Caspase-8 Antibody - BSA Free
Novus Biologicals | Catalog # NB100-56116

![Immunohistochemistry: Caspase-8 Antibody [NB100-56116] Immunohistochemistry: Caspase-8 Antibody [NB100-56116]](https://resources.rndsystems.com/images/products/Caspase-8-Antibody-active-cleaved-Immunohistochemistry-NB100-56116-img0009.jpg)
Key Product Details
Species Reactivity
Validated:
Cited:
Applications
Validated:
Cited:
Label
Antibody Source
Format
Product Specifications
Immunogen
Reactivity Notes
Specificity
Clonality
Host
Isotype
Theoretical MW
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for Caspase-8 Antibody - BSA Free
Immunohistochemistry: Caspase-8 Antibody [NB100-56116]
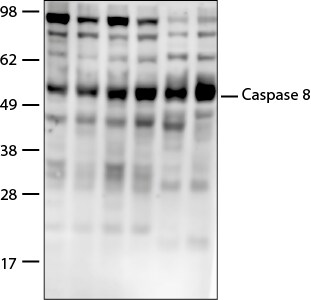
Caspase-8-Antibody-active-cleaved-Immunohistochemistry-NB100-56116-img0009.jpgWestern Blot: Caspase-8 Antibody [NB100-56116]
Western Blot: Caspase-8 Antibody - (active/cleaved) [NB100-56116] - Analysis of active/cleaved Caspase 8 in NRK whole cell lysate using anti-active/cleaved Caspase 8 antibody. WB image submitted by a verified customer review.Immunohistochemistry-Paraffin: Caspase-8 Antibody [NB100-56116]
Immunohistochemistry-Paraffin: Caspase-8 Antibody - (active/cleaved) [NB100-56116] - Sections from a brain tumor tissue array stained for Caspase-8 expression using NB100-56116 at 1:2000. A. Anaplastic glioma (Grade III, left) and Gemistocytoma (Grade II, right) cores showing negative and positive staining for Caspase-8, respectively. B. Higher magnification of the Gemistocytoma tumor (from A).Immunohistochemistry-Paraffin: Caspase-8 Antibody [NB100-56116]
Immunohistochemistry-Paraffin: Caspase-8 Antibody - (active/cleaved) [NB100-56116] - Sections from a brain tissue array stained for Caspase-8 expression using NB100-56116 at 1:2000. A. Normal brain stem (1) and cortex (2). B. Higher magnification of cortex (from A). C. Higher magnification of brain stem (from A). D. Normal cerebellum showing caspase-8 staining in the Purkinge cells.Immunohistochemistry: Caspase-8 Antibody [NB100-56116] -
Immunohistochemistry: Caspase-8 Antibody [NB100-56116] - Necroptosis marker p-MLKL can be detected in OA cartilage & after TNF/CHX stimulation.Exemplary images of impacted & differently stimulated cartilage explants after immunohistochemical staining of a cleaved CASP8, b RIPK3, & c p-MLKL. In addition, percentage of cleaved CASP8− (d) & p-MLKL− (e) positive cells was quantified (n = 3). Images were acquired using a 20× objective; the bars represent 200 µm Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31527653), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunohistochemistry: Caspase-8 Antibody [NB100-56116] -
Immunohistochemistry: Caspase-8 Antibody [NB100-56116] - Expression of necroptosis- & apoptosis-related markers is elevated in OA cartilage.Necroptosis- & apoptosis-related markers in highly degenerated cartilage tissue of OA patients (ICRS grade ≥ 3) were determined by a gene expression analysis of apoptosis & necroptosis markers as well as immunohistochemical analysis (IHC) of b cleaved CASP8, c RIPK3, & d p-MLKL. Macroscopically intact cartilage (ICRS grade ≤ 1) served as control. Bars in the IHC images represent 200 µm. Statistical analysis was performed by an unpaired multiple t test; error bars indicate median & range from min to max; n = 13. Significant differences between groups were depicted as: *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31527653), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: Caspase-8 Antibody [NB100-56116] -
Immunocytochemistry/ Immunofluorescence: Caspase-8 Antibody [NB100-56116] - Analysis of the cell type susceptible to apoptotic cell death in the hypothalamus of IRS2−/− mice. Immunoblots probed with antibodies against GFAP (A) & Tuj-1 (B) in the hypothalamus of wild-type (WT), non-diabetic IRS2-deficient (ND IRS2−/−) & diabetic IRS2-deficient (D IRS2−/−) mice. The average of three independent assays performed in duplicate is shown. Statistical significance by ANOVA: *P<0.05 vs WT mice. n=6/group. (C) Colocalization of TUNEL & GFAP in the hypothalamus of WT, ND IRS2−/− & D IRS2−/− mice. Arrows indicate astrocytes with apoptotic nuclei. (D) Colocalization of cleaved caspase-8 & GFAP, & of cleaved caspase-8 & NeuN, in the hypothalamus of WT, ND IRS2−/− & D IRS2−/− mice. Arrows indicate colocalization of GFAP & cleaved caspase-8. Scale bar: 50 µm; inset, 100 µm. Image collected & cropped by CiteAb from the following publication (https://journals.biologists.com/dmm/article/doi/10.1242/dmm.023515/2571…), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for Caspase-8 Antibody - BSA Free
Immunocytochemistry/ Immunofluorescence
Immunohistochemistry
Immunohistochemistry-Frozen
Immunohistochemistry-Paraffin
Immunoprecipitation
Simple Western
Western Blot
Reviewed Applications
Read 2 reviews rated 4 using NB100-56116 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Formulation
Format
Preservative
Concentration
Shipping
Stability & Storage
Background: Caspase-8
Alternate Names
Gene Symbol
UniProt
Additional Caspase-8 Products
Product Documents for Caspase-8 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for Caspase-8 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for Caspase-8 Antibody - BSA Free
Customer Reviews for Caspase-8 Antibody - BSA Free (2)
Have you used Caspase-8 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Application: Western BlotSample Tested: Mouse Colon and Mouse whole cell lysateSpecies: MouseVerified Customer | Posted 08/16/2021PVDF membrane was probed with Caspase-8 antibody (cat no. NB100-56116) followed by HRP-conjugated Anti-Rabbit IgG Secondary Antibody (CST, Catalog # 7074). I could only detect the pro form.
