Rat Glycine R Pan Specific Antibody
R&D Systems | Catalog # PPS009

Discontinued Product
PPS009 has been discontinued.
View all Glycine R products.
Key Product Details
Species Reactivity
Rat
Applications
Immunohistochemistry, Western Blot
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Peptide from the N-terminus region of the alpha 1 subunit of the rat Glycine Receptor
Specificity
Specific for the ~48 kDa alpha 1 and alpha 2 subunits of the Glycine Receptor in Western blots of rat spinal cord, brain stem and cell extracts. Immunolabeling is blocked by preadsorption of antibody with the peptide immunogen. This antibody does not recognize other Glycine Receptor subunits.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Scientific Data Images for Rat Glycine R Pan Specific Antibody
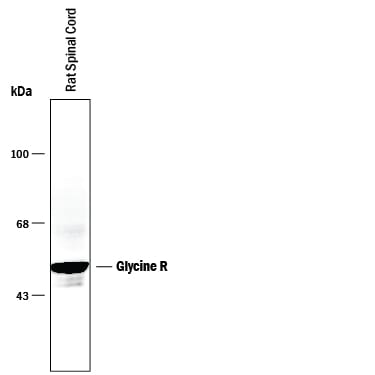
Detection of Rat Glycine R by Western Blot.
Western blot of 10 μg of rat spinal cord. As shown in the autoradiograph, the Anti-Glycine Receptor antibody is specific for the ~48 kDa subunits recognizing both the alpha 1 and alpha 2 subunits of the Glycine Receptor.Applications for Rat Glycine R Pan Specific Antibody
Application
Recommended Usage
Immunohistochemistry
1:1000 dilution
Sample: Frozen sections; unpublished observations, 1:1000
Sample: Frozen sections; unpublished observations, 1:1000
Western Blot
1:1000 dilution
Sample: Rat spinal cord lysate
Sample: Rat spinal cord lysate
Formulation, Preparation, and Storage
Purification
Antigen Affinity-purified
Reconstitution
This antibody should be reconstituted in 100 μL phosphate buffered saline (137 mM NaCl, 7.5 mM Na2HPO4, 2.7 mM KCl, 1.5 mM KH2PO4, pH 7.4) before use.
Formulation
Lyophilized in 5 mM ammonium bicarbonate
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
The lyophilized product is stable at ≤- 20° C for at least 1 year. After reconstitution the antibody should be aliquoted and stored at ≤ -20° C.
Calculators
Background: Glycine R
Product Documents for Rat Glycine R Pan Specific Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Product Specific Notices for Rat Glycine R Pan Specific Antibody
For research use only
Related Research Areas
Customer Reviews for Rat Glycine R Pan Specific Antibody
There are currently no reviews for this product. Be the first to review Rat Glycine R Pan Specific Antibody and earn rewards!
Have you used Rat Glycine R Pan Specific Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
