Simultaneous Detection of OPRM1 Protein and Its Message in Rat Brain Using Immunohistochemistry and RNAscope In Situ Hybridization Technology
by Rachelle Reed, Jodi Hagen, Nate Hopp, Michael Grahek, Ana Ptak, John Humphrey, Greg Du, Patricia Mur.
Scientific Meeting PostersABSTRACT
The actions of the narcotic analgesics widely used in hospitals, including morphine and fentanyl, are mediated by the µ Opioid Receptor (OPRM1), also known as MOR and MOR1, which is encoded by the OPRM1 gene. Research has shown that multiple OPRM1 splice variants exist, including MOR1A and MOR1B, which are C-terminal splice variants. MOR1A and MOR1B have been shown to be widely distributed in the rat central nervous system; however, expression levels of OPRM1 and MOR1A are higher than MOR1B. Additionally, it has been suggested that the different OPRM1 splice variants may mediate different pharmacological actions of opioids.
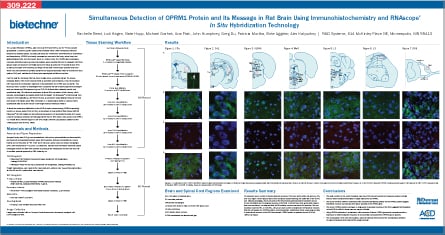
To more closely examine the anatomical distribution of MOR1A expressing neurons among all OPRM1 neurons in rat brain, we combined RNAscope® in situ hybridization (ISH) technology with immunohistochemistry (IHC). We developed a two-color fluorescence detection protocol that identified neurons expressing both the OPRM1 protein and MOR1A mRNA in 15 µm, formaldehyde-fixed, frozen rat brain tissue sections. This protocol utilized a rabbit monoclonal antibody that recognizes and binds the N-terminus of rat OPRM1, and a rat MOR1A RNAscope® probe.
A low number of neurons expressing both the OPRM1 protein and MOR1A mRNA was observed in the rat cortex, hippocampus, and caudate putamen. A higher number of dual-labeled neurons were seen in the ventrolateral periaqueductal gray and medulla, two areas that are part of the descending antinociceptive brainstem circuit. Additionally, we observed that all ISH-positive neurons were always IHC-positive, indicating the rabbit monoclonal antibody that was used is highly specific for rat OPRM1. However, not all neurons expressing the OPRM1 protein stained positive for MOR1A mRNA, suggesting these neurons express other splice variants of the OPRM1 gene.
These findings show that our protocol, which combines ISH and IHC, can be used to study the ratio of neurons expressing different OPRM1 splice variants in different brain regions.
Download Poster