APE Antibody - BSA Free
Novus Biologicals | Catalog # NB100-101

![Simple Western: APE AntibodyBSA Free [NB100-101] Simple Western: APE AntibodyBSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Simple-Western-NB100-101-img0011.jpg)
Key Product Details
Species Reactivity
Validated:
Human, Mouse, Rat, Primate, Rabbit
Cited:
Human, Mouse, Rat, Rabbit
Applications
Validated:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Immunohistochemistry Free-Floating, Western Blot, Block/Neutralize, Flow Cytometry, Flow (Intracellular), Immunocytochemistry/ Immunofluorescence, Simple Western, Immunoprecipitation, Chromatin Immunoprecipitation (ChIP)
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Western Blot, Immunocytochemistry/ Immunofluorescence, Immunoprecipitation, Chemotaxis, IF/IHC
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Format
BSA Free
Loading...
Product Specifications
Immunogen
Affinity purified human APE1 [UniProt# P27695]
Reactivity Notes
Rabbit reactivity reported in the scientific literature (PMID: 15276530).
Localization
In immunohistochemical experiments punctate nuclear staining is seen in most cell types. However, in hepatocytes, large neurons and granule cells, cytoplasmic staining is observed.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Theoretical MW
37 kDa.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for APE Antibody - BSA Free
Simple Western: APE AntibodyBSA Free [NB100-101]
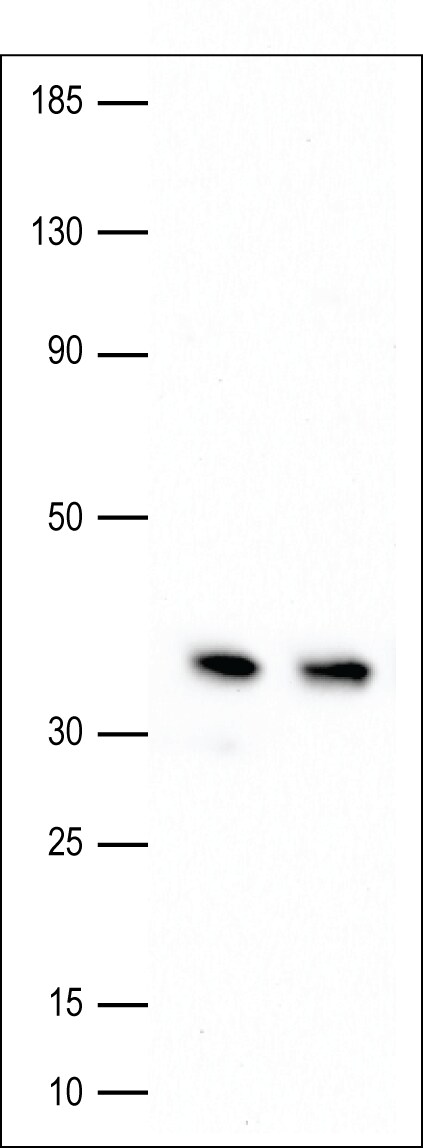
Simple Western: APE Antibody [NB100-101] - Lane view shows a specific band for APE1 in 0.1 mg/ml of HeLa lysate. This experiment was performed under reducing conditions using the 12-230 kDa separation system.Western Blot: APE AntibodyBSA Free [NB100-101]
Western Blot: APE Antibody [NB100-101] - Analysis of APE1 in cell lysates: 1. HeLa, 2. Ntera2, 3. A431, 4. HepG2, 5. MCF7, 6. NIH 3T3, 7. PC12, and 8. Cos 7.Immunohistochemistry: APE Antibody - BSA Free [NB100-101]
Immunohistochemistry: APE Antibody [NB100-101] - Immunohistochemical staining of APE-ref-1 in prostate cancer.Western Blot: APE AntibodyBSA Free [NB100-101]
APE-Antibody-Western-Blot-NB100-101-img0012.jpgImmunocytochemistry/ Immunofluorescence: APE Antibody - BSA Free [NB100-101]
Immunocytochemistry/Immunofluorescence: APE Antibody [NB100-101] - IF on HeLa cells. Image from verified customer review.Western Blot: APE AntibodyBSA Free [NB100-101]
Western Blot: APE Antibody [NB100-101] - SH-SY5Y (nuclear extract) tested at 1:1000 dilution. Image provided by verified customer review.Flow Cytometry: APE Antibody - BSA Free [NB100-101]
Flow Cytometry: APE Antibody [NB100-101] - Spectral properties of Alexa Fluor(R) 488: an excitation maximum of 490 nm and an emission maximum of 525 nm. Use the Novus Spectra Viewer to identify the optimal laser(s) and filters for Alexa Fluor(R) 488 and to determine its compatibility with other fluorophores when designing a multicolor experiment. https://www.novusbio.com/spectraviewerWestern Blot: APE AntibodyBSA Free [NB100-101]
Western Blot: APE Antibody [NB100-101] - Analysis of APE in human melanoma cell lysate using anti-APE antibody. Image from verified customer review.Immunocytochemistry/ Immunofluorescence: APE Antibody - BSA Free [NB100-101]
Immunocytochemistry/Immunofluorescence: APE Antibody [NB100-101] - Detection of APE1 (Green) in HepG2 cells using NB100-101. Nuclei (Blue) are counterstained with Hoechst 33258.Western Blot: APE Antibody - BSA Free [NB100-101] -
