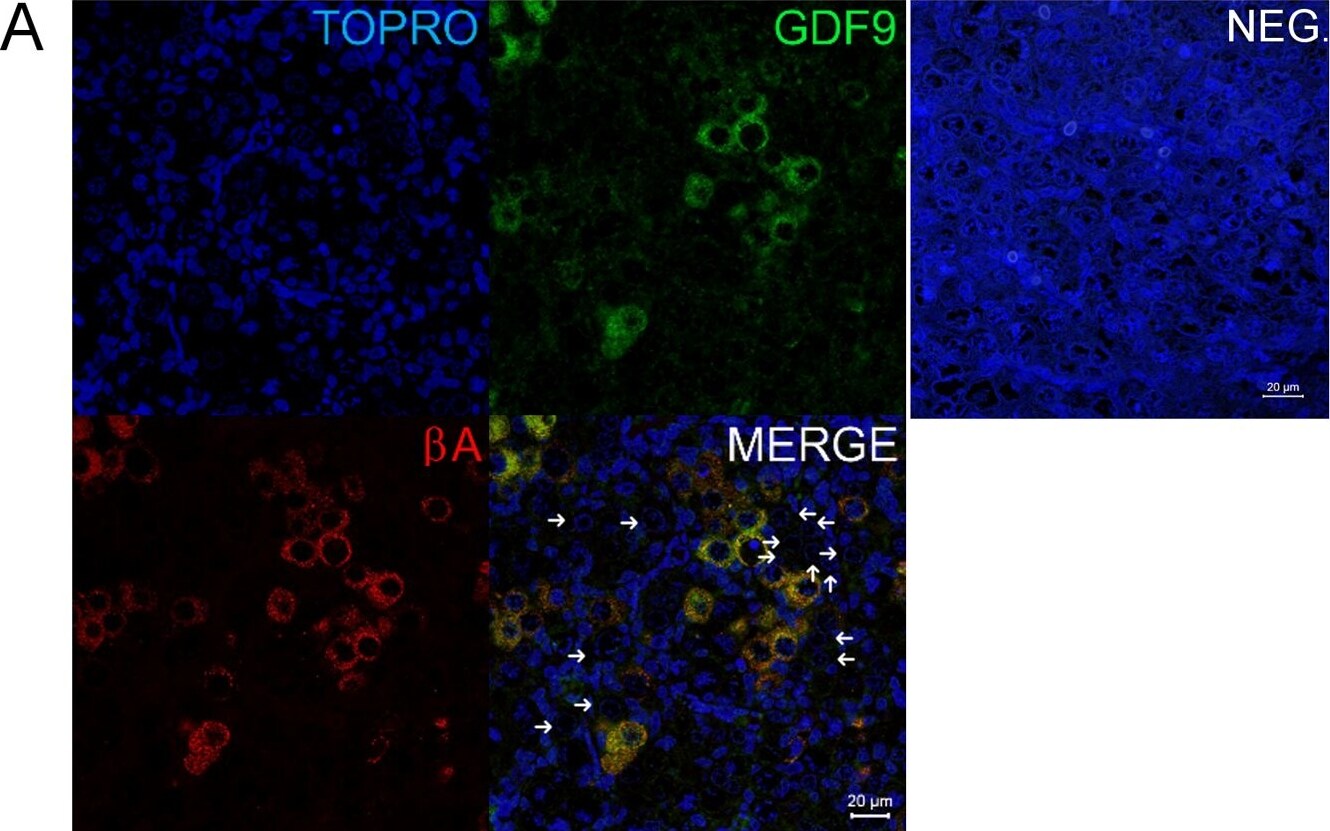
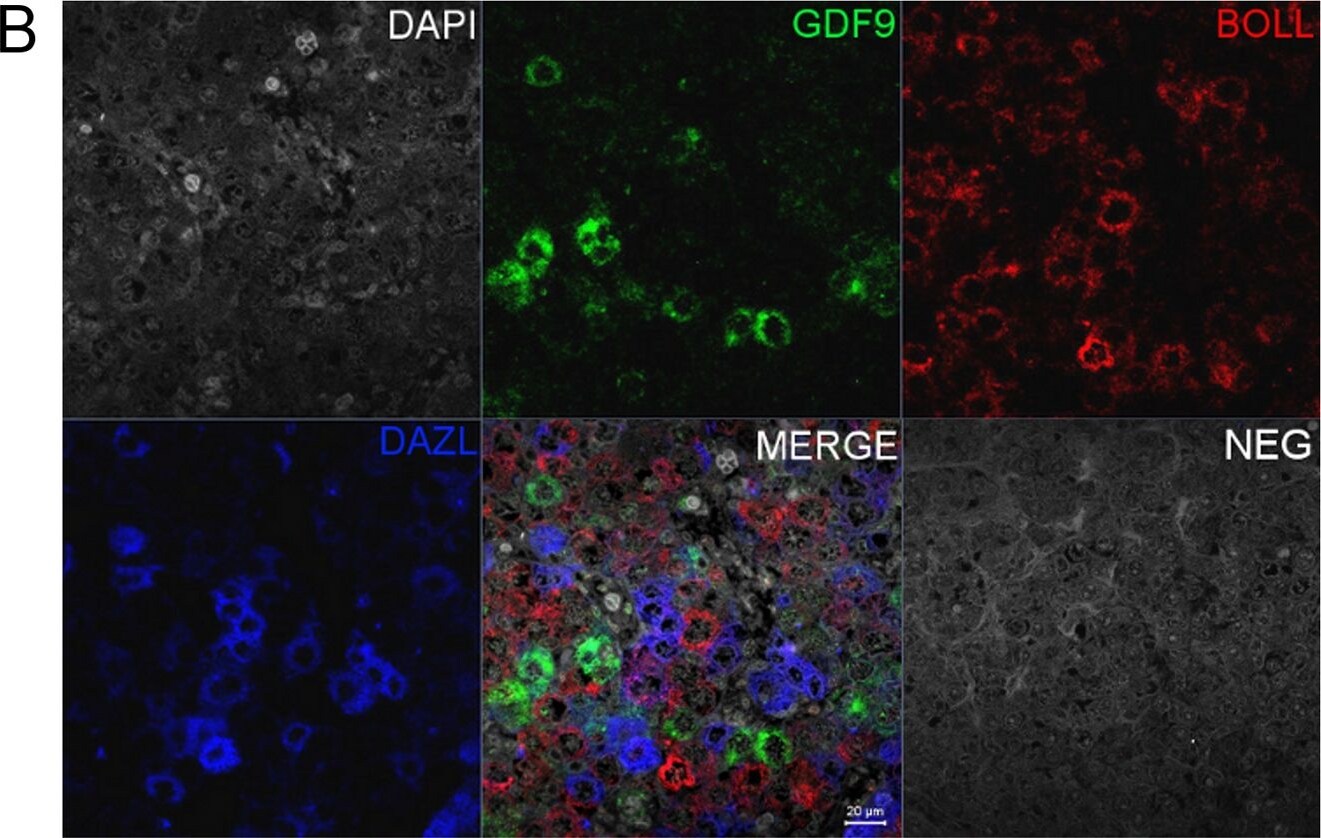
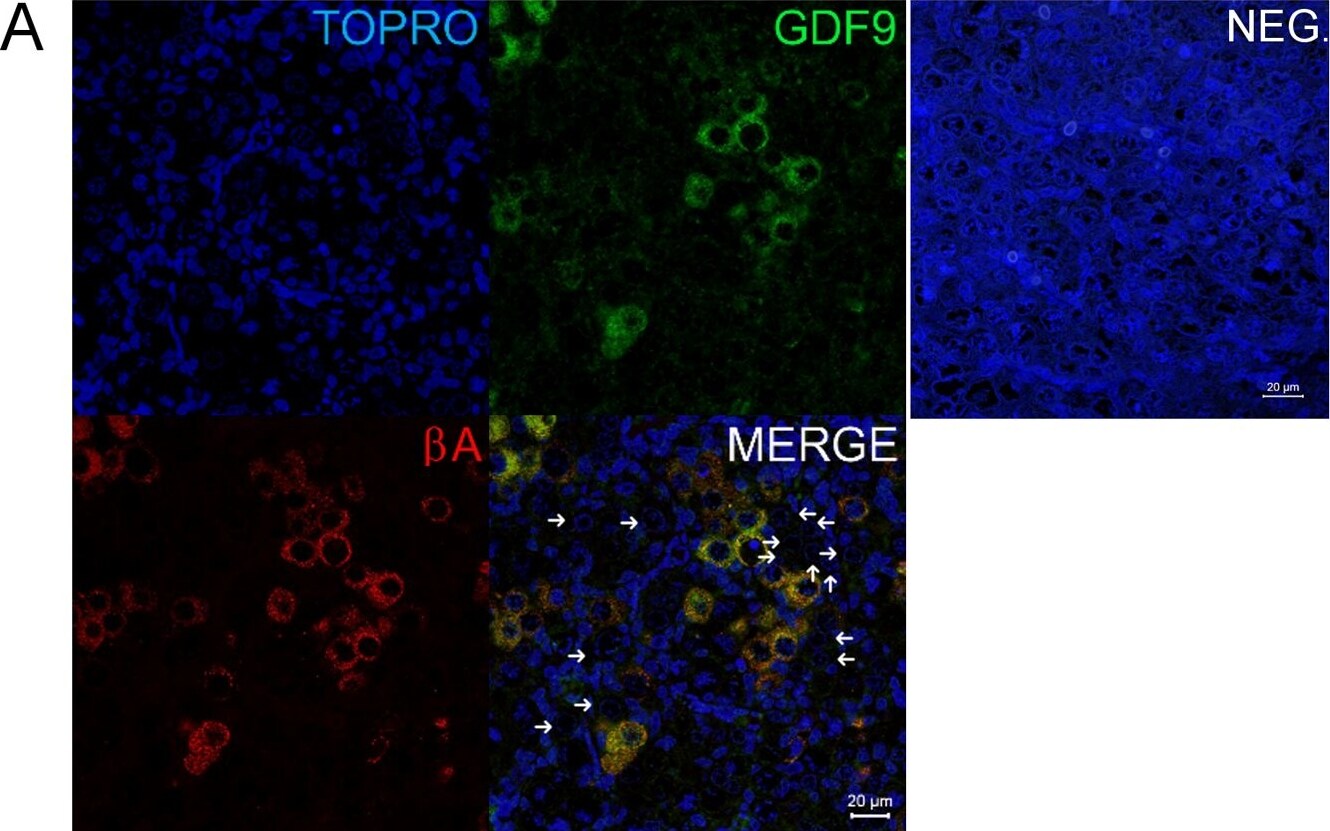
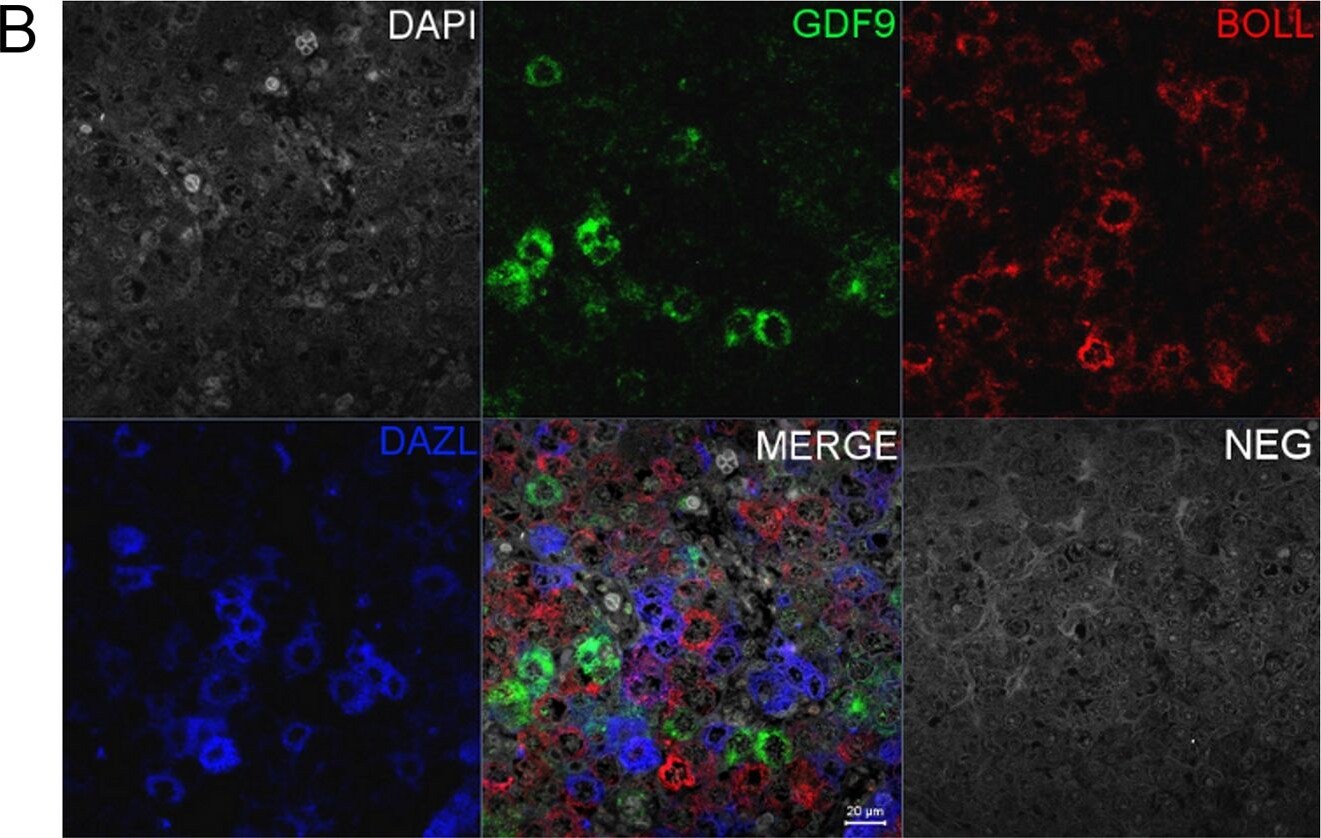
Co-localisation of GDF9 with activin beta A but not DAZL or BOLL prior to follicle formation.(A) Double immunohistochemistry of 18 week fetal ovary stained for GDF9 (green) and activin beta A (red), thus in the merged image co-expression is yellow. Unstained germ cells are indicated with arrows. Counterstain is TOPRO. (B) Triple fluorescent immunohistochemistry for GDF9 (green), DAZL (blue) and BOLL (red) in 20 week human fetal ovary with DAPI as counterstain (grey). Split channel and merged images in (A) and (B) are shown as are merged images of non-immune serum negative control (NEG). Scale bars are 20μm. (C) Nuclear diameters of DAZL, BOLL and GDF9 stained germ cells indicates that GDF9 positive cells are significantly larger (p<0.001) than DAZL but not BOLL expressing cells (bars indicate mean ± sem). (D) Higher magnification merged image of GDF9/DAZL/BOLL immunohistochemistry showing one large primordial follicle is positive for both GDF9 and DAZL but other follicles are positive only for DAZL. