Dendritic Cell Lineage Development Pathways
Cell (HSC)
Progenitor Cell (CMP)
Progenitor (GMP)
Progenitor (MDP)
Progenitor (CDP)
Progenitor (cMoP)
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Dendritic Cell (pDC)
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Erythromeyloid
Progenitor Cell
HSC Progenitor
Progenitor
Cell
Erythromyeloid
Progenitor Cell
Monocyte
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Monocyte
Cell
CD207+ Dermal DC
Dermal DC
Dermal DC
CD207- Dermal DC
Non-Lymphoid
Tissue cDCs
Resident cDCs
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
CD11b+ cDC
CD11b- cDC
Lymphoid Tissue
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Propria/Peyer's Patch
CD11b- cDC
cDC
cDC
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
B220/CD45 R+
Bst-2/PDCA-1+
CD11b/Integrin alpha M-
CD11c+
CLEC9a+
DC-SIGN/CD209+
Flt-3/Flk-2+
Ly-6C+
M-CSF R/CD115+
MHC class II+
Siglec-H+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Draining Lymph Nodes
During Inflammation
CD11b/Integrin alpha M+
CD11c+
CD107b/Mac-3+
F4/80+
Fc gamma RI/CD64+
Fc epsilon RI+
Ly-6C+
MHC class II+
MMR/CD206+
Secreted Molecules
IL-12
NO
TNF-alpha
Inflammatory
Monocyte-
derived DC
CD11b/Integrin alpha M+
CD11c+
CD107b/Mac-3+
F4/80+
Fc gamma RI/CD64+
Fc epsilon RI+
Ly-6C+
MHC class II+
MMR/CD206+
Secreted Molecules
IL-12
NO
TNF-alpha
Progenitor (MLP)
MDP or CDP
Progenitor (cMoP)
Lin-
CD1a-
CD11clow
DLEC/BDCA-2+
HLA-DR+
IL-3 R alpha/CD123+
Neuropilin-1/BDCA-4+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Dendritic Cell
(pDC)
Lin-
CD1a-
CD11clow
DLEC/BDCA-2+
HLA-DR+
IL-3 R alpha/CD123+
Neuropilin-1/BDCA-4+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Monocyte
Lin-
CD1a-
CD11clow
DLEC/BDCA-2+
HLA-DR+
IL-3 R alpha/CD123+
Neuropilin-1/BDCA-4+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Lin-
CD1a-
CD11clow
DLEC/BDCA-2+
HLA-DR+
IL-3 R alpha/CD123+
Neuropilin-1/BDCA-4+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Lin-
CD1a-
CD1c/BDCA-1-
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+
CLEC9a+
CX3CR1-
DEC-205/CD205high
HLA-DR+
IGSF4a/SynCAM1/Necl2+
SIRP alpha/CD172a-
XCR1+
Secreted Molecules
IL-12
cDC
Lin-
CD1a-
CD1c/BDCA-1-
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+
CLEC9a+
CX3CR1-
DEC-205/CD205high
HLA-DR+
IGSF4a/SynCAM1/Necl2+
SIRP alpha/CD172a-
XCR1+
Secreted Molecules
IL-12
Lin-
CD1a-
CD1c/BDCA-1+