Western Blot: APE Antibody - BSA Free [NB100-101] - Defective p53 activity leads to failure of the BER coordination system. (A) High-throughput microscopy analysis of TIG-1 cells transfected with plasmids expressing wild-type or mutant p53. Boxplots showing the distribution of APE1 staining intensity (in arbitrary units) in p53 low versus p53 overexpressing cells. Each p53 mutant is reported on top of the relevant plot. The dashed line highlights the median APE1 intensity in p53 low cells (N > 5000). NS: not statistically significant at P < 0.05. (B) Representative western blotting analysis comparing WI38 & WI38 (SV40) cells upon transfection with the indicated siRNAs. Failure to downregulate Sp1 correlates with the inability to modulate APE1. Actin was used as loading control. (C) qPCR analysis on APE1 transcript in WI38 & WI38 (SV40) cells upon transfection with the indicated siRNAs. APE1 transcription is reduced in WI38 cells only. Note the higher transcript content in WI38 (SV40) cells. (D) Representative Western blotting analysis comparing WI38 & WI38 (SV40) cells upon transfection with the indicated siRNAs. Failure to modulate BER correlates with gamma H2AX staining in WI38 (SV40) cells; gamma H2AX increases further upon XRCC1 depletion. (E) Neutral Comet assay on WI38 & WI38 (SV40) fibroblasts shows accumulation of DSBs upon XRCC1-depletion in transformed cells only. Results are expressed as mean ± SD of three independent experiments. Image collected & cropped by CiteAb from the following publication (https://academic.oup.com/nar/article-lookup/doi/10.1093/nar/gkw015), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: APE Antibody - BSA Free [NB100-101] -
Western Blot: APE Antibody - BSA Free [NB100-101] - APE1 expression in HCC tissues(A) qPCR of tumor (HCC), peri-HCC, & surrounding liver cirrhosis (SLC), & normal liver (CTRL) (left panel). Ratio between HCC & SLC & HCC & peri-HCC within the same patients (right panel). APE1 mRNA quantification was normalized to two reference genes 18srRNA & Actin. Bar graphs indicate mean & SEM. (B) APE1 protein quantification in HCC & SLC tissue lysates from HCC cancer patients. Graphs indicate the different distributions of the fold of protein expression for each sample as the ratio between APE1 & actin. (see Supplementary Figure 1). *P < 0.05. (C) Western blot analysis of HCC & SLC tissue lysates patients performed on pooled samples from HCC cancer. Actin was used as loading control & for the relative normalization. A representative image of western blot analysis is shown. Data represent the means of ± SD of three independent experiments. *P < 0.05. (D) Immunohistochemistry of HCC, SLC, & normal (CTRL) tissue. Red & yellow arrows indicate nuclear & cytoplasmic positivity, respectively. (E) Scan of HCC nodules & its corresponding sAPE1 from 3 patients representing for each low, median, & high sAPE1. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/30719231), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: APE Antibody - BSA Free [NB100-101] -
Western Blot: APE Antibody - BSA Free [NB100-101] - Persistent SSBs decrease APE1 levels by affecting its transcription. (A) Western blotting analysis on a representative XRCC1 knockdown time-course. TIG-1 cells were incubated with either a control siRNA (72 h), or a XRCC1-targeting siRNA, & harvested at the indicated time points. DNA damage accumulation is highlighted by PAR formation; APE1 is downregulated in a time-dependent manner. Actin was used as loading control. (B) Alkaline Comet assay on TIG-1 cells harvested 72 h after XRCC1 depletion shows accumulation of SSBs (N = 9). (C) Histogram showing the downregulation in APE1 protein amount 72 h after XRCC1 knockdown, as measured by western blotting (N = 10). (D) Histogram showing the downregulation in APE1 transcript level 72 h after XRCC1 depletion, as measured by qPCR (N = 7). Results depicted in histograms are presented as mean ± SD of the indicated number (N) of independent experiments. Image collected & cropped by CiteAb from the following publication (https://academic.oup.com/nar/article-lookup/doi/10.1093/nar/gkw015), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: APE Antibody - BSA Free [NB100-101] -
