ADMP: A Ventralizing BMP in the Dorsal Embryo
More than a century ago, pioneering experiments by Hans Spemann resulted in some of the most fascinating, early observations in developmental embryology. Using his infant daughter’s hair as a ligature, he studied the effect of constriction along different planes of the early salamander embryo.1,2 If a ligature was made bisecting the blastopore along the median plane of the embryo, both segments were capable of forming a complete body plan. In contrast, if the dorsal and ventral halves were separated, the presumptive dorsal half self-regulated, forming a complete embryo, while the ventral portion formed a baüchstuck, or “belly-piece,” that lacked axial structures (Figure 1). These observations were a prelude to the classic experiments showing that the dorsal lip of the blastopore (Spemann’s organizer) contained factors that had the ability to induce a secondary body axis.3
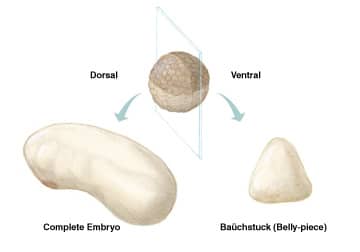
Figure 1. When a Xenopus embryo is bisected into dorsal and ventral halves, the ventral half forms a mass of tissue without axial structures, while the dorsal half self-regulates, forming a proportionally normal embryo. New studies suggest that dorsally expressed ADMP compensates for the removal of ventralizing BMPs.
Only recently have the molecules responsible for the inductive potency of Spemann’s organizer been identified. Most are not inducers, but inhibitors of BMPs (bone morphogenetic proteins).4 Despite the recognition of these molecules, questions remained regarding some of Spemann’s fundamental observations. For instance, what factors in the presumptive dorsal embryo were responsible for embryonic self-regulation? A recent study by Reversade & De Robertis provides evidence that TGF-β superfamily member, anti-dorsalizing morphogenetic protein (ADMP), is critical for this process.5
ADMP is structurally related to BMPs and is expressed in the dorsal organizing center (Spemann’s organizer).6 It has been shown to bind the BMP modulators twisted gastrulation, follistatin, and chordin, as well as the receptor activin RIA/ALK-2, and orthologs have thus far been identified in Xenopus, zebrafish, and chicken.5-9 ADMP is shown to exhibit ventralizing activity despite its paradoxical localization in the dorsal center.6,7 Therefore, it was postulated that it might be the factor conferring self-regulatory capacity in the dorsal embryo.5 Reminiscent of Spemann’s ligature studies, knockdown of ADMP blocks the ability of the dorsal embryo to self-regulate, and the majority of the ectoderm adopts a neural fate.5 In whole embryos, quadruple knockdown of ADMP and ventral BMP-2/4/7 causes all ectoderm to adopt a neural fate at the expense of epidermal tissues. Remarkably, DV patterning could be rescued in these embryos by transplantation of wild-type tissues containing either a dorsal or ventral morphogenetic center. These experiments suggest that molecules in either the dorsal (ADMP) or ventral (BMP-2/4/7) centers are capable of supporting the development of ventral structures.5
What is happening at the molecular level? The authors provide evidence to suggest that BMPs and ADMP are under opposite transcriptional regulation, and that their interactions with BMP regulators establish self-regulating morphogenetic fields (Figure 2).5 Recombinant BMP-4 injection into the blastocoel cavity enhances both the transcription of ventrally expressed BMPs and BMP inhibitors (BAMBI, Sizzled), forming what may be a self-regulating negative feedback loop. In contrast, dorsal transcription of ADMP and chordin decrease in response to BMP-4. In addition, when BMP levels are suppressed, as would presumably be the case in Spemann’s self-regulating dorsal embryo, ADMP transcription increases. These experiments provide a molecular explanation for Spemann’s ligature studies and support the notion that ADMP is critical for embryonic self-regulation.

Figure 2. BMPs/ADMP at opposite poles of the embryo are under opposite transcriptional regulation. Ventrally, BMPS induce themselves and their inhibitors, forming a self-regulating negative feedback loop. In contrast, BMPs suppress ADMP and chordin transcription, and ADMP and chordin reciprocally antagonize each other. Ventralizing molecules shown in green, dorsalizing molecules in red. Adapted from Kimelman, D. & U. J. Pyati (2005) Cell 123:982.
References
- Spemann, H. (1903) Arch. f. Entw. mech. 16:551.
- Sander, K. & P.E. Faessler (2001) Int. J. Dev. Biol. 45:1.
- Spemann, H. & H. Mangold (1924) Reprinted in (2001) Int. J. Dev. Biol.45:13.
- De Robertis, E.M. & H. Kuroda (2004) Annu. Rev. Cell Dev. Biol. 20:285.
- Reversade, B. & E.M. De Robertis (2005) Cell 123:1147.
- Moos, M. Jr. et al. (1995) Development. 121:4293.
- Dosch, R. & C. Niehrs (2000) Mech. Dev. 90:195.
- Dickmeis, T. et al. (2001) Dev. Genes Evol. 211:568.
- Joubin, K. & C.D. Stern (1999) Cell 98:559.
- Oelgeschläger, M. et al. (2000) Nature 405:757.
- Oelgeschläger, M. et al. (2003) Development 130:4047.
