A Look Inside A Tumor: Mechanisms of Tumor Evasion and Immunosuppression in the Tumor Microenvironment Wall Poster
Life Science PostersLife Science Posters Summary
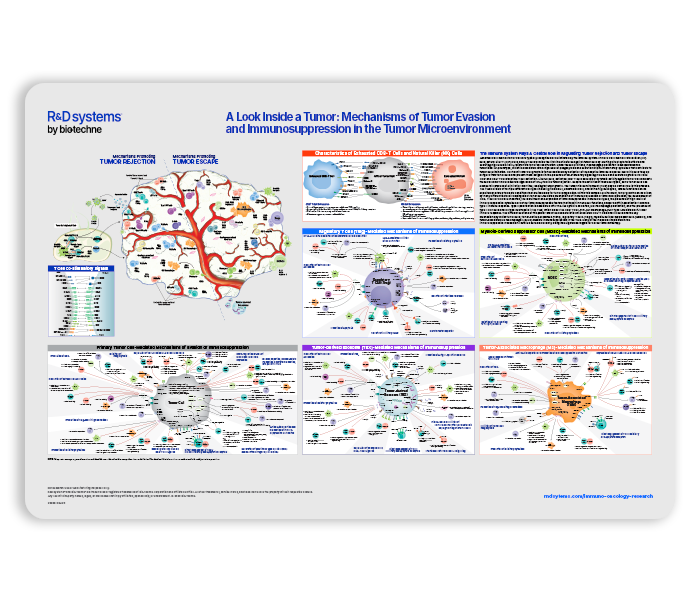
The tumor microenvironment (TME) is an intricate network of diverse cell types that can either inhibit or promote tumor progression. Key components of the TME include stromal cells, endothelial cells, and a heterogeneous array of immune cell populations.
Innate immune cells, such as natural killer (NK) cells, natural killer T (NKT) cells, and gamma delta (γδ) T cells, provide the first line of defense against tumor cells. These cells can mediate direct cytotoxicity against malignant cells without requiring classical antigen sensitization, enabling rapid responses to transformed or stressed cells. Adaptive immune effector cells, including CD8⁺ cytotoxic T lymphocytes and CD4⁺ Th1 helper T cells, are subsequently primed and activated to mount a specialized, secondary immune response that drives targeted tumor destruction.
Despite ongoing immune surveillance, cancer cells frequently acquire changes over time that allow them to escape recognition and elimination by the immune system. Tumor evolution is marked by genetic instability and relentless proliferation, which can drive immunoediting processes that reduce tumor immunogenicity. This decline is further exacerbated by functional exhaustion of NK cells and T cells, and the progressive enrichment of immunosuppressive cell subsets within the TME. Collectively, these alterations converge to suppress effective anti-tumor immunity, enabling cancer cells to evade immune detection and thereby promote tumor progression.
Request our Tumor Microenvironment poster to learn more about:
- Different immune cell types involved in tumor rejection and tumor escape
- Characteristics of exhausted NK and CD8⁺ T cells
- Tumor cell-mediated mechanisms of immune cell evasion and immunosuppression
- Pathways by which regulatory T cells, myeloid-derived suppressor cells, and tumor-associated macrophages promote tumor escape
Request Literature
To access this literature content please fill out the form below.