Western Blot: APE Antibody - BSA Free [NB100-101] - Downregulation of APE1 in response to BER unbalance is dependent of p53. (A) Representative western blotting analysis on TIG-1 cells depleted of XRCC1 & p53. APE1 is downregulated upon XRCC1 knockdown in a p53-dependent manner. Actin was used as loading control. (B) Left: histogram showing the quantification of APE1 protein amount in the experiment showed in panel A (N = 5). Right: histogram illustrating the amount of APE1 transcript upon combined XRCC1/p53 depletion, as measured through qPCR (N = 3). Results are expressed as mean ± SD of the indicated number (N) of independent experiments. (C) Boxplot showing the distribution of APE1 staining intensity (in arbitrary units) in p53 low versus p53 overexpressing cells. The dashed line highlights the median APE1 intensity in p53 low cells (N > 8000). (D) Representative high-throughput immuno-fluorescence pictures showing TIG-1 cells stained for APE1 (right panel) & p53 (middle panel) after transfection with a p53 expressing plasmid. Cells that downregulating APE1 in response to p53 overexpression are marked by a contour line. Image collected & cropped by CiteAb from the following publication (https://academic.oup.com/nar/article-lookup/doi/10.1093/nar/gkw015), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: APE Antibody - BSA Free [NB100-101] -
Western Blot: APE Antibody - BSA Free [NB100-101] - Excessive AP-endonuclease activity in cells with p53 impairment leads to accumulation of genomic instability. (A) Representative western blotting analysis on WI38 (SV40) cells shows rescue of gamma H2AX staining upon co-depletion of XRCC1 & APE1. (B) Neutral Comet assay on WI38 (SV40) fibroblasts shows accumulation of DSBs upon XRCC1-depletion & rescue after co-depletion of XRCC1 & APE1. Results are expressed as mean ± SD of three independent experiments. (C) Representative western blotting analysis on WI38 (SV40) cells shows rescue of gamma H2AX staining after depletion of XRCC1 in presence of APE1 inhibitor III (APE1i III, 5 μM for 24 h), or AR03 (2.5 μM for 24 h). Actin was used as loading control in all western blotting experiments. Image collected & cropped by CiteAb from the following publication (https://academic.oup.com/nar/article-lookup/doi/10.1093/nar/gkw015), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for APE Antibody - BSA Free
Application
Recommended Usage
Chromatin Immunoprecipitation (ChIP)
1:10-1:500
Flow (Intracellular)
1.0 ug/ml
Flow Cytometry
1.0 ug/ml
Immunocytochemistry/ Immunofluorescence
1:50-1:200
Immunohistochemistry
1:100
Immunohistochemistry Free-Floating
reported in scientific literature (PMID 17332344)
Immunohistochemistry-Frozen
1:100
Immunohistochemistry-Paraffin
1:100
Immunoprecipitation
7 ug/ml
Simple Western
1:12.5
Western Blot
1:1000
Application Notes
In WB this antibody detects a single band at 37 kDa. SH-SY5Y Lysate (nuclear extract) image in western blot provided via verified customer review. In IHC it can be competitively inhibited from recognizing the APE1 antigen in tissues using APE1 protein. This antibody can be used on frozen sections, fixed-paraffin sections and cytospin preps. NB100-101 can also be used following the apoptosis (TUNEL) procedure with the Boehringer-Mannheim TUNEL assay kit. Antibody staining should be performed AFTER the TUNEL assay. NB100-101 can inhibit the repair activity of APE1 protein. In Simple Western only 10 - 15 uL of the recommended dilution is used per data point.