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunocytochemistry/ Immunofluorescence
Co-localisation of GDF9 with activin beta A but not DAZL or BOLL prior to follicle formation.(A) Double immunohistochemistry of 18 week fetal ovary stained for GDF9 (green) and activin beta A (red), thus in the merged image co-expression is yellow. Unstained germ cells are indicated with arrows. Counterstain is TOPRO. (B) Triple fluorescent immunohistochemistry for GDF9 (green), DAZL (blue) and BOLL (red) in 20 week human fetal ovary with DAPI as counterstain (grey). Split channel and merged images in (A) and (B) are shown as are merged images of non-immune serum negative control (NEG). Scale bars are 20μm. (C) Nuclear diameters of DAZL, BOLL and GDF9 stained germ cells indicates that GDF9 positive cells are significantly larger (p<0.001) than DAZL but not BOLL expressing cells (bars indicate mean ± sem). (D) Higher magnification merged image of GDF9/DAZL/BOLL immunohistochemistry showing one large primordial follicle is positive for both GDF9 and DAZL but other follicles are positive only for DAZL. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry
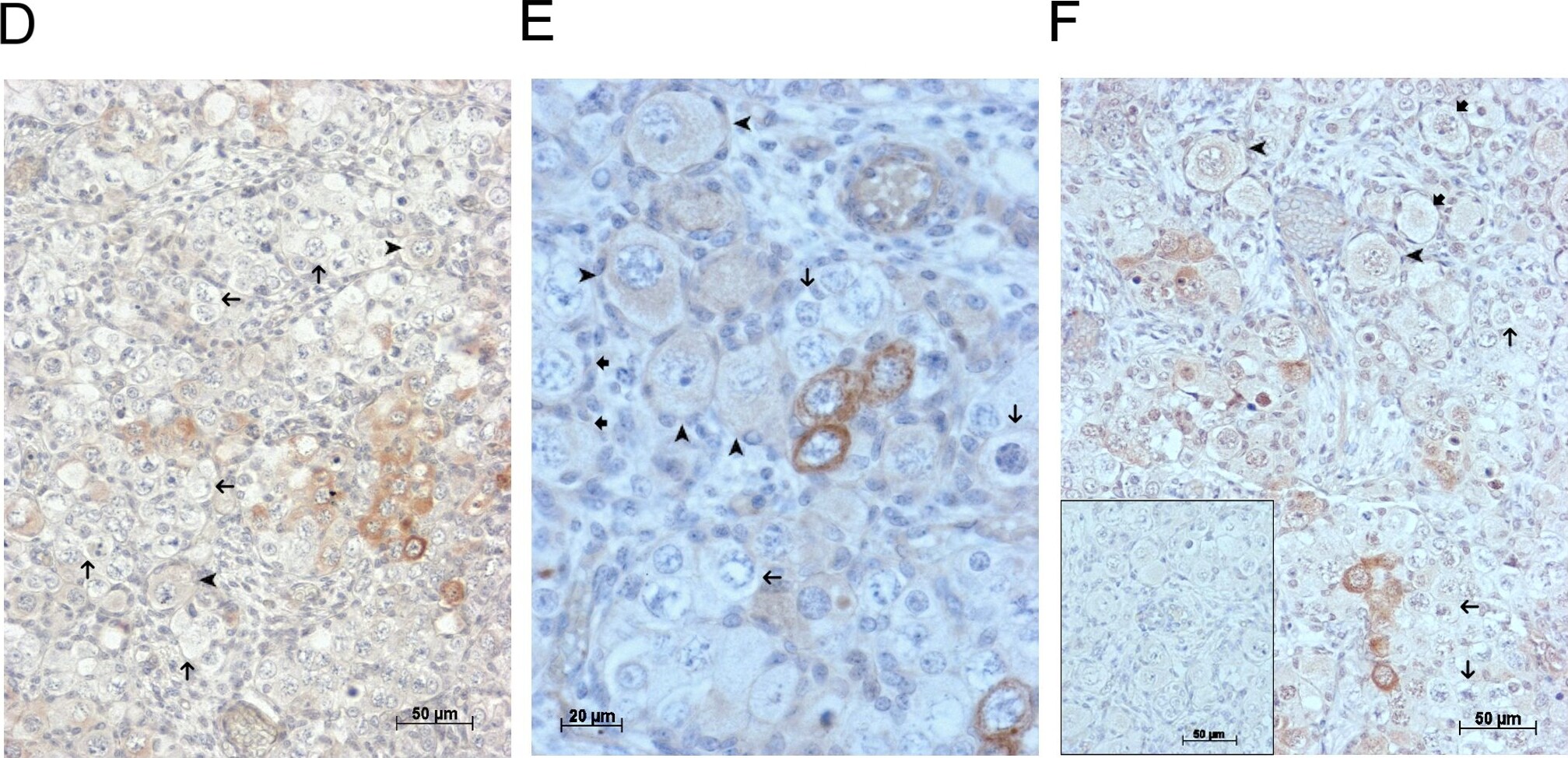
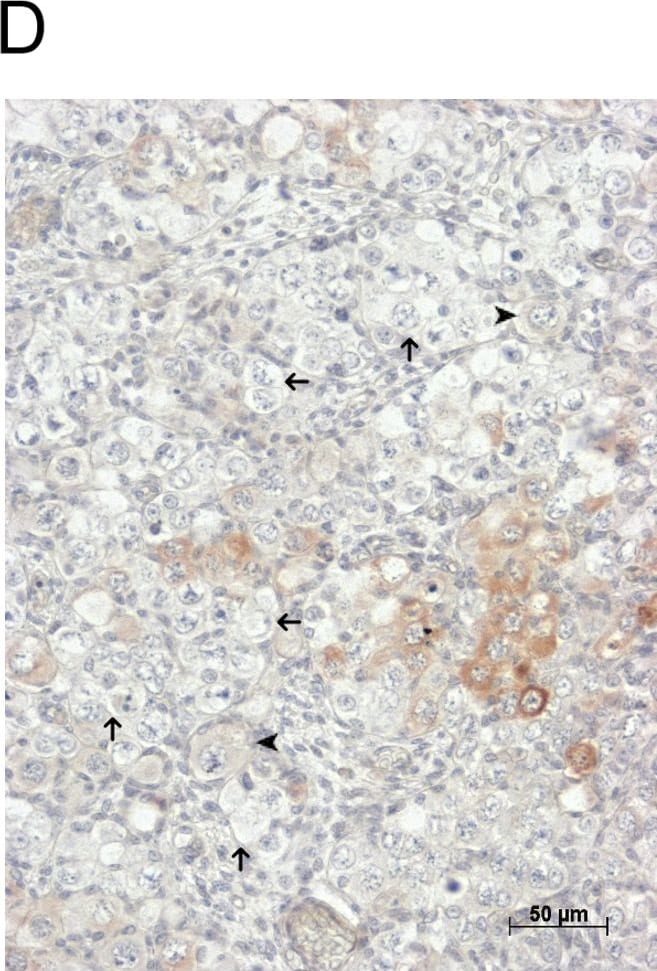
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
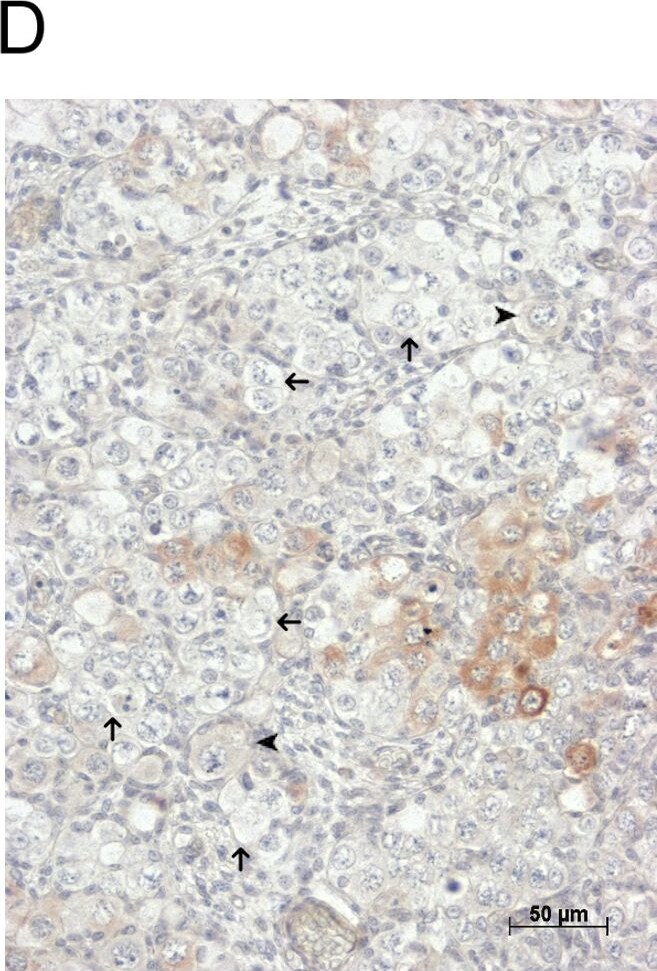
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunocytochemistry/ Immunofluorescence
Co-localisation of GDF9 with activin beta A but not DAZL or BOLL prior to follicle formation.(A) Double immunohistochemistry of 18 week fetal ovary stained for GDF9 (green) and activin beta A (red), thus in the merged image co-expression is yellow. Unstained germ cells are indicated with arrows. Counterstain is TOPRO. (B) Triple fluorescent immunohistochemistry for GDF9 (green), DAZL (blue) and BOLL (red) in 20 week human fetal ovary with DAPI as counterstain (grey). Split channel and merged images in (A) and (B) are shown as are merged images of non-immune serum negative