CD45 in the Stem Cell Niche
The hematopoietic stem cell (HSC) niche is a complex milieu that supports both the survival of quiescent HSCs, and allows for the expansion, differentiation, and migration of precursors during the process of hematopoietic cell replenishment.1, 2 Stromal cells that support the most primitive HSCs in the endosteal niche are actually a mixture of fibroblasts, CXCL12/SDF-1-expressing reticular cells, endothelial cells, and bone-forming osteoblasts.1, 2 Bone-resorbing osteoclasts have recently been suggested to play a role in the niche as well.3 Current evidence indicates that this role involves the hematopoietic transmembrane tyrosine phosphatase, CD45.4
CD45 is present on the surface of all hematopoietic cells, including HSCs and osteoclasts, which are of hematopoietic origin.4, 5 Src family kinases (SFK) are the most well-studied of its substrates. CD45 can affect SFK activity either negatively or positively, depending on which phosphotyrosine it dephosphorylates.5 Deletion mutations within CD45 in humans are associated with severe immunodeficiency. This is primarily due to the absence of CD45 on T cells, where it is typically abundant and required to modulate SFK activity during antigen responses.5 CD45-deficient (CD45-/- ) mouse bone marrow contains normal numbers of hematopoietic cells, but the most primitive HSCs are reduced in number, and their mobilization in response to G-CSF is impaired.4 In part, this defect is intrinsic to the HSC; without CD45-mediated downregulation of SFK activity, integrin-mediated adhesion is high and HSCs are more likely to remain in the niche. CD45-/- HSCs are also deficient in G-CSF-stimulated mobilization and homing to the chemokine CXCL12/SDF-1, which negatively affects cell engraftment following transplantation.4 These deficiencies can be restored by supplementation with SFK inhibitors, indicating that this role is usually performed by CD45.4 Likewise, CD45-/- recipients also show deficient engraftment and subsequent mobilization of normal HSCs, indicating a role for CD45 in the niche, as well as in the HSC.4
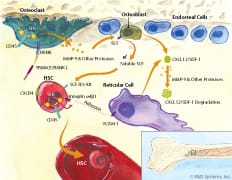
Processes downstream of CD45 are involved in HSC mobilization. CD45 dephosphorylates and downregulates the activity of Src family kinases (SFK) on hematopoietic stem cells (HSCs). This decreases integrin activity and adhesion as occurs, for example, between HSC Integrin alpha 4 beta 1 and reticular cell VCAM-1. CD45 also downregulates osteoclast SFK activity, which allows TRANCE/RANK L to induce the secretion of MMP-9 and other proteases. These proteases catalyze the release of soluble Stem Cell Factor (SCF) from osteoblasts and the degradation of CXCL12/SDF-1, which normally interact with HSC SCF R/c-kit and CXCR4, respectively. Loss of these interactions results in HSC mobilization from the bone marrow into the blood.
While G-CSF has been widely used to induce stem cell mobilization into the peripheral circulation, many other "stress signals" also have a mobilizing effect.6 TRANCE (TNF-related activation-induced cytokine), also known as RANK L (receptor activator of NF-kB ligand) is one such mobilizing agent.3, 4 However, in CD45-/- mice, RANK L is no longer able to mobilize HSCs. Since osteoclasts are the main cell type in the niche that express the receptor for RANK L, this implicates osteoclast CD45 in RANK L-induced mobilization.4 RANK L is better known as a mediator that is required for osteoclast differentiation, and this function also appears to be impaired in the CD45-/- mice. In addition to abnormal morphology and niche remodeling, CD45-/- osteoclasts show impaired G-CSF-stimulated metalloproteinase secretion, and a decrease in both RANK L-stimulated osteopontin degradation and soluble stem cell factor (SCF; c-kit ligand) release.4 These events are known to be regulated by SFK and important for HSC mobilization.3, 7, 8, 9
A role for CD45 in osteoclasts has not previously been shown, although its role in dephosphorylating SFK and other substrates has been demonstrated in many classes of leukocytes.5 Several questions remain concerning CD45, including the reason for its highly regulated expression of isotypes and other differentially glycosylated forms, as well as the identity of its putative extracellular ligand.10 However, the involvement of osteoclast CD45 in endosteal niche architecture and HSC mobilization provides new evidence that the role that osteoclasts play, whether direct or indirect, is important for HSC homeostasis.
References
- Raaijmakers, M.H. & D.T. Scadden (2008) Curr. Opin. Hematol. 15:301.
- Kiel, M.J. & S.J. Morrison (2008) Nat. Rev. Immunol. 8:290.
- Kollet, O. et al. (2006) Nat. Med. 12:657.
- Shivtiel, S. et al. (2008) J. Exp. Med. 205:2381.
- Hermiston, M.L. et al. (2003) Annu. Rev. Immunol. 21:107.
- Spiegel, A. et al. (2008) Cell Stem Cell 3:484.
- Borneo, J. et al. (2007) Exp. Hematol. 35:1026.
- Jin, F. et al. (2008) Bone Marrow Transplant. 42:581.
- Heissig, B. et al. (2002) Cell 109:625.
- Holmes, N. (2005) Immunology 117:145.
This symbol denotes references that cite the use of R&D Systems products.
Related Information
Hematopoietic Stem Cell Markers
Hematopoietic Stem Cell Molecules
