Having trouble trying to find the right product? Chat with our scientists below - we're here to help!
286 results for "Apolipoprotein E/ApoE" in Products
Apolipoprotein E/ApoE Products
The apolipoproteins are a structurally-unrelated group of proteins that have some association with the transport of lipids in blood. Apolipoproteins, plus phospholipids, cholesterol and triglycerides, form spherical particles with a lipid/hydrophobic center and a (apolipo)protein coat. The apolipoprotein coat promotes aqueous solubility and serves as a ligand for lipoprotein receptors. HDL may contain apolipoproteins A, C, D, E, J, L and M, while LDL contains apolipoproteins B and E.
ApoAI ...
| Reactivity: | Human, Mouse, Rat (Negative) |
| Details: | Mouse IgG1 kappa Monoclonal Clone #WUE-4 |
| Applications: | IHC, WB, ELISA, Flow, ICC/IF, + 2 More |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, ICC/IF, Simple Western, + 1 More |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB, Simple Western |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #1H4 |
| Applications: | IHC, WB, ELISA, Flow |
Loading...
| Reactivity: | Human |
| Sensitivity: | 4.3 ng/mL |
| Assay Range: | 0.016 to 1 ug/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ICC/IF |
Loading...
Recombinant Monoclonal Antibody
| Reactivity: | Human, Mouse |
| Details: | Rabbit IgG Monoclonal Clone #SC0536 |
| Applications: | IHC, WB, ICC/IF, IP |
Loading...
| Reactivity: | Human |
| Details: | Rat IgG2A Monoclonal Clone #395004 |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #960319 |
| Applications: | IHC, ICC/IF, WB (-) |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #GT27711 |
| Applications: | ELISA |
Loading...
| Reactivity: | Human, Mouse, Rat |
| Details: | Rabbit IgG Polyclonal |
| Applications: | IHC, WB, ELISA, ICC/IF |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG1 Monoclonal Clone #960318 |
| Applications: | IHC |
Loading...
| Reactivity: | Human |
| Source: | HEK293 |
| Accession #: | P02649.1 |
| Applications: | Bioactivity |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #377108 |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2A Monoclonal Clone #377109 |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Mouse IgG2B Monoclonal Clone #GT1627 |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Rabbit IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Rat |
| Sensitivity: | 0.38 ng/mL |
| Assay Range: | 0.63-40 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Rabbit |
| Sensitivity: | 0.38 ng/mL |
| Assay Range: | 0.63 - 40 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Mouse |
| Sensitivity: | 0.19 ng/mL |
| Assay Range: | 0.31-20 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Sensitivity: | 14.06 ng/mL |
| Assay Range: | 23.44 - 1500 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Mouse |
| Sensitivity: | 0.94 ng/mL |
| Assay Range: | 1.56 - 100 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Rat |
| Sensitivity: | 1.88 ng/mL |
| Assay Range: | 3.13 - 200 ng/mL |
| Applications: | ELISA |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...
| Reactivity: | Human |
| Details: | Goat IgG Polyclonal |
| Applications: | WB |
Loading...


![Immunohistochemistry-Paraffin: Apolipoprotein E/ApoE Antibody (WUE-4) - BSA Free [NB110-60531] Immunohistochemistry-Paraffin: Apolipoprotein E/ApoE Antibody (WUE-4) - BSA Free [NB110-60531]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-WUE-4-Immunohistochemistry-Paraffin-NB110-60531-img0011.jpg)
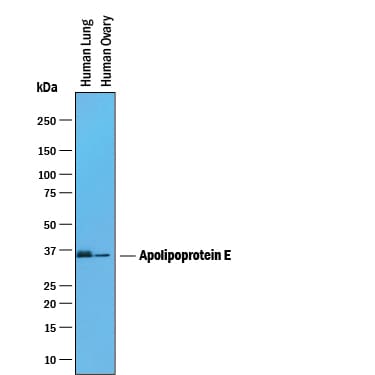
![Western Blot: Apolipoprotein E/ApoE AntibodyBSA Free [NBP1-31123] Western Blot: Apolipoprotein E/ApoE AntibodyBSA Free [NBP1-31123]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-Western-Blot-NBP1-31123-img0032.jpg)