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+/-
CX3CR1+
HLA-DR+
SIRP alpha/CD172a+
Secreted Molecules
IL-1 beta
IL-6
IL-10
IL-12
TNF-alpha
cDC
Lin-
CD1a-
CD1c/BDCA-1+
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+/-
CX3CR1+
HLA-DR+
SIRP alpha/CD172a+
Secreted Molecules
IL-1 beta
IL-6
IL-10
IL-12
TNF-alpha
Draining Lymph Nodes
During Inflammation
Lin-
CD1a+
CD1c/BDCA-1+
CD11b/Integrin alpha M+/-
CD11c+
CD14+
Fc gamma RI/CD64+
Fc epsilon RI+
HLA-DR+
MMR/CD206+
SIRP alpha/CD172a+
Secreted Molecules
IL-23
NO
TNF-alpha
Inflammatory
Monocyte-
derived DC
Lin-
CD1a+
CD1c/BDCA-1+
CD11b/Integrin alpha M+/-
CD11c+
CD14+
Fc gamma RI/CD64+
Fc epsilon RI+
HLA-DR+
MMR/CD206+
SIRP alpha/CD172a+
Secreted Molecules
IL-23
NO
TNF-alpha
Dermal DC
Dermal DC
Dermal DC
Lin-
CD1a-
CD11clow
DLEC/BDCA-2+
HLA-DR+
IL-3 R alpha/CD123+
Neuropilin-1/BDCA-4+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Lin-
CD1a-
CD11clow
DLEC/BDCA-2+
HLA-DR+
IL-3 R alpha/CD123+
Neuropilin-1/BDCA-4+
Intracellular Markers
E2-2/Tcf4+
IRF7+
IRF8+
Spi-B+
TLR7+
TLR9+
Secreted Molecules
IFN-alpha
IFN-beta
IL-6
TNF-alpha
Lin-
CD1a-
CD1c/BDCA-1-
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+
CLEC9a+
CX3CR1-
DEC-205/CD205high
HLA-DR+
IGSF4a/SynCAM1/Necl2+
SIRP alpha/CD172a-
XCR1+
Secreted Molecules
IL-12
cDC
Lin-
CD1a-
CD1c/BDCA-1-
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+
CLEC9a+
CX3CR1-
DEC-205/CD205high
HLA-DR+
IGSF4a/SynCAM1/Necl2+
SIRP alpha/CD172a-
XCR1+
Secreted Molecules
IL-12
Lin-
CD1a-
CD1c/BDCA-1+
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+/-
CX3CR1+
HLA-DR+
SIRP alpha/CD172a+
Secreted Molecules
IL-1 beta
IL-6
IL-10
IL-12
TNF-alpha
cDC
Lin-
CD1a-
CD1c/BDCA-1+
CD11b/Integrin alpha Mlow
CD11c+
CD141/BDCA-3+/-
CX3CR1+
HLA-DR+
SIRP alpha/CD172a+
Secreted Molecules
IL-1 beta
IL-6
IL-10
IL-12
TNF-alpha
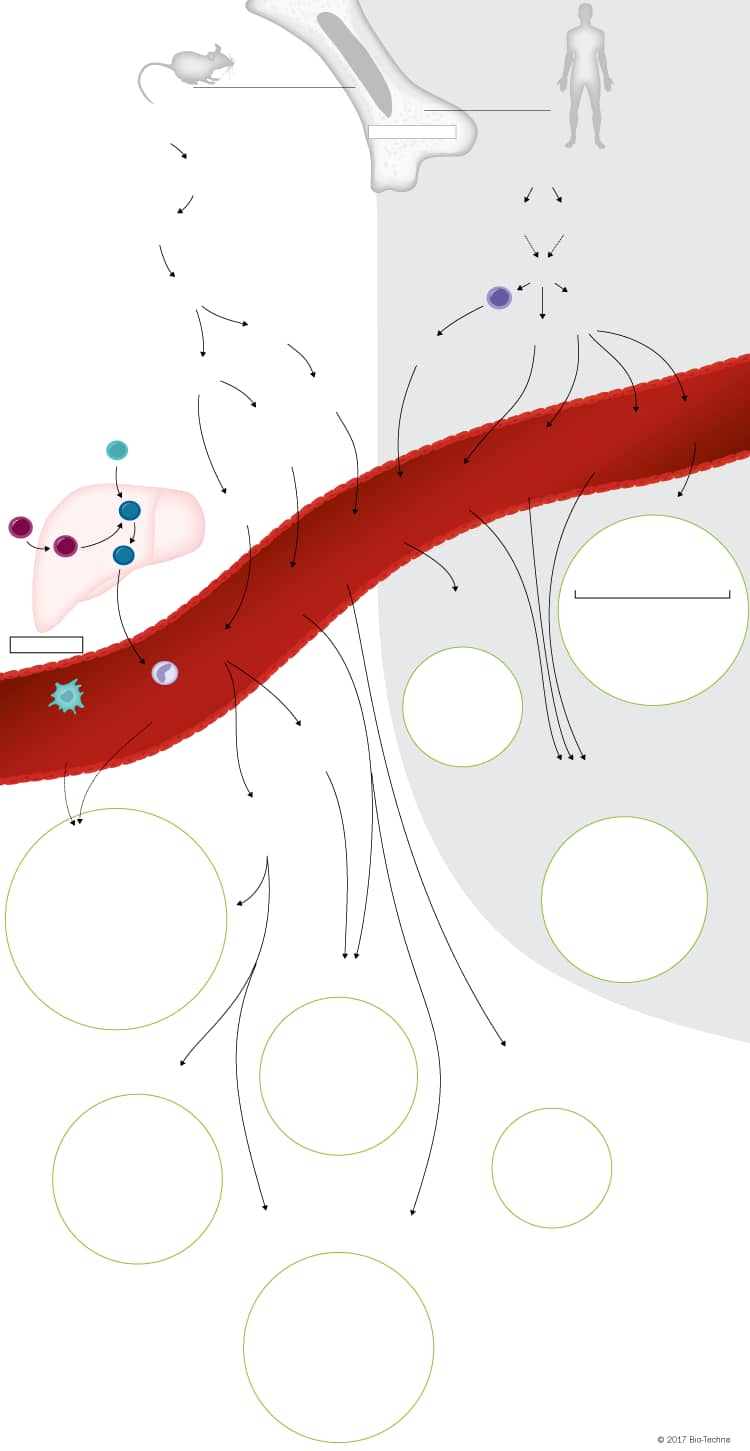
Overview of Dendritic Cell Lineage Development
Dendritic cells (DCs) are key mediators of the innate and adaptive immune responses due to their abilities to: 1) upregulate MHC molecules and costimulatory receptors upon pathogen recognition, 2) capture, process, and present antigens to naïve T cells, and 3) produce polarizing cytokines that promote pathogen-specific effector T cell differentiation and activation. In addition, DCs can promote self-tolerance by secreting tolerogenic cytokines that induce the differentiation of regulatory T cells.
