Free Radicals and Oxidative Stress
Until the 1970s, oxygen free radicals were fringe topics in biology. Three key discoveries changed that: i) The report by McCord and Fridovitch1 of the superoxide dismutase (SOD) activity of erythrocuprein and the finding that almost all mammalian cells are equipped with SOD suggested that superoxide is a physiological product; ii) Babior et al.2 showed that the bactericidal action of neutrophils was associated with the generation of superoxide, linking oxygen radicals with inflammation; and iii) Granger et al.3 demonstrated that reperfusion injury after intestinal ischemia was likely caused by oxygen radicals. Many diseases are linked to free radical damage arising from an imbalance between radical-generating and radical-scavenging systems, a condition called oxidative stress.
Sources of Oxygen Radicals
Cells generate energy aerobically by reducing molecular oxygen (O2) to water. The cytochrome c oxidase-catalyzed reaction involves transfer of four electrons (e) to oxygen, in principle without intermediates, but, in fact, partially reduced oxygen species are produced. Other enzymes, especially flavin enzymes, also generate partially reduced oxygen species.
Transfer of one electron to oxygen leads to multiple reactive oxygen species through the superoxide anion radical, O2-• (Figure 1). Superoxide dismutates to hydrogen peroxide (O2-•) + O2-• Further addition of electrons requires cleavage of the bond between the oxygen atoms, a reaction catalyzed by iron (Fenton Reaction) to form the highly reactive hydroxyl radical (OH•). OH• reacts instantaneously with any molecule from which it can abstract a hydrogen atom (Equation 1). (A recently discovered iron-independent pathway to NO2• from Nitric oxide (NO) leads to nitrogen dioxide radicals (NO2•) as well.) Hypochlorous acid (HOCl), a strong oxidant that acts physiologically as a bactericidal agent and pathologically as an inflammogen, is generated enzymatically by myeloperoxidase (MPO) in neutrophils. Reaction of hypochlorous acid with hydrogen peroxide yields singlet oxygen (1O2) + H2O. The biological significance of singlet oxygen is unclear.