control (NEG). Scale bars are 20μm. (C) Nuclear diameters of DAZL, BOLL and GDF9 stained germ cells indicates that GDF9 positive cells are significantly larger (p<0.001) than DAZL but not BOLL expressing cells (bars indicate mean ± sem). (D) Higher magnification merged image of GDF9/DAZL/BOLL immunohistochemistry showing one large primordial follicle is positive for both GDF9 and DAZL but other follicles are positive only for DAZL. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunocytochemistry/ Immunofluorescence
Co-localisation of GDF9 with activin beta A but not DAZL or BOLL prior to follicle formation.(A) Double immunohistochemistry of 18 week fetal ovary stained for GDF9 (green) and activin beta A (red), thus in the merged image co-expression is yellow. Unstained germ cells are indicated with arrows. Counterstain is TOPRO. (B) Triple fluorescent immunohistochemistry for GDF9 (green), DAZL (blue) and BOLL (red) in 20 week human fetal ovary with DAPI as counterstain (grey). Split channel and merged images in (A) and (B) are shown as are merged images of non-immune serum negative control (NEG). Scale bars are 20μm. (C) Nuclear diameters of DAZL, BOLL and GDF9 stained germ cells indicates that GDF9 positive cells are significantly larger (p<0.001) than DAZL but not BOLL expressing cells (bars indicate mean ± sem). (D) Higher magnification merged image of GDF9/DAZL/BOLL immunohistochemistry showing one large primordial follicle is positive for both GDF9 and DAZL but other follicles are positive only for DAZL. Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Detection of Human GDF-9 by Immunohistochemistry-Paraffin
GDF9 is expressed in the human fetal ovary.qRT-PCR analysis of GDF9 (A), BMP15 (B) and NOBOX (C) mRNA expression in human fetal ovary across the gestational range of 8 to 20 weeks. Ovaries (n = 5–7 per group) were grouped according to developmental stage and transcript levels measured relative to those of RPL32. Bars indicate mean±sem. Statistically different levels are indicated by asterisks above the columns, thus expression of GDF9 at 18–20 weeks was significantly