Profiling the effects of MEK inhibition on kinase phosphorylation and protease secretion using antibody arrays
by Greta Wegner, Erin Eleria, Wen-Chieh Liao, Dave Finkel, and Amy James.
Scientific Meeting PostersABSTRACT
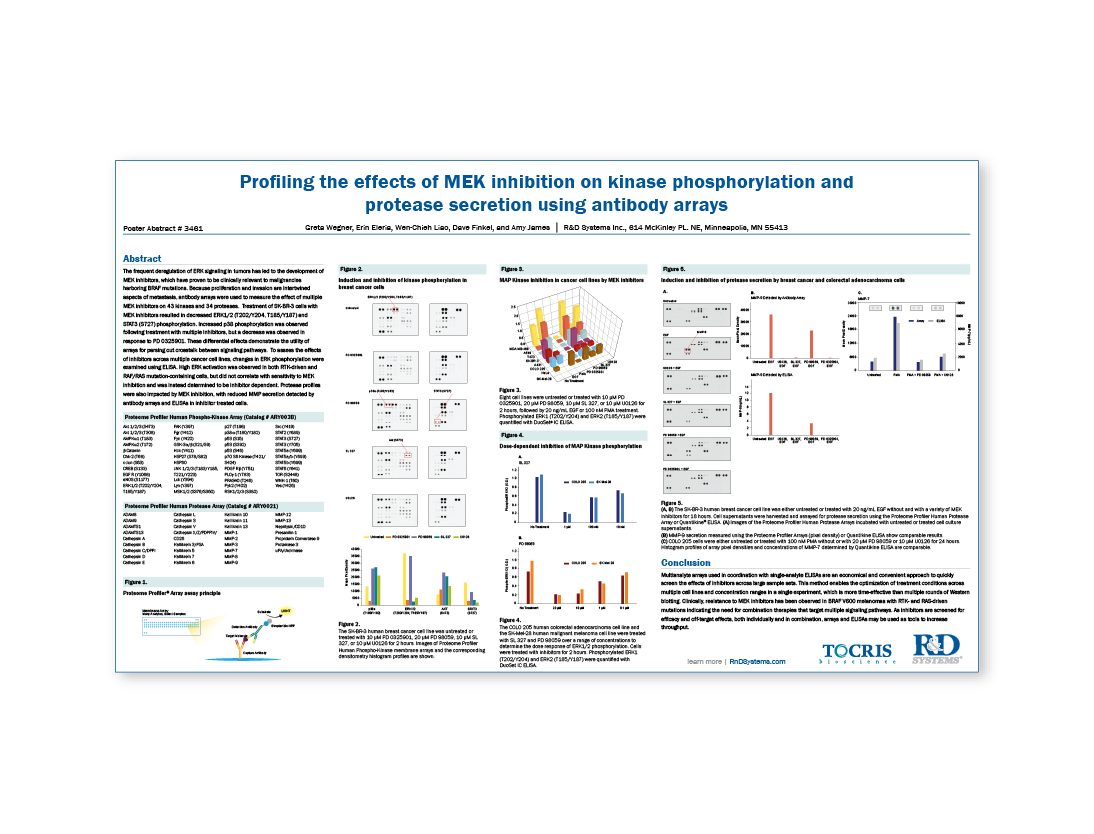
The frequent deregulation of ERK signaling in tumors has led to the development of MEK inhibitors, which have proven to be clinically relevant to malignancies harboring BRAF mutations. Because proliferation and invasion are intertwined aspects of metastasis, antibody arrays were used to measure the effect of multiple MEK inhibitors on 43 kinases and 34 proteases. Treatment of SK-BR-3 cells with MEK inhibitors resulted in decreased ERK1/2 (T202/Y204, T185/Y187) and STAT3 (S727) phosphorylation. Increased p38 phosphorylation was observed following treatment with multiple inhibitors, but a decrease was observed in response to PD 0325901. These differential effects demonstrate the utility of arrays for parsing out crosstalk between signaling pathways. To assess the effects of inhibitors across multiple cancer cell lines, changes in ERK phosphorylation were examined using ELISA. High ERK activation was observed in both RTK-driven and RAF/RAS mutation-containing cells, but did not correlate with sensitivity to MEK inhibition and was instead determined to be inhibitor dependent. Protease profiles were also impacted by MEK inhibition, with reduced MMP secretion detected by antibody arrays and ELISAs in inhibitor treated cells.
Related Information
Proteome Profiler™ Antibody Arrays
Erk Signal Transduction Pathway
Download PDF