![Western Blot: Apolipoprotein E/ApoE Antibody (1H4) [NBP2-22110] Western Blot: Apolipoprotein E/ApoE Antibody (1H4) [NBP2-22110]](https://resources.rndsystems.com/images/products/ApoE-Antibody-1H4-Western-Blot-NBP2-22110-img0002.jpg)
![ELISA: Human Apolipoprotein E/ApoE ELISA Kit (Colorimetric) [KA1031] ELISA: Human Apolipoprotein E/ApoE ELISA Kit (Colorimetric) [KA1031]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-ELISA-Kit-[HRP]-ELISA-KA1031-img0001.jpg)
![Immunohistochemistry-Paraffin: Apolipoprotein E/ApoE Antibody [NBP2-49450] Immunohistochemistry-Paraffin: Apolipoprotein E/ApoE Antibody [NBP2-49450]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-Immunohistochemistry-Paraffin-NBP2-49450-img0011.jpg)
![Immunohistochemistry-Paraffin: Apolipoprotein E/ApoE Antibody (SC0536) [NBP2-67565] Immunohistochemistry-Paraffin: Apolipoprotein E/ApoE Antibody (SC0536) [NBP2-67565]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-SC0536-Immunohistochemistry-Paraffin-NBP2-67565-img0001.jpg)
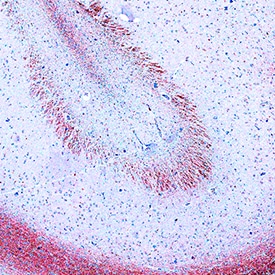
![Immunocytochemistry: Apolipoprotein E/ApoE Antibody (960319) - Azide and BSA Free [MAB41442] Immunocytochemistry: Apolipoprotein E/ApoE Antibody (960319) - Azide and BSA Free [MAB41442]](https://resources.rndsystems.com/images/products/Apolipoprotein-E3-ApoE3-Antibody-960319-Immunocytochemistry-MAB41442-img0003.jpg)
![ELISA: Apolipoprotein E/ApoE Antibody (GT27711) [NBP3-13692] ELISA: Apolipoprotein E/ApoE Antibody (GT27711) [NBP3-13692]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-GT27711-ELISA-NBP3-13692-img0001.jpg)
![Western Blot: Apolipoprotein E/ApoE AntibodyBSA Free [NBP2-41216] Western Blot: Apolipoprotein E/ApoE AntibodyBSA Free [NBP2-41216]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-Western-Blot-NBP2-41216-img0001.jpg)
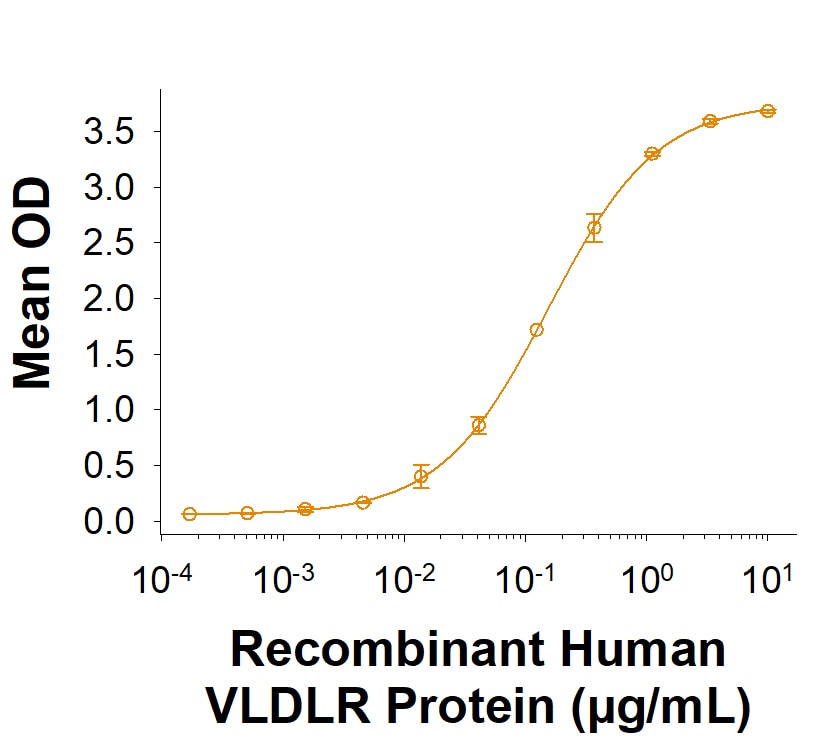
![ELISA: Apolipoprotein E/ApoE Antibody (GT1627) [NBP3-13693] ELISA: Apolipoprotein E/ApoE Antibody (GT1627) [NBP3-13693]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody-GT1627-ELISA-NBP3-13693-img0001.jpg)
![Western Blot: Apolipoprotein E/ApoE AntibodyR136S Mutant [NBP3-13452] Western Blot: Apolipoprotein E/ApoE AntibodyR136S Mutant [NBP3-13452]](https://resources.rndsystems.com/images/products/Apolipoprotein-E-ApoE-Antibody---R136S-Mutant-Western-Blot-NBP3-13452-img0001.jpg)
![ELISA: Rat Apolipoprotein E/ApoE ELISA Kit (Chemiluminescence) [NBP2-66737] - Rat Apolipoprotein E/ApoE ELISA Kit (Chemiluminescence)](https://resources.rndsystems.com/images/products/nbp2-66737_rat-apolipoprotein-e-apoe-elisa-kit-chemiluminescence-132202416225223.jpg)
![ELISA: Rabbit Apolipoprotein E/ApoE ELISA Kit (Colorimetric) [NBP2-66738] - Rabbit Apolipoprotein E/ApoE ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-66738_rabbit-apolipoprotein-e-apoe-elisa-kit-colorimetric-132202416225212.jpg)
![ELISA: Mouse Apolipoprotein E/ApoE ELISA Kit (Chemiluminescence) [NBP2-66739] - Mouse Apolipoprotein E/ApoE ELISA Kit (Chemiluminescence)](https://resources.rndsystems.com/images/products/nbp2-66739_mouse-apolipoprotein-e-apoe-elisa-kit-chemiluminescence-132202416102275.jpg)
![ELISA: Human Apolipoprotein E/ApoE ELISA Kit (Colorimetric) [NBP2-82117] - Human Apolipoprotein E/ApoE ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-82117_human-apolipoprotein-e-apoe-elisa-kit-colorimetric-132202416184813.jpg)
![ELISA: Mouse Apolipoprotein E/ApoE ELISA Kit (Colorimetric) [NBP2-82118] - Mouse Apolipoprotein E/ApoE ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-82118_mouse-apolipoprotein-e-apoe-elisa-kit-colorimetric-132202416201928.jpg)
![ELISA: Rat Apolipoprotein E/ApoE ELISA Kit (Colorimetric) [NBP2-82119] - Rat Apolipoprotein E/ApoE ELISA Kit (Colorimetric)](https://resources.rndsystems.com/images/products/nbp2-82119_rat-apolipoprotein-e-apoe-elisa-kit-colorimetric-132202416254098.jpg)