With the exception of Langerhans cells which develop from precursor cells in the yolk sac and fetal liver, dendritic cells in mice develop from macrophage-dendritic cell precursors (MDPs) in the bone marrow. MDPs give rise to common monocyte progenitors (cMoPs) and common DC progenitors (CDPs). cMoPs develop into blood monocytes, which migrate to inflamed tissues, where they differentiate into monocyte-derived dendritic cells (MoDCs). In contrast, CDPs give rise to plasmacytoid dendritic cells (pDCs) and pre-DCs (cDC progenitors). pDCs fully mature in the bone marrow prior to entering the bloodstream, while pre-DCs migrate through the blood to lymphoid and non-lymphoid tissues, where they then differentiate into classical DC (cDC) subsets. Dendritic cell development in humans is not as clearly established but both granulocyte-macrophage progenitors (GMPs) and multi-lymphoid progenitors (MLPs) have been suggested to have the potential to differentiate into a MDP- or CDP-like progenitor. These progenitors are thought to subsequently differentiate into cMoPs, pDCs, and the human equivalent of pre-DCs. Similar to what is seen in mice, human cMoPs develop into blood monocytes, which differentiate into MoDCs in inflamed tissues, and fully mature pDCs along with immature pre-DCs migrate through the blood to tissues. Immature human pre-DCs differentiate either in the bloodstream or in tissues following migration.
Several mouse and human cDC subsets have been characterized based on the differential expression of specific cell surface and intracellular markers on DCs isolated from different tissues. In mouse lymphoid tissue, two cDC subsets have been characterized based on their expression of either CD8 alpha or CD11b. CD8 alpha+ cDCs are specialized in their ability to cross-present antigens on MHC class I molecules to CD8+ T cells and prime Th1 and cytotoxic T cell responses, while CD11b+ cDCs preferentially interact with CD4+ T cells through MHC class II-antigenic peptide complexes. In addition to the lymphoid tissue-resident mouse cDCs subsets, several tissue migratory/non-lymphoid tissue cDC subsets have also been characterized. In mouse skin, five different cDC subsets have been identified including Langerhans cells and four other dermal DC subtypes that are distinguished based on the expression or lack of expression of CD103, CD11b, and CD207. In mouse lung, liver, and kidney, two cDC subsets have been identified that are either CD103+ CD11b- or CD103- CD11b+. Similarly, in mouse intestinal lamina propria and Peyer’s patches, both CD103+ CD11b- and CD103- CD11b+ cDCs have been found, along with a third subset that expresses both CD103 and CD11b.
Significant differences between the markers expressed on mouse and human cDCs have hindered the characterization of cDC subsets in humans. To date, only two subsets of human cDCs have been identified in the blood, spleen, and tonsils based on the expression of either CD141/BDCA-3 or CD1c/BDCA-1. Human CD141/BDCA-3+ cDCs are thought to be phenotypically and functionally similar to mouse CD8 alpha+ cDCs, while human CD1c/BDCA-3+ cDCs correspond to mouse CD11b+ cDCs. In addition to these two human cDC subsets, four other DC subsets have been characterized in the human skin including Langerhans cells, CD1a+ CD1c/BDCA-1+, CD1a+ CD141/BDCA-3+, and CD1a- CD14+ dermal DCs.
Unlike cDCs where multiple subsets have been identified, only one type of pDC has been characterized in both mouse and human. pDCs are relatively rare and are morphologically and functionally distinct from cDCs. They fully mature in the bone marrow before entering the bloodstream, have a spherical morphology, and express low levels of MHC class II prior to activation. Once activated, pDCs secrete large amounts of type I interferons and pro-inflammatory cytokines, and upregulate MHC class II and T cell co-stimulatory molecule expression, allowing them to function as antigen-presenting cells.
To learn more, please visit our Dendritic Cell Subsets and Lineage-specific Markers research topic page.