Other sources of reactive oxygen species include radiation (e.g., UV light), toxic chemicals (e.g., paraquat), and drugs (e.g., adriamycin, bleomycin).
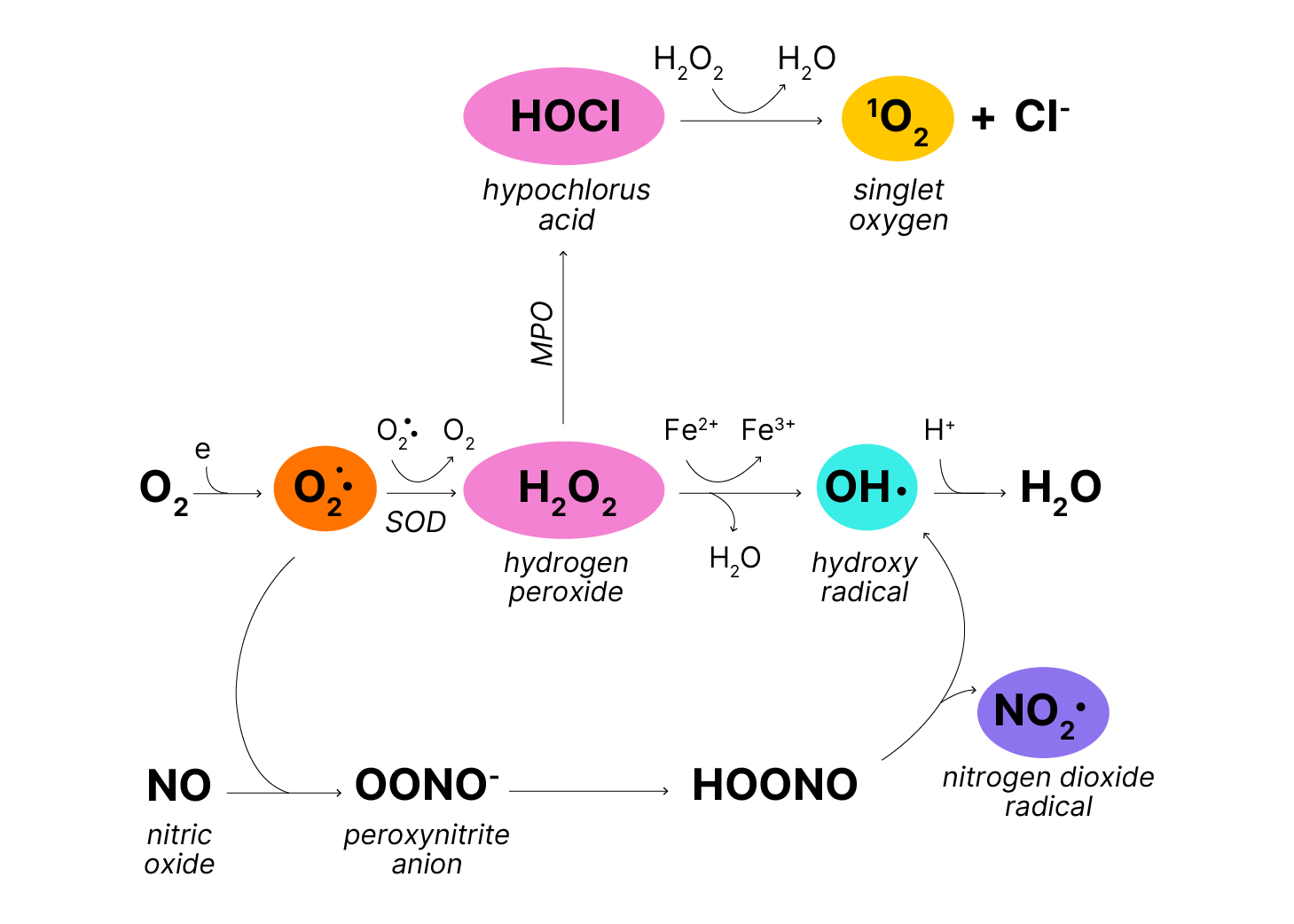
Figure 1. Generation of reactive oxygen species where MPO is myeloperoxidase and SOD is superoxide dismutase.
Mechanism of Damage
Reactive oxygen species, in particular OH•, can react with all biological macromolecules (lipids, proteins, nucleic acids, and carbohydrates). The initial reaction generates a second radical, which can react with a second macromolecule to continue the chain reaction.
Among the more susceptible targets are polyunsaturated fatty acids. Abstraction of a hydrogen atom initiates the process of lipid peroxidation (Figure 2). Numerous products are formed, presenting special analytical problems. The choice is between simple, nonspecific assays for classes of lipid peroxidation products (e.g., thiobarbituric acid reaction for aldehydes), more specific but less sensitive assays (e.g., uv absorption of conjugated dienes) or specific, highly sensitive methods that require expensive instrumentation (e.g., mass spectral analysis of hydroxy fatty acids). A sensitive and specific colorimetric assay based on measurement of malondialdehyde and 4-hydroxyalkenals is a good compromise.
Proteins are modified in structure and function by radical reactions. Metal-catalyzed protein oxidation results in addition of carbonyl groups or cross-linking or fragmentation of proteins. Lipid (peroxidation) aldehydes can react with sulfhydryl (cysteine) or basic amino acids (histidine, lysine). Similarly, modification of individual nucleotide bases, single-strand breaks and cross-linking are the typical effects of reactive oxygen species on nucleic acids.