-
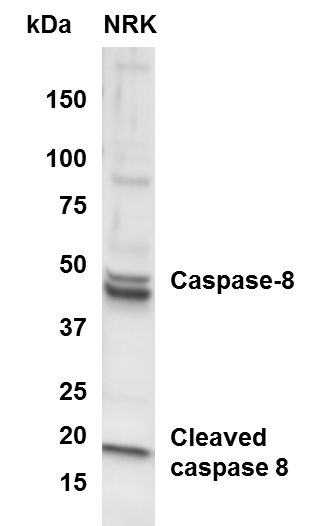
Application: Western BlotSample Tested: NRK whole cell lysateSpecies: RatVerified Customer | Posted 05/22/2015Western Blot of caspase 8 in NRK cells
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Immunoprecipitation Protocol
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Caspase-8 Antibody - BSA Free
-
Q: Hello,I want to perform a Western using this antibody and be able to see both inactive and active forms of caspase-8 using mouse cell line. Can I see an example of such experiment at least with human cell line?
A: I apologize but I do not have an example of a western blot using NB100-56116 to send you. However, we will guarantee it 100% in western blotting using human, mouse and rat samples.
-
Q: May we ask the MW difference between the active form and the proform of Caspase 8 while performing WB?
A: In treated cells induced to undergo apoptosis, caspase-8 migrates as a 55/53 kDa (pro-form), 41/42 kDa (a cleaved/active or intermediate form), and 18 kDa (active form). The proform (55/53 kDa) is still seen in treated cells because not all cells undergo apoptosis at once.
-
Q: We would like to know if any antigen retrieval is required for this antibody (NB100-56116)? And if antigen retrieval is required, could you let us know the recommended protocol?
A:
The requirement of antigen retrieval mainly depends upon for how long the samples were kept in the fixative solution. This step is generally optional if the fixation is done for a brief period, however, I would recommend you include retrieval step to be on the safer side. Different antigen retrieval methods can be found at our Support webpage. In our lab, we generally employ Heat Induced Epitope Retrieval (HIER) method and the protocol for that would be: 1.) Citrate Buffer (10mM Citric Acid, 0.05% Tween 20, pH 6.0): 1.92 g Citric acid (anhydrous) + 1000 ml Distilled water; Mix to dissolve. Adjust pH to 6.0 with 1N NaOH and add 0.5 ml of Tween 20, mix well. 2.) Pre-heat steamer or water bath with staining dish containing Citrate Buffer until temperature reaches 95-100C. Immerse slides in the staining dish. Place the lid loosely on the staining dish and incubate for 20-40 minutes. 3.) Remove the staining dish to room temperature and allow the slides to cool for 20 minutes before proceeding with normal staining procedure.
-
Q: Hello,I want to perform a Western using this antibody and be able to see both inactive and active forms of caspase-8 using mouse cell line. Can I see an example of such experiment at least with human cell line?
A: I apologize but I do not have an example of a western blot using NB100-56116 to send you. However, we will guarantee it 100% in western blotting using human, mouse and rat samples.
-
Q: May we ask the MW difference between the active form and the proform of Caspase 8 while performing WB?
A: In treated cells induced to undergo apoptosis, caspase-8 migrates as a 55/53 kDa (pro-form), 41/42 kDa (a cleaved/active or intermediate form), and 18 kDa (active form). The proform (55/53 kDa) is still seen in treated cells because not all cells undergo apoptosis at once.