See Simple Western Antibody Database for Simple Western validation: Tested in HeLa lysate 0.1 mg/mL, separated by Size, antibody dilution of 1:12.5, apparent MW was 46 kDa. Separated by Size-Wes, Sally Sue/Peggy Sue.
See Simple Western Antibody Database for Simple Western validation: Tested in HeLa lysate 0.1 mg/mL, separated by Size, antibody dilution of 1:12.5, apparent MW was 46 kDa. Separated by Size-Wes, Sally Sue/Peggy Sue.
Reviewed Applications
Read 3 reviews rated 5 using NB100-101 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Immunogen affinity purified
Formulation
PBS
Format
BSA Free
Preservative
0.02% Sodium Azide
Concentration
1.0 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles.
Background: APE
Long Name
Apurinic/Apyrimidinic Endonuclease
Alternate Names
APEX1
Gene Symbol
APEX1
Additional APE Products
Product Documents for APE Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for APE Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for APE Antibody - BSA Free
Customer Reviews for APE Antibody - BSA Free (3)
5 out of 5
3 Customer Ratings
Have you used APE Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
3 of
3 reviews
Showing All
Filter By:
-
Application: Western BlotSample Tested: Melanoma whole cell lysateSpecies: HumanVerified Customer | Posted 11/20/2014Ape1 in Western
-
Application: ImmunofluorescenceSample Tested: HeLa cellsSpecies: HumanVerified Customer | Posted 07/31/2014IF/Ape1 ab
-
Application: Western BlotSample Tested: SH-SY5Y lysate (nuclear extract)Species: HumanVerified Customer | Posted 02/28/2014Ape1 (1:1,000) detected in nuclear extracts of SH-SY5Y neuroblastoma cells (10 ug total protein per lane)
There are no reviews that match your criteria.
Protocols
View specific protocols for APE Antibody - BSA Free (NB100-101):
Protocol for Flow Cytometry Intracellular Staining
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer (NBP2-26247).
5. Aliquot out 100 uL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeabilization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Generally, our Intracellular Flow Assay Kit (NBP2-29450) is a good place to start as it contains an optimized combination of reagents for intracellular staining as well as an inhibitor of intracellular protein transport (necessary if staining secreted proteins). Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabilization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 15 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 15 minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 1 minute at 400 RCF.
5. Discard supernatant and re-suspend in 100 uL of staining buffer + 0.1% permeabilizer.
6. Add appropriate amount of each antibody (eg. 1 test or 1 ug per sample, as experimentally determined).
7. Mix well and incubate at room temperature for 30 minutes- 1 hour. Gently mix samples every 10-15 minutes.
8. Following the primary/conjugate incubation, add 1-2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 1 minute at 400 RCF.
9. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
10. Add appropriate amount of secondary antibody (as experimentally determined) to each sample.
11. Incubate at room temperature in dark for 20 minutes.
12. Add 1-2 mL of staining buffer and centrifuge at 400 RCF for 1 minute and discard supernatant.
13. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
14. Resuspend in an appropriate volume of staining buffer (usually 500 uL per sample) and proceed with analysis on your flow cytometer.
Sample Preparation.
1. Grow cells to 60-85% confluency. Flow cytometry requires between 2 x 105 and 1 x 106 cells for optimal performance.
2. If cells are adherent, harvest gently by washing once with staining buffer and then scraping. Avoid using trypsin as this can disrupt certain epitopes of interest. If enzymatic harvest is required, use Accutase, Collagenase, or TrypLE Express for a less damaging option.
3. Reserve 100 uL for counting, then transfer cell volume into a 50 mL conical tube and centrifuge for 8 minutes at 400 RCF.
a. Count cells using a hemocytometer and a 1:1 trypan blue exclusion stain to determine cell viability before starting the flow protocol. If cells appear blue, do not proceed.
4. Re-suspend cells to a concentration of 1 x 106 cells/mL in staining buffer (NBP2-26247).
5. Aliquot out 100 uL samples in accordance with your experimental samples.
Tip: When cell surface and intracellular staining are required in the same sample, it is advisable that the cell surface staining be performed first since the fixation and permeabilization steps might reduce the availability of surface antigens.
Intracellular Staining.