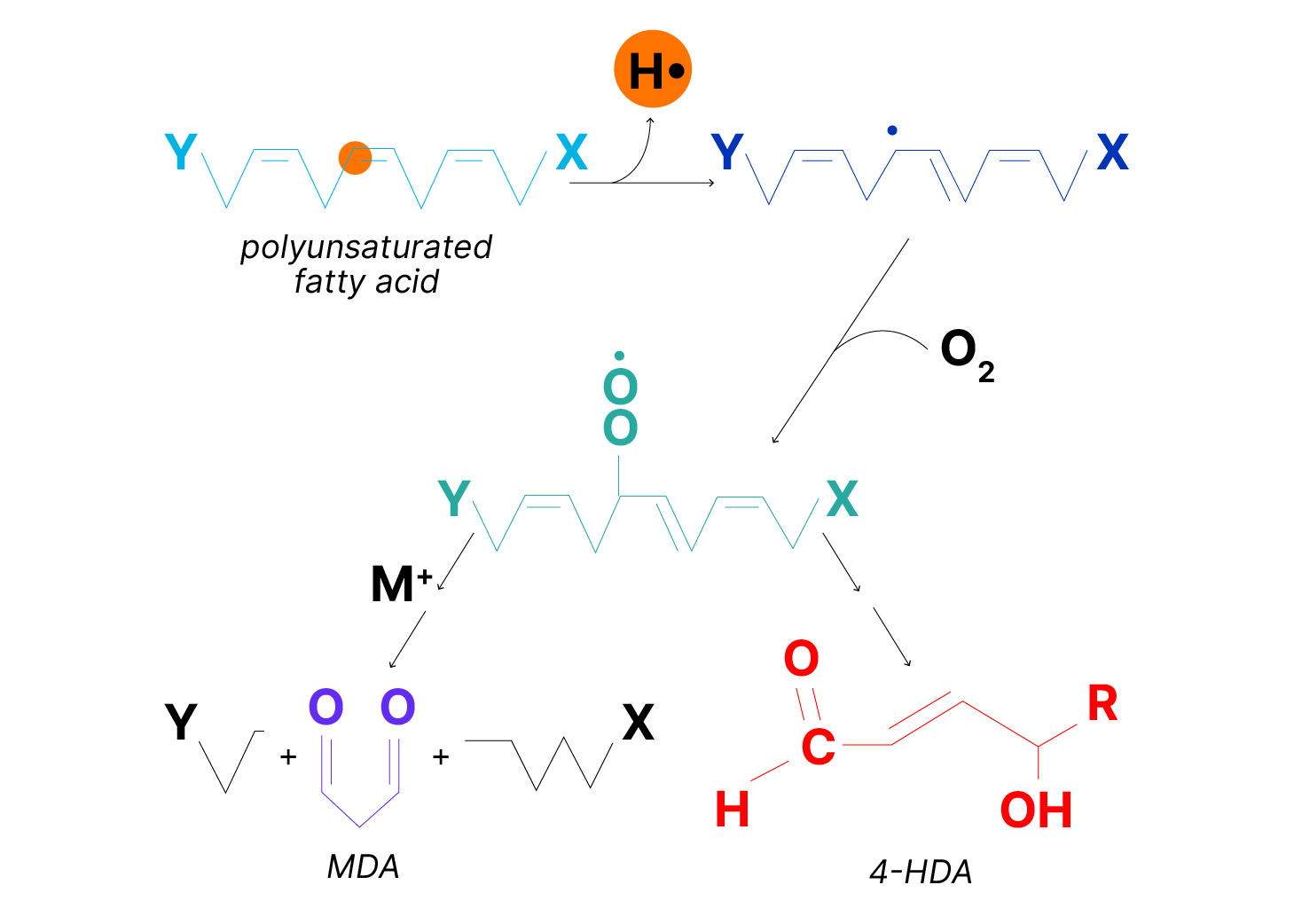
Figure 2. Peroxidation of unsaturated lipids. The variety of lipids and the random nature of free radical reactions leads to many products. These include 4-hydroxyalkenals (4-HDA) and, when there are 3 or more unsaturated bonds, malondialdehyde (MDA). These can serve as targets for the measurement of fatty acid peroxidation. The initiating event can be reaction with another radical, UV light or radiation. Since a radical is also produced in the process, it is a chain reaction.
Defense Mechanisms
Mammalian cells possess elaborate defense mechanisms to detoxify (metabolize) radicals (Figure 3). SOD catalyzes the dismutation of O2-• to H2O2 + O2. H2O2 is converted to O2 + H2O by catalase or by glutathione peroxidase (GPx), which uses glutathione (GSH) as the reducing agent. Redox-active metals, such as iron, bind to storage and transport proteins (e.g., ferritin, transferrin, lactoferrin) to minimize formation, and radical-scavenging antioxidants (e.g., vitamin E) interrupt the chain reactions.
References
- McCord, J.M. and I. Fridovitch (1969) J. Biol. Chem. 244:6049.
- Babior, B.M. et al. (1973) J. Clin. Invest. 52:741.
