Apoptosis: Caspase Pathways
A group of intracellular proteases called caspases are responsible for the deliberate disassembly of the cell into apoptotic bodies during apoptosis.1 Caspases are present as inactive pro-enzymes that are activated by proteolytic cleavage. Caspases 8, 9 and 3 are situated at pivotal junctions in apoptosis pathways. Caspase 8 initiates disassembly in response to extracellular apoptosis-inducing ligands and is activated in a complex associated with the cytoplasmic death domain of many cell surface receptors for the ligands.2 Caspase 9 activates disassembly in response to agents or insults that trigger the release of cytochrome c from mitochondria3, 4 and is activated when complexed with apoptotic protease activating factor 1 (APAF-1) and extra-mitochondrial cytochrome c.5 Caspase 3 appears to amplify caspase 8 and caspase 9 initiation signals into full-fledged commitment to disassembly.1, 6 Caspase 8 and caspase 9 activate caspase 3 by proteolytic cleavage and caspase 3 then cleaves vital cellular proteins or other caspases.1, 6
Work with cells from caspase 3-/- and from caspase 9-/- mice suggests that the caspase pathway used for disassembly is cell-type specific.7-10 Embryonic stem cells (ESC), embryonic fibroblasts (EF), thymocytes, and splenocytes from caspase 3-/- and from caspase 9-/- mice were subjected to a wide array of agents or insults that induce apoptosis in cells from mice homozygous or heterozygous for the wild-type alleles. Whereas, ESC, EF, and thymocytes from caspase 9-/- mice were resistant to etoposide, splenocytes were sensitive. Caspase 9-/- ESC and EF were resistant to UV irradiation, however, thymocytes and splenocytes were sensitive. Cells from the caspase 3-/- mice showed a cell-type specificity for sensitivity to UV and gamma irradiation.
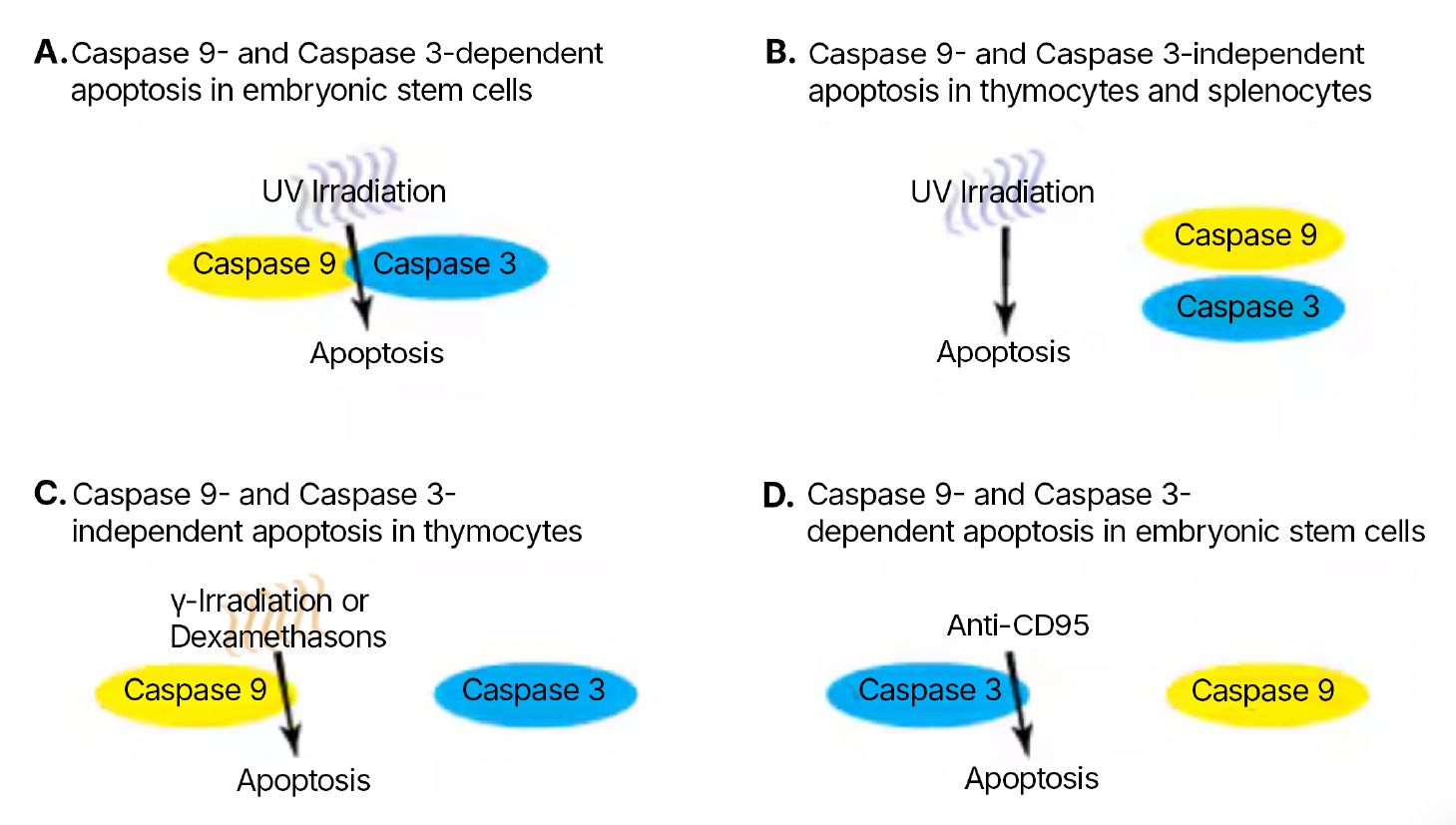
Figure 1. Four caspase pathways involved in apoptosis.