higher than at 8–11 weeks (p<0.005) as was expression of BMP15 and of NOBOX (both p<0.01). DAB immunohistochemical detection of GDF9: 19 week (D, E) and 20 week (F) human fetal ovary stained with anti-GDF9 antibody or normal goat IgG negative control (F inset)—positive staining is brown. Thick arrows indicate primordial follicles and thin arrows germ cells that are not stained for GDF9 while the arrowheads indicate primordial follicles that are positive for GDF9. Scale bars are 50μm (D and F) and 20μm (E). Image collected and cropped by CiteAb from the following open publication (https://pubmed.ncbi.nlm.nih.gov/25790371), licensed under a CC-BY license. Not internally tested by R&D Systems.Applications for Human/Mouse/Hamster GDF‑9 Antibody
Application
Recommended Usage
Immunohistochemistry
5-15 µg/mL
Sample: Immersion fixed frozen sections of mouse embryo (E11-13) and perfusion fixed frozen sections of mouse ovary
Sample: Immersion fixed frozen sections of mouse embryo (E11-13) and perfusion fixed frozen sections of mouse ovary
Western Blot
1 µg/mL
Sample: Human ovary tissue, mouse ovary tissue, and CHO Chinese hamster ovary cell line
Sample: Human ovary tissue, mouse ovary tissue, and CHO Chinese hamster ovary cell line
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
Reconstitute at 0.2 mg/mL in sterile PBS. For liquid material, refer to CoA for concentration.
Formulation
Lyophilized from a 0.2 μm filtered solution in PBS with Trehalose. *Small pack size (SP) is supplied either lyophilized or as a 0.2 µm filtered solution in PBS.
Shipping
Lyophilized product is shipped at ambient temperature. Liquid small pack size (-SP) is shipped with polar packs. Upon receipt, store immediately at the temperature recommended below.
Stability & Storage
Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
- 12 months from date of receipt, -20 to -70 °C as supplied.
- 1 month, 2 to 8 °C under sterile conditions after reconstitution.
- 6 months, -20 to -70 °C under sterile conditions after reconstitution.
Calculators
Background: GDF-9
References
- McGrath, S.A. et al. (1995) Mol. Endocrinol. 9:131.