Tip: When performing intracellular staining, it is important to use appropriate fixation and permeabilization reagents based upon the target and its subcellular location. Generally, our Intracellular Flow Assay Kit (NBP2-29450) is a good place to start as it contains an optimized combination of reagents for intracellular staining as well as an inhibitor of intracellular protein transport (necessary if staining secreted proteins). Certain targets may require more gentle or transient permeabilization protocols such as the commonly employed methanol or saponin-based methods.
Protocol for Cytoplasmic Targets:
1. Fix the cells by adding 100 uL fixation solution (such as 4% PFA) to each sample for 10-15 minutes.
2. Permeabilize cells by adding 100 uL of a permeabilization buffer to every 1 x 106 cells present in the sample. Mix well and incubate at room temperature for 15 minutes.
a. For cytoplasmic targets, use a gentle permeabilization solution such as 1X PBS + 0.5% Saponin or 1X PBS + 0.5% Tween-20.
b. To maintain the permeabilized state throughout your experiment, use staining buffer + 0.1% of the permeabilization reagent (i.e. 0.1% Tween-20 or 0.1% Saponin).
3. Following the 15 minute incubation, add 2 mL of the staining buffer + 0.1% permeabilizer to each sample.
4. Centrifuge for 1 minute at 400 RCF.
5. Discard supernatant and re-suspend in 100 uL of staining buffer + 0.1% permeabilizer.
6. Add appropriate amount of each antibody (eg. 1 test or 1 ug per sample, as experimentally determined).
7. Mix well and incubate at room temperature for 30 minutes- 1 hour. Gently mix samples every 10-15 minutes.
8. Following the primary/conjugate incubation, add 1-2 mL/sample of staining buffer +0.1% permeabilizer and centrifuge for 1 minute at 400 RCF.
9. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
10. Add appropriate amount of secondary antibody (as experimentally determined) to each sample.
11. Incubate at room temperature in dark for 20 minutes.
12. Add 1-2 mL of staining buffer and centrifuge at 400 RCF for 1 minute and discard supernatant.
13. Wash twice by re-suspending cells in staining buffer (2 mL for tubes or 200 uL for wells) and centrifuging at 400 RCF for 5 minutes. Discard supernatant.
14. Resuspend in an appropriate volume of staining buffer (usually 500 uL per sample) and proceed with analysis on your flow cytometer.
Immunocytochemistry Protocol
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 10% formalin to the dish and fix at room temperature for 10 minutes.
2. Remove the formalin and wash the cells in PBS.
3. Permeablize the cells with 0.1% Triton X100 or other suitable detergent for 10 min.
4. Remove the permeablization buffer and wash three times for 10 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 10 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 10 minutes each.
10. Counter stain DNA with DAPi if required.
Culture cells to appropriate density in 35 mm culture dishes or 6-well plates.
1. Remove culture medium and wash the cells briefly in PBS. Add 10% formalin to the dish and fix at room temperature for 10 minutes.
2. Remove the formalin and wash the cells in PBS.
3. Permeablize the cells with 0.1% Triton X100 or other suitable detergent for 10 min.
4. Remove the permeablization buffer and wash three times for 10 minutes each in PBS. Be sure to not let the specimen dry out.
5. To block nonspecific antibody binding, incubate in 10% normal goat serum from 1 hour to overnight at room temperature.
6. Add primary antibody at appropriate dilution and incubate overnight at 4C.
7. Remove primary antibody and replace with PBS. Wash three times for 10 minutes each.
8. Add secondary antibody at appropriate dilution. Incubate for 1 hour at room temperature.
9. Remove secondary antibody and replace with PBS. Wash three times for 10 minutes each.
10. Counter stain DNA with DAPi if required.
Immunohistochemistry-Paraffin Embedded Sections
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
Antigen Unmasking:
Bring slides to a boil in 10 mM sodium citrate buffer (pH 6.0) then maintain at a sub-boiling temperature for 10 minutes. Cool slides on bench-top for 30 minutes (keep slides in the sodium citrate buffer at all times).
