B Cell Development
Life Science PostersLife Science Posters Summary
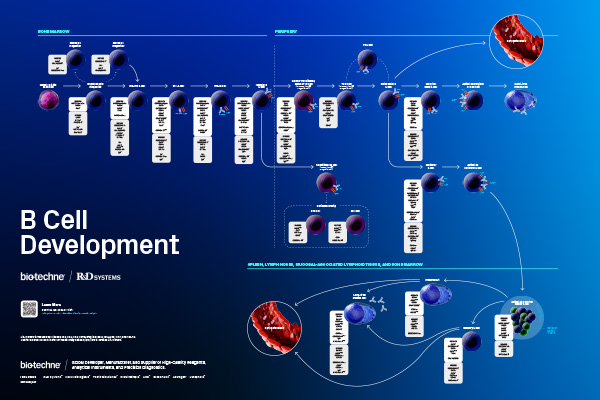
B lymphocytes differentiate from hematopoietic stem cells (HSCs) in the bone marrow in a multi-stage maturation and selection process. Initially, HSCs differentiate into multipotent progenitor cells that can become either common myeloid or common lymphoid progenitor cells. Common lymphoid progenitor cells subsequently develop into B cells, T cells, or natural killer cells. In the B cell lineage pathway, a common lymphoid progenitor cell gives rise to a B lymphocyte progenitor that then differentiates into a Pre-Pro B Cell, a Pro-B Cell, and a Pre-B cell prior to the generation of an immature B cell. These stages of B cell development are antigen-independent. Immunoglobulin gene rearrangement that occurs during these stages of development results in the expression of a mature B cell receptor (BCR) that is capable of binding to antigen. This is followed by a selection process that involves BCR editing or clonal deletion, designed to eliminate self-reactive immature B cells. Immature B cells that survive this selection process migrate from the bone marrow to the spleen, where they differentiate into immunocompetent naïve mature B cells. A small percentage of naïve mature B cells become marginal zone B cells, while the majority of these cells differentiate into follicular B cells. During the antigen-dependent phase of development, marginal zone B cells differentiate into short-lived plasma cells, while follicular B cells differentiate into short-lived plasma cells or take part in germinal center reactions in the spleen, lymph nodes, and mucosal –associated lymphoid tissue, where follicular B cells differentiate into long-lived, antibody-secreting plasma cells or memory B cells. The poster shows the cell surface and intracellular markers that can be used to distinguish the different stages of B cell development.
Request Literature
To access this literature content please fill out the form below.
