Interleukin-2 (IL-2) is a O-glycosylated, four alpha -helix bundle cytokine that has potent stimulatory activity for antigen-activated T cells. It is expressed by CD4+ and CD8+ T cells, gamma delta T cells, B cells, dendritic cells, and eosinophils (1-3). Mature human IL-2 shares 56% and 66% aa sequence identity with mouse and rat IL-2, respectively. Human and mouse IL-2 exhibit cross-species activity (4). The receptor for IL-2 consists of three subunits that are present on the cell surface in varying preformed complexes (5-7). The 55 kDa IL-2 R alpha is specific for IL-2 and binds with low affinity. The 75 kDa IL-2 R beta, which is also a component of the IL-15 receptor, binds IL-2 with intermediate affinity. The 64 kDa common gamma chain gamma c/IL-2 R gamma, which is shared with the receptors for IL-4, -7, -9, -15, and -21, does not independently interact with IL-2. Upon ligand binding, signal transduction is performed by both IL-2 R beta and gamma c. IL-2 is best known for its autocrine and paracrine activity on T cells. It drives resting T cells to proliferate and induces IL-2 and IL-2 R alpha synthesis (1, 2). It contributes to T cell homeostasis by promoting the Fas-induced death of naïve CD4+ T cells but not activated CD4+ memory lymphocytes (8). IL-2 plays a central role in the expansion and maintenance of regulatory T cells, although it inhibits the development of Th17 polarized cells (9-11). Thus, IL-2 may be a key cytokine in the natural suppression of autoimmunity (12, 13).
GMP
Recombinant Human IL-2 GMP Protein, CF
R&D Systems | Catalog # 202-GMP
Animal-Free.

Discontinued Product
202-GMP has been discontinued.
An alternative/replacement product is available:
BT-002-GMP. View all IL-2 products.
Key Product Details
Source
E. coli
Accession Number
Applications
Bioactivity
Loading...
Product Specifications
Source
E. coli-derived human IL-2 protein
Ala21-Thr153, with an N-terminal Met
Produced using non-animal reagents in an animal-free laboratory.
Manufactured and tested under cGMP guidelines.
Ala21-Thr153, with an N-terminal Met
Produced using non-animal reagents in an animal-free laboratory.
Manufactured and tested under cGMP guidelines.
Purity
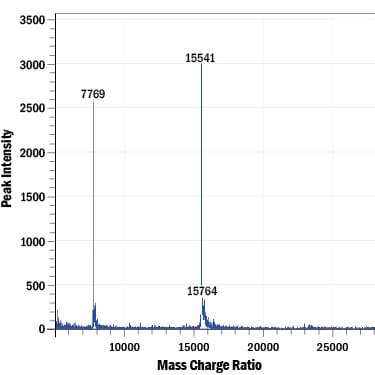
>97%, by SDS-PAGE with silver staining, under reducing conditions. Intact mass analysis of recombinant human IL-2 confirms the predicted molecular mass of 15549 Da.
Endotoxin Level
<0.01 EU per 1 μg of the protein by the LAL method.
N-terminal Sequence Analysis
Met-Ala21-Pro-Thr-Ser-Ser-Ser-Thr-Lys-Lys
Predicted Molecular Mass
15.5 kDa
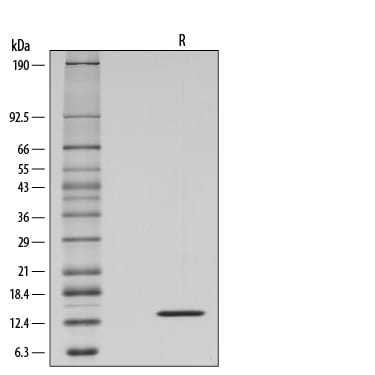
SDS-PAGE
13 kDa, reducing conditions
Activity
Measured in a cell proliferation assay using CTLL‑2 mouse cytotoxic T cells. Gearing, A.J.H. and C.B. Bird (1987) in Lymphokines and Interferons, A Practical Approach. Clemens, M.J. et al. (eds): IRL Press. 295.