Staining:
1. Wash sections in deionized water three times for 5 minutes each.
2. Wash sections in PBS for 5 minutes.
3. Block each section with 100-400 ul blocking solution (1% BSA in PBS) for 1 hour at room temperature.
4. Remove blocking solution and add 100-400 ul diluted primary antibody. Incubate overnight at 4 C.
5. Remove antibody solution and wash sections in wash buffer three times for 5 minutes each.
6. Add 100-400 ul HRP polymer conjugated secondary antibody. Incubate 30 minutes at room temperature.
7. Wash sections three times in wash buffer for 5 minutes each.
8. Add 100-400 ul DAB substrate to each section and monitor staining closely.
9. As soon as the sections develop, immerse slides in deionized water.
10. Counterstain sections in hematoxylin.
11. Wash sections in deionized water two times for 5 minutes each.
12. Dehydrate sections.
13. Mount coverslips.
Western Blot Protocol
1. Perform SDS-PAGE on samples to be analyzed, loading 10-25 ug of total protein per lane.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturer's instructions.
1. Perform SDS-PAGE on samples to be analyzed, loading 10-25 ug of total protein per lane.
2. Transfer proteins to PVDF membrane according to the instructions provided by the manufacturer of the membrane and transfer apparatus.
3. Stain the membrane with Ponceau S (or similar product) to assess transfer success, and mark molecular weight standards where appropriate.
4. Rinse the blot TBS -0.05% Tween 20 (TBST).
5. Block the membrane in 5% Non-fat milk in TBST (blocking buffer) for at least 1 hour.
6. Wash the membrane in TBST three times for 10 minutes each.
7. Dilute primary antibody in blocking buffer and incubate overnight at 4C with gentle rocking.
8. Wash the membrane in TBST three times for 10 minutes each.
9. Incubate the membrane in diluted HRP conjugated secondary antibody in blocking buffer (as per manufacturer's instructions) for 1 hour at room temperature.
10. Wash the blot in TBST three times for 10 minutes each (this step can be repeated as required to reduce background).
11. Apply the detection reagent of choice in accordance with the manufacturer's instructions.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- ChIP Protocol Video
- Chromatin Immunoprecipitation (ChIP) Protocol
- Chromatin Immunoprecipitation Protocol
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

![Western Blot: APE AntibodyBSA Free [NB100-101] Western Blot: APE AntibodyBSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Western-Blot-NB100-101-img0002.jpg)
![Immunohistochemistry: APE Antibody - BSA Free [NB100-101] Immunohistochemistry: APE Antibody - BSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Immunohistochemistry-NB100-101-img0006.jpg)
![Western Blot: APE AntibodyBSA Free [NB100-101] Western Blot: APE AntibodyBSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Western-Blot-NB100-101-img0012.jpg)
![Immunocytochemistry/ Immunofluorescence: APE Antibody - BSA Free [NB100-101] Immunocytochemistry/ Immunofluorescence: APE Antibody - BSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Immunocytochemistry-Immunofluorescence-NB100-101-img0009.jpg)
![Western Blot: APE AntibodyBSA Free [NB100-101] Western Blot: APE AntibodyBSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Western-Blot-NB100-101-img0008.jpg)
![Flow Cytometry: APE Antibody - BSA Free [NB100-101] Flow Cytometry: APE Antibody - BSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Flow-Cytometry-NB100-101-img0013.jpg)
![Western Blot: APE AntibodyBSA Free [NB100-101] Western Blot: APE AntibodyBSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Western-Blot-NB100-101-img0010.jpg)
![Immunocytochemistry/ Immunofluorescence: APE Antibody - BSA Free [NB100-101] Immunocytochemistry/ Immunofluorescence: APE Antibody - BSA Free [NB100-101]](https://resources.rndsystems.com/images/products/APE-Antibody-Immunocytochemistry-Immunofluorescence-NB100-101-img0007.jpg)
![Western Blot: APE Antibody - BSA Free [NB100-101] - APE Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-101_rabbit-polyclonal-ape-antibody-310202415482249.jpg)
![Western Blot: APE Antibody - BSA Free [NB100-101] - APE Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-101_rabbit-polyclonal-ape-antibody-310202415483616.jpg)
![Western Blot: APE Antibody - BSA Free [NB100-101] - APE Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-101_rabbit-polyclonal-ape-antibody-310202415483620.jpg)
![Western Blot: APE Antibody - BSA Free [NB100-101] - APE Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-101_rabbit-polyclonal-ape-antibody-310202415483627.jpg)
![Western Blot: APE Antibody - BSA Free [NB100-101] - APE Antibody - BSA Free](https://resources.rndsystems.com/images/products/nb100-101_rabbit-polyclonal-ape-antibody-31020241548519.jpg)

-(01-ml)_NB100-101_9121.jpg)