- Granger, D.N. et al. (1981) Gastroenterology 81:22.
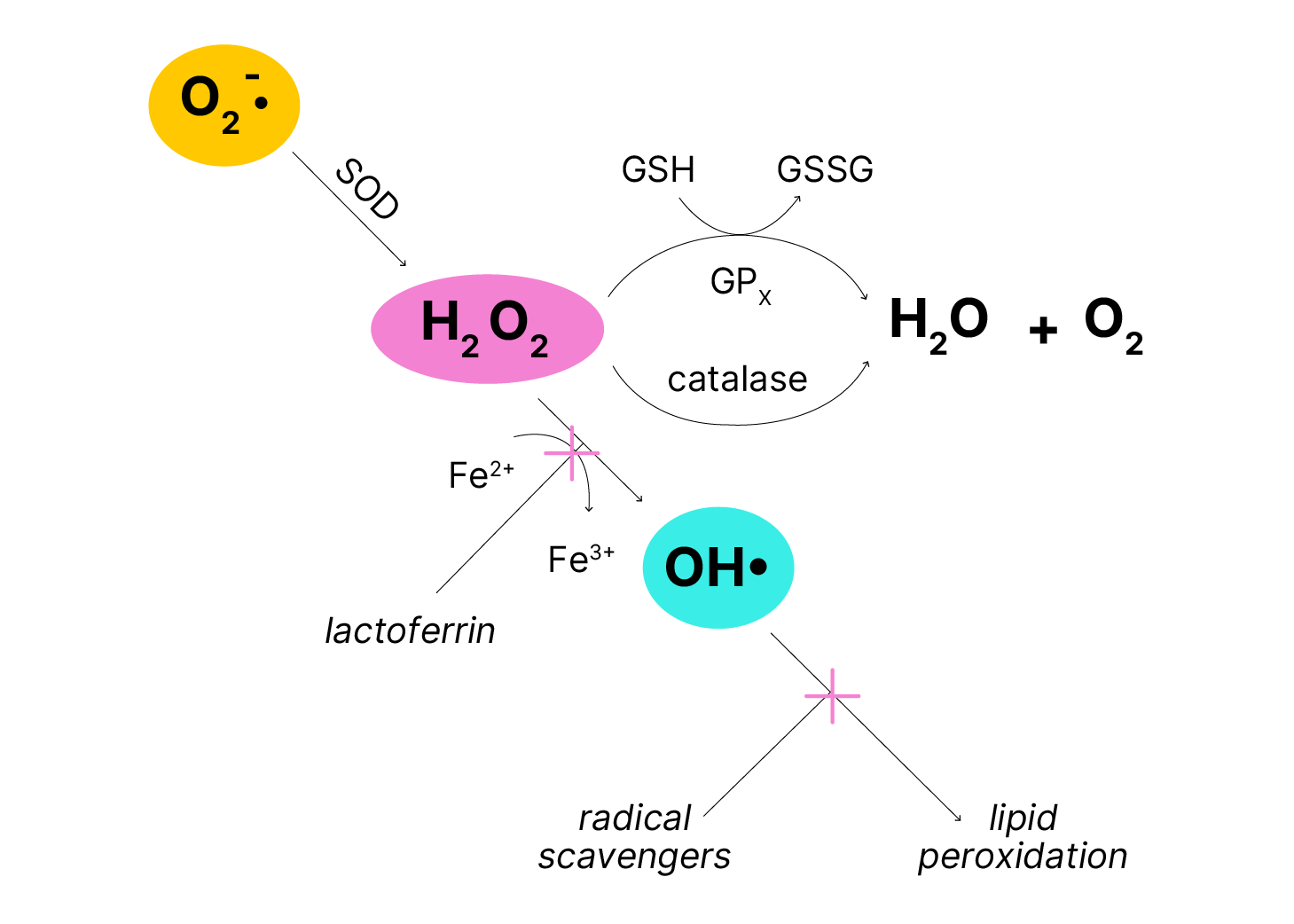
Figure 3. Defense mechanism against damage by reactive oxygen. Superoxide dismutase (SOD) plus catalase or glutathione peroxidase (GPx) eliminate many damaging oxygen species. Lactoferrin (binds iron) and radical scavengers such as vitamin E, further limit damage.