-
Q: We would like to know if any antigen retrieval is required for this antibody (NB100-56116)? And if antigen retrieval is required, could you let us know the recommended protocol?
A:
The requirement of antigen retrieval mainly depends upon for how long the samples were kept in the fixative solution. This step is generally optional if the fixation is done for a brief period, however, I would recommend you include retrieval step to be on the safer side. Different antigen retrieval methods can be found at our Support webpage. In our lab, we generally employ Heat Induced Epitope Retrieval (HIER) method and the protocol for that would be: 1.) Citrate Buffer (10mM Citric Acid, 0.05% Tween 20, pH 6.0): 1.92 g Citric acid (anhydrous) + 1000 ml Distilled water; Mix to dissolve. Adjust pH to 6.0 with 1N NaOH and add 0.5 ml of Tween 20, mix well. 2.) Pre-heat steamer or water bath with staining dish containing Citrate Buffer until temperature reaches 95-100C. Immerse slides in the staining dish. Place the lid loosely on the staining dish and incubate for 20-40 minutes. 3.) Remove the staining dish to room temperature and allow the slides to cool for 20 minutes before proceeding with normal staining procedure.
-
Q: Hello,I want to perform a Western using this antibody and be able to see both inactive and active forms of caspase-8 using mouse cell line. Can I see an example of such experiment at least with human cell line?
A: I apologize but I do not have an example of a western blot using NB100-56116 to send you. However, we will guarantee it 100% in western blotting using human, mouse and rat samples.
-
Q: May we ask the MW difference between the active form and the proform of Caspase 8 while performing WB?
A: In treated cells induced to undergo apoptosis, caspase-8 migrates as a 55/53 kDa (pro-form), 41/42 kDa (a cleaved/active or intermediate form), and 18 kDa (active form). The proform (55/53 kDa) is still seen in treated cells because not all cells undergo apoptosis at once.
-
Q: We would like to know if any antigen retrieval is required for this antibody (NB100-56116)? And if antigen retrieval is required, could you let us know the recommended protocol?
A:
The requirement of antigen retrieval mainly depends upon for how long the samples were kept in the fixative solution. This step is generally optional if the fixation is done for a brief period, however, I would recommend you include retrieval step to be on the safer side. Different antigen retrieval methods can be found at our Support webpage. In our lab, we generally employ Heat Induced Epitope Retrieval (HIER) method and the protocol for that would be: 1.) Citrate Buffer (10mM Citric Acid, 0.05% Tween 20, pH 6.0): 1.92 g Citric acid (anhydrous) + 1000 ml Distilled water; Mix to dissolve. Adjust pH to 6.0 with 1N NaOH and add 0.5 ml of Tween 20, mix well. 2.) Pre-heat steamer or water bath with staining dish containing Citrate Buffer until temperature reaches 95-100C. Immerse slides in the staining dish. Place the lid loosely on the staining dish and incubate for 20-40 minutes. 3.) Remove the staining dish to room temperature and allow the slides to cool for 20 minutes before proceeding with normal staining procedure.
Associated Pathways


![Western Blot: Caspase-8 Antibody [NB100-56116] Western Blot: Caspase-8 Antibody [NB100-56116]](https://resources.rndsystems.com/images/products/Caspase-8-Antibody-active-cleaved-Western-Blot-NB100-56116-img0003.jpg)
![Immunohistochemistry-Paraffin: Caspase-8 Antibody [NB100-56116] Immunohistochemistry-Paraffin: Caspase-8 Antibody [NB100-56116]](https://resources.rndsystems.com/images/products/Caspase-8-Antibody-active-cleaved-Immunohistochemistry-Paraffin-NB100-56116-img0004.jpg)
![Immunohistochemistry-Paraffin: Caspase-8 Antibody [NB100-56116] Immunohistochemistry-Paraffin: Caspase-8 Antibody [NB100-56116]](https://resources.rndsystems.com/images/products/Caspase-8-Antibody-active-cleaved-Immunohistochemistry-Paraffin-NB100-56116-img0005.jpg)
![Immunohistochemistry: Caspase-8 Antibody [NB100-56116] - Caspase-8 Antibody](https://resources.rndsystems.com/images/products/nb100-56116_rabbit-polyclonal-caspase-8-antibody-active-cleaved-imgenex-img-5703-310202415392576.jpg)
![Immunohistochemistry: Caspase-8 Antibody [NB100-56116] - Caspase-8 Antibody](https://resources.rndsystems.com/images/products/nb100-56116_rabbit-polyclonal-caspase-8-antibody-active-cleaved-imgenex-img-5703-310202415394549.jpg)
![Immunocytochemistry/ Immunofluorescence: Caspase-8 Antibody [NB100-56116] - Caspase-8 Antibody](https://resources.rndsystems.com/images/products/nb100-56116_rabbit-polyclonal-caspase-8-antibody-active-cleaved-imgenex-img-5703-31020241683918.jpg)