- Mottershead, D.G. et al. (2008) Mol. Cell. Endocrinol. 283:58.
- McPherron, A.C. and S.-J. Lee (1992) J. Biol. Chem. 268:3444.
- Gilchrist, R.B. et al. (2006) J. Cell. Sci. 119:3811.
- Dong, J. et al. (1996) Nature 383:531.
- Roh, J.S. et al. (2003) Endocrinology 144:172.
- Kaivo-Oja, N. et al. (2003) J. Clin. Endocrinol. Metab. 88:755.
- Mazerbourg, S. et al. (2004) Mol. Endocrinol. 18:653.
- Vitt, U.A. et al. (2002) Biol. Reprod. 67:473.
Long Name
Growth Differentiation Factor 9
Alternate Names
GDF9
Gene Symbol
GDF9
Additional GDF-9 Products
Product Documents for Human/Mouse/Hamster GDF‑9 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Human/Mouse/Hamster GDF‑9 Antibody
For research use only
Related Research Areas
Citations for Human/Mouse/Hamster GDF‑9 Antibody
Customer Reviews for Human/Mouse/Hamster GDF‑9 Antibody
There are currently no reviews for this product. Be the first to review Human/Mouse/Hamster GDF‑9 Antibody and earn rewards!
Have you used Human/Mouse/Hamster GDF‑9 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...