The results (summarized in Table 1 below) suggest four possible caspase pathways. The first (Figure 1A) requires both caspase 3 and caspase 9. ESC from caspase 3-/- and from caspase 9-/- mice are resistant to UV irradiation and many other agents suggesting that this pathway involves cytochrome c release from mitochondria, caspase 9 activation, and subsequent cleavage of caspase 3 by caspase 9. The second pathway (Figure 1B) requires neither caspase 9 nor caspase 3. Thymocytes and splenocytes from caspase 3-/- and from caspase 9-/- mice are sensitive to UV irradiation suggesting the involvement of other caspases. The third (Figure 1C) requires caspase 9 but not caspase 3. Thymocytes from caspase 9-/- mice are resistant to gamma irradiation and dexamethasone, whereas thymocytes from caspase 3-/- mice are sensitive, suggesting that other caspases are directly activated by caspase 9. The fourth (Figure 1D) requires caspase 3 but not caspase 9. Activated splenocytes from caspase 3-/- mice are resistant to anti-CD95 whereas splenocytes from caspase 9-/- mice are sensitive. This pathway likely involves caspase 8 which then activates caspase 3.
| Table 1. | Cell Type-specific Effect of Apoptosis-inducing Agents on Caspase 9 or Caspase 3 Deficient Mice. | |||
|---|---|---|---|---|
| Caspase 9 -/- | Caspase 3 -/- | |||
| resistant to | sensitive to | resistant to | sensitive to | |
| Embryonic Stem Cells | adriamycin cisplatinum anisomycin etoposide osmotic shock γ irradiation UV irradiation | adriamycin cisplatinum anisomycin etoposide osmotic shock γ irradiation UV irradiation | ||
| Embryonic Fibroblasts | adriamycin etoposide γ irradiation UV irradiation |
CTL * killing | adriamycin etoposide γ irradiation UV irradiation |
CTL killing |
| Thymocytes | dexamethasone γ irradiation etoposide |
UV irradiation heat shock anti-CD95 TNF-α | dexamethasone γ irradiation
UV irradiation | |
| Splenocytes | γ irradiation dexamethasone |
UV irradiation adriamycin cisplatinum etoposide osmotic shock | γ irradiation |
UV irradiation |
| Activated Splenocytes | anti-CD95 | anti-CD95 | ||
| * Cytotoxic T-lymphocytes | ||||
Interpretation of the results is complicated by the existence of inhibitors that act by binding to caspases11 or by competing against pro-caspases for binding to the protein complexes that activate caspases.12 Generation of the caspase 9 mutations involved substitution for a segment of caspase 9 required for enzymatic activity. The pro-domain that interacts with CARD (caspase recruitment domain) found in APAF-1 was left intact. Thus, mutant caspase 9s could compete with other caspases for binding to APAF-1.
The effects of caspase 3 and caspase 9 mutations in vivo are striking. Both cause perinatal mortality, and the most obvious effect is on brain development. Brain morphology in caspase 9-/- mice was more aberrant than in caspase 3-/- mice. Caspase 9-/- mice often developed brain tissue outside the skull. This difference suggests that both caspase 9-, caspase 3-dependent (Fig. 1A) and caspase 9-dependent, caspase 3-independent (Fig. 1C) apoptosis pathways are involved in brain development. Altered brain morphology appeared to result, in part, from decreased apoptosis of neuroepithelial progenitors indicating that caspase 9 and caspase 3 are required to cull this population of cells.
Other gross morphological defects were not observed. Tissue remodeling involving retraction of the interdigital webbing in the fetal hand occurred normally in caspase 9-/- mice indicating that apoptosis during this developmental process is caspase 9-independent.8 Although the mutations confer resistance to thymocytes and splenocytes to many inducers of apoptosis, the mutation did not appear to affect thymic selection. Lymph nodes were, however, enlarged in the caspase 9-/- mice that survived 2 weeks.
References
- Thornberry, N.A. and Y. Lazebnik (1998) Science 281:1312.
- Ashkenazi, A. and V.M. Dixit (1998) Science 281:1305.
- Liu, X. et al. (1996) Cell 86:147.
- Green, D.R. and J.C. Reed (1998) Science 281:1309.
- Li, P. et al. (1997) Cell 91:479.
- Cryns, V. and J. Yuan (1998) Genes Dev. 12:1551.
- Hakem, R. et al. (1998) Cell 94:339.
- Kuida, K. et al. (1998) Cell 94:325.
- Kuida, K. et al. (1996) Nature 384:368.
- Woo, M. et al. (1998) Genes Dev. 12:806.
- Deveraux, Q.L. et al. (1997) Nature 388:300.
- Irmler, M. et al. (1998) Nature 388:190.
Related Information
Apoptosis Detection Kits & Reagents
Caspase Activation & Apoptosis
Cysteine Proteases and Regulators