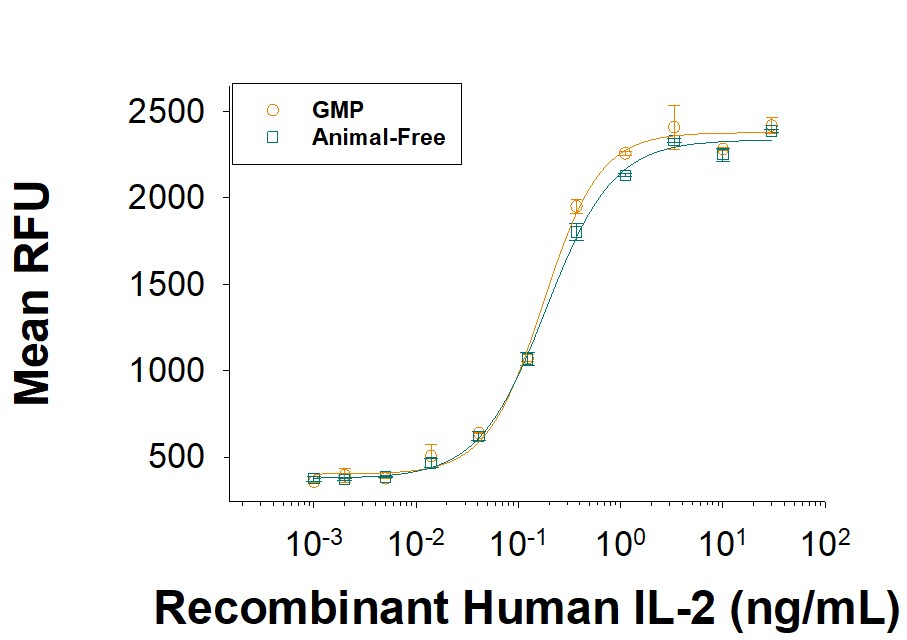
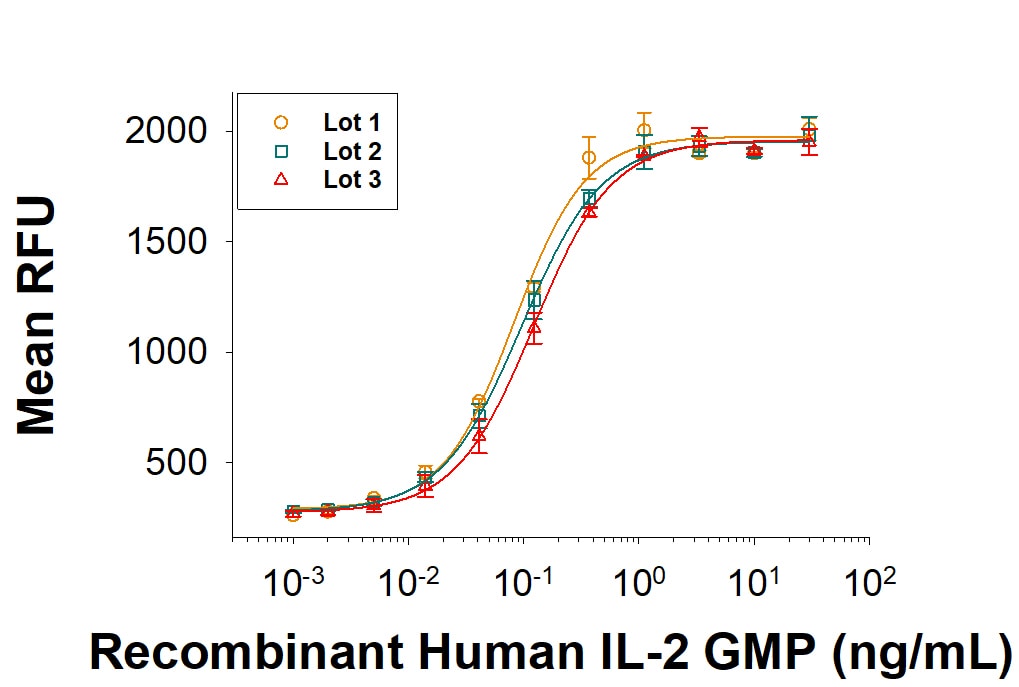
The ED50 for this effect is 0.05-0.25 ng/mL. The specific activity of recombinant human IL-2 is >5.00 x 106 IU/mg, which is calibrated against an internal reference standard value assigned against the human IL-2 WHO International Standard (NIBSC code: 86/500).
