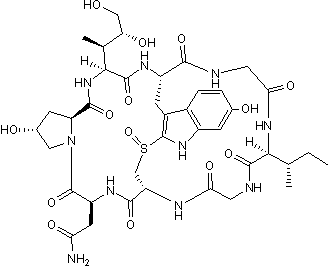
α-Amanitin
Tocris Bioscience | Catalog # 4025

Key Product Details
Description
Product Description
α-Amanitin is an inhibitor of RNA polymerase II. Inhibits transcription in eukaryotic cells. Binds and blocks the largest subunits of RNA polymerase II, preventing new ribonucleotides from incorporating into the nascent RNA chain. Potent amatoxin.
Product Specifications for α-Amanitin
Molecular Weight
Formula
Storage
Purity
Chemical Name
CAS Number
PubChem ID
InChI Key
SMILES
The technical data provided above is for guidance only. For batch specific data refer to the Certificate of Analysis.
Solubility
| Solvent | Max Conc. mg/mL | Max Conc. mM | |
|---|---|---|---|
| Solubility | |||
| water | 4.59 | 5 |
Preparing Stock Solutions for α-Amanitin
The following data is based on the product molecular weight 918.97.
Batch specific molecular weights may vary from batch to batch due to the degree of hydration, which all affect the solvent volumes required to prepare stock solutions.
| Concentration / Solvent Volume / Mass | 1 mg | 5 mg | 10 mg |
|---|---|---|---|
| 0.05 mM | 21.76 mL | 108.82 mL | 217.63 mL |
| 0.25 mM | 4.35 mL | 21.76 mL | 43.53 mL |
| 0.5 mM | 2.18 mL | 10.88 mL | 21.76 mL |
| 2.5 mM | 0.44 mL | 2.18 mL | 4.35 mL |
Calculators
Background References
References are publications that support the biological activity of the product. See our Citations tab to view 7 publications citing the usage of this product.
- Raha Close association of RNA polymerase II and many transcription factors with Pol III genes. Proc.Natl.Acad.Sci.
- Casse The transcriptional inhibitors, actinomycin D and α-amanitin, activate the HIV-1 promoter and favor phosphorylation of the RNA polymerase II C-terminal domain. J.Biol.Chem. 1999 PMID: 10347161
Product Documents for α-Amanitin
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for α-Amanitin
For research use only
Related Research Areas
Citations for α-Amanitin
Customer Reviews for α-Amanitin (1)
Have you used α-Amanitin?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Species: HumanVerified Customer | Posted 02/20/2019alpha -Amanitin was used in antibacterial resistance experiments.We looked into RNAP translocation along the DNA without dissociating from either the template or the growing RNA product until it encounters a termination factor or signal that causes the transcription cycle end with dissociation of the transcribing complex and release of RNA polymerase for a new round of transcription.
There are no reviews that match your criteria.