Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
47 results for "COMT" in Products
COMT Products
COMT (Catechol-O-MethylTransferase) is a 29-30 kDa member of the COMT family of enzymes. It shares the same acronym with plant COMT, or caffeic acid 3-O-methyltransferase that is involved in phenylpropanoid synthesis. Mammalian COMT is widely expressed, and catalyzes the O-methylation of catecholamines, dopamine and estrogen. Human COMT is a 271 amino acid (aa) type II transmembrane (TM) protein. It contains a six aa cytoplasmic N-terminus, an 20 aa TM segment, and a 245 aa ...
| Reactivity: | Human, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | KO, IHC, WB |
Loading...
| Reactivity: | Human, Mouse |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, ELISA, Flow |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | IHC, WB, ELISA |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Source: | E. coli |
| Accession #: | AAA68929 |
| Applications: | Enzyme Activity |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2b Kappa Monoclonal Clone #1G4-1A1 |
| Applications: | WB, ELISA |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human |
| Details: | Rabbit IgG Monoclonal Clone #8E1 |
| Applications: | IHC, WB, ELISA |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human, Rat |
| Details: | Rabbit IgG Monoclonal Clone #9Y2A3 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Sensitivity: | 0.058 ng/mL (example only; lot dependent) |
| Assay Range: | 0.156 - 10 ng/mL (example only; lot dependent) |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Sensitivity: | 0.058 ng/mL (example only; lot dependent) |
| Assay Range: | 0.156 - 10 ng/mL (example only; lot dependent) |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Source: | E. coli |
| Applications: | AC |
Loading...
| Reactivity: | Human |
| Source: | Synthetic |
| Applications: | AC |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Sheep IgG Polyclonal |
| Applications: | WB |
Loading...


![Western Blot: COMT Antibody [NBP2-15954] Western Blot: COMT Antibody [NBP2-15954]](https://resources.rndsystems.com/images/products/COMT-Antibody-Western-Blot-NBP2-15954-img0005.jpg)
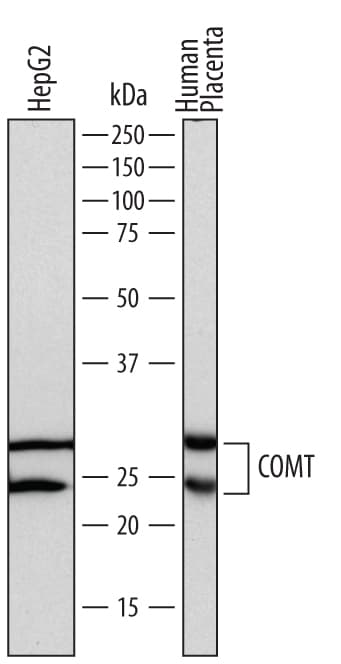
![Western Blot: COMT Antibody [NB100-1780] Western Blot: COMT Antibody [NB100-1780]](https://resources.rndsystems.com/images/products/COMT-Antibody-Western-Blot-NB100-1780-img0002.jpg)
![Western Blot: COMT Antibody [NB100-41387] Western Blot: COMT Antibody [NB100-41387]](https://resources.rndsystems.com/images/products/COMT-Antibody-Western-Blot-NB100-41387-img0007.jpg)


![Western Blot: COMT Antibody (1G4-1A1) [H00001312-M01] Western Blot: COMT Antibody (1G4-1A1) [H00001312-M01]](https://resources.rndsystems.com/images/products/COMT-Antibody-1G4-1A1-Western-Blot-H00001312-M01-img0005.jpg)
![Immunohistochemistry: COMT Antibody (8E1) [NBP3-26646] - COMT Antibody (8E1)](https://resources.rndsystems.com/images/products/nbp3-26646_rabbit-comt-mab-8e1-262202415355741.jpg)
![Western Blot: COMT Antibody - BSA Free [NBP1-85818] COMT Antibody - BSA Free Western Blot: COMT Antibody - BSA Free [NBP1-85818]](https://resources.rndsystems.com/images/products/nbp1-85818_rabbit-polyclonal-comt-antibody-24420259421717.jpg)
![Western Blot: COMT Antibody (9Y2A3) [NBP3-16413] Western Blot: COMT Antibody (9Y2A3) [NBP3-16413]](https://resources.rndsystems.com/images/products/COMT-Antibody-9Y2A3-Western-Blot-NBP3-16413-img0001.jpg)
![Western Blot: COMT Antibody [H00001312-B01P] Western Blot: COMT Antibody [H00001312-B01P]](https://resources.rndsystems.com/images/products/COMT-Antibody-Western-Blot-H00001312-B01P-img0004.jpg)
![ELISA: Human COMT ELISA Kit (Colorimetric) [NBP3-27406] -](https://resources.rndsystems.com/images/products/nbp3-27406_human-comt-elisa-kit-colorimetric-114202410262137.png)
![ELISA: Human COMT - Ready-To-Use ELISA Kit (Colorimetric) [NBP3-31201] - Human COMT - Ready-To-Use ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp3-31201_human-comt-ready-to-use-elisa-kit-colorimetric-206202415314894.png)
![Product Feature: CoraFluor Probes for TR-FRET COMT Antibody [CoraFluor™ 1]](https://resources.rndsystems.com/images/products/af7386cl1_sheep-comt-pab-corafluor-1-1292025124120.png)