The ED50 for this effect is 0.05-0.25 ng/mL. The specific activity of recombinant human IL-2 is >5.00 x 106 IU/mg, which is calibrated against an internal reference standard value assigned against the human IL-2 WHO International Standard (NIBSC code: 86/500).
Host Cell Protein
<0.5 ng per µg of protein when tested by ELISA.
Mycoplasma
Negative for Mycoplasma.
Host Cell DNA
<0.0015 ng per µg of protein when tested by PCR.
Reviewed Applications
Read 1 review rated 5 using 202-GMP in the following applications:
Scientific Data Images for Recombinant Human IL-2 GMP Protein, CF
Recombinant Human IL-2 GMP Protein Bioactivity
GMP-grade Recombinant Human IL-2 (Catalog # 202-GMP) stimulates cell proliferation of the CTLL‑2 mouse cytotoxic T cell line. The ED50 for this effect is 0.05-0.25 ng/mL. Three independent lots were tested for activity and plotted on the same graph to show lot-to-lot consistency of GMP IL-2.Recombinant Human IL-2 GMP Protein SDS-PAGE
1 μg/lane of GMP-grade Recombinant Human IL-2 (Catalog # 202-GMP) was resolved with SDS-PAGE under reducing (R) conditions and visualized by silver staining, showing a single band at 13 kDa.Equivalent Bioactivity of GMP and Animal-Free grades of Recombinant Human IL-2
Equivalent bioactivity of GMP (Catalog # 202-GMP) and Animal-Free (AFL202) grades of Recombinant Human IL-2 as measured in a cell proliferation assay using CTLL 2 mouse cytotoxic T cells (orange and green, respectively).Recombinant Human IL-2 GMP Protein Mass Spectrometry
MALDI-TOF analysis of GMP-grade Recombinant Human IL-2 (Catalog # 202-GMP). The major peak corresponds to the calculated molecular mass, 15549 Da. The minor peak at 15764 Da is a matrix-associated artifact of the MALDI-TOF.Formulation, Preparation, and Storage
202-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in Acetonitrile and TFA. |
| Reconstitution | Reconstitute at 100 μg/mL in 100 mM acetic acid. Alternatively, reconstitute at 100 μg/mL in sterile deionized water and use within 24 hours, store at 2 to 8 °C. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Calculators
Background: IL-2
References
- Ma, A. et al. (2006) Annu. Rev. Immunol. 24:657.
- Gaffen, S.L. and K.D. Liu (2004) Cytokine 28:109.
- Taniguchi, T. et al. (1983) Nature 302:305.
- Mosmann, T.R. et al. (1987) J. Immunol. 138:1813.
- Liparoto, S.F. et al. (2002) Biochemistry 41:2543.
- Wang, X. et al. (2005) Science 310:1159.
- Bodnar, A. et al. (2008) Immunol. Lett. 116:117.
- Jaleco, S. et al. (2003) J. Immunol. 171:61.
- Malek, T.R. (2003) J. Leukoc. Biol. 74:961.
- Laurence, A. et al. (2007) Immunity 26:371.
- Kryczek, I. et al. (2007) J. Immunol. 178:6730.
- Afzali, B. et al. (2007) Clin. Exp. Immunol. 148:32.
- Fehervari, Z. et al. (2006) Trends Immunol. 27:109.
Long Name
Interleukin 2
Alternate Names
Aldesleukin, IL2, Proleukin, TCGF
Entrez Gene IDs
Gene Symbol
IL2
UniProt
Additional IL-2 Products
Product Documents for Recombinant Human IL-2 GMP Protein, CF
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented processes and QA control of documentation and process changes
- Personnel training programs
- Raw material testing and vendor qualification/monitoring
- Fully validated equipment, processes and test methods
- Equipment calibration schedules using a computerized calibration program
- Facility maintenance, safety programs and pest control
- Material review process for variances
- Monitoring of stability over product shelf-life
R&D Systems strives to provide our customers with the analytical characteristics of each product so that customers may determine whether our products are appropriate for their research. The Certificate of Analysis provided contains the following lot specific information:
- N-terminal amino acid analysis, SDS-PAGE analysis, and endotoxin level (as determined by LAL assay) performed on each bulk QC lot, not on individual bottlings of each QC lot
- Post-bottling lot-specific bioassay results (compliance with an established range) and results of microbial testing according to USP
- Host Cell Protein testing performed by ELISA
- Mycoplasma testing by ribosomal RNA hybridization assay
Additional testing and documentation requested by the customer can be arranged at an additional cost.
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo use. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Animal-Free Manufacturing Conditions
Our dedicated controlled-access animal-free laboratories ensure that at no point in production are the products exposed to potential contamination by animal components or byproducts. Every stage of manufacturing is conducted in compliance with R&D Systems' stringent Standard Operating Procedures (SOPs). Production and purification procedures use equipment and media that are confirmed animal-free.
Production
- All molecular biology procedures use animal-free media and dedicated labware.
- Dedicated fermentors are utilized in committed animal-free areas.
- Protein purification columns are animal-free.
- Bulk proteins are filtered using animal-free filters.
- Purified proteins are stored in animal-free containers in a dedicated cold storage room.
Quality Assurance
- Low Endotoxin Level.
- No impairment of biological activity.
- High quality product obtained under stringent conditions.
Product Specific Notices for Recombinant Human IL-2 GMP Protein, CF
Full terms and conditions of sale can be found online in the Protein Sciences Segment T&Cs at: Terms & Conditions.
For preclinical, or clinical ex vivo use
Related Research Areas
Citations for Recombinant Human IL-2 GMP Protein, CF
Customer Reviews for Recombinant Human IL-2 GMP Protein, CF (1)
5 out of 5
1 Customer Rating
Have you used Recombinant Human IL-2 GMP Protein, CF?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Stem/Immune cell maintenance or differentiationVerified Customer | Posted 10/07/2020
There are no reviews that match your criteria.
Loading...
Associated Pathways
 Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
Hematopoietic Stem Cell Differentiation Pathways & Lineage-specific Markers
 IL-2 Signaling Pathways
IL-2 Signaling Pathways
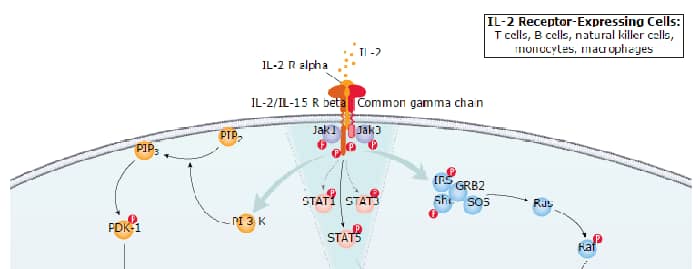 IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-2 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
 IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types
IL-15 Signaling Pathways and their Primary Biological Effects in Different Immune Cell Types

Innate Lymphoid Cell Differentiation Pathways
 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th2 Differentiation Pathway
Th2 Differentiation Pathway

 Jak/STAT Signaling Pathway
Jak/STAT Signaling Pathway
 Th1 Differentiation Pathway
Th1 Differentiation Pathway
 Th2 Differentiation Pathway
Th2 Differentiation Pathway