Collagens comprise a large family of insoluble extracellular glycoproteins that are essential components of connective tissues such as tendons, ligaments, cartilage, bone and skin. The mature polypeptides are secreted as coiled, left-handed helices that subsequently assemble into rope-like collagen fibers. Collagen I is a fibril-forming collagen that requires N- and C- terminal processing. Collagen IV is a network forming collagen whose C-terminus forms dimers and N-terminus forms tetramers.
Cultrex 3-D Culture Matrix Rat Collagen I
R&D Systems | Catalog # 3447-020-01

Key Product Details
Features
Species
Product Summary for Cultrex 3-D Culture Matrix Rat Collagen I
Negative for the presence of bacteria and fungi.
Product Specifications
Source
Protein Concentration
Endotoxin Level
Sterility Testing
Cell Culture Testing
Cell Attachment - Tested for the ability to Supports cell attachment and spreading of MG63 human osteosarcoma cells.
Viral Testing
Formulation, Preparation, and Storage
Shipping
Storage
Stability
Background: Collagen I
Alternate Names
Additional Collagen I Products
Product Documents for Cultrex 3-D Culture Matrix Rat Collagen I
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Citations for Cultrex 3-D Culture Matrix Rat Collagen I
Customer Reviews for Cultrex 3-D Culture Matrix Rat Collagen I (2)
Have you used Cultrex 3-D Culture Matrix Rat Collagen I?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Verified Customer | Posted 07/15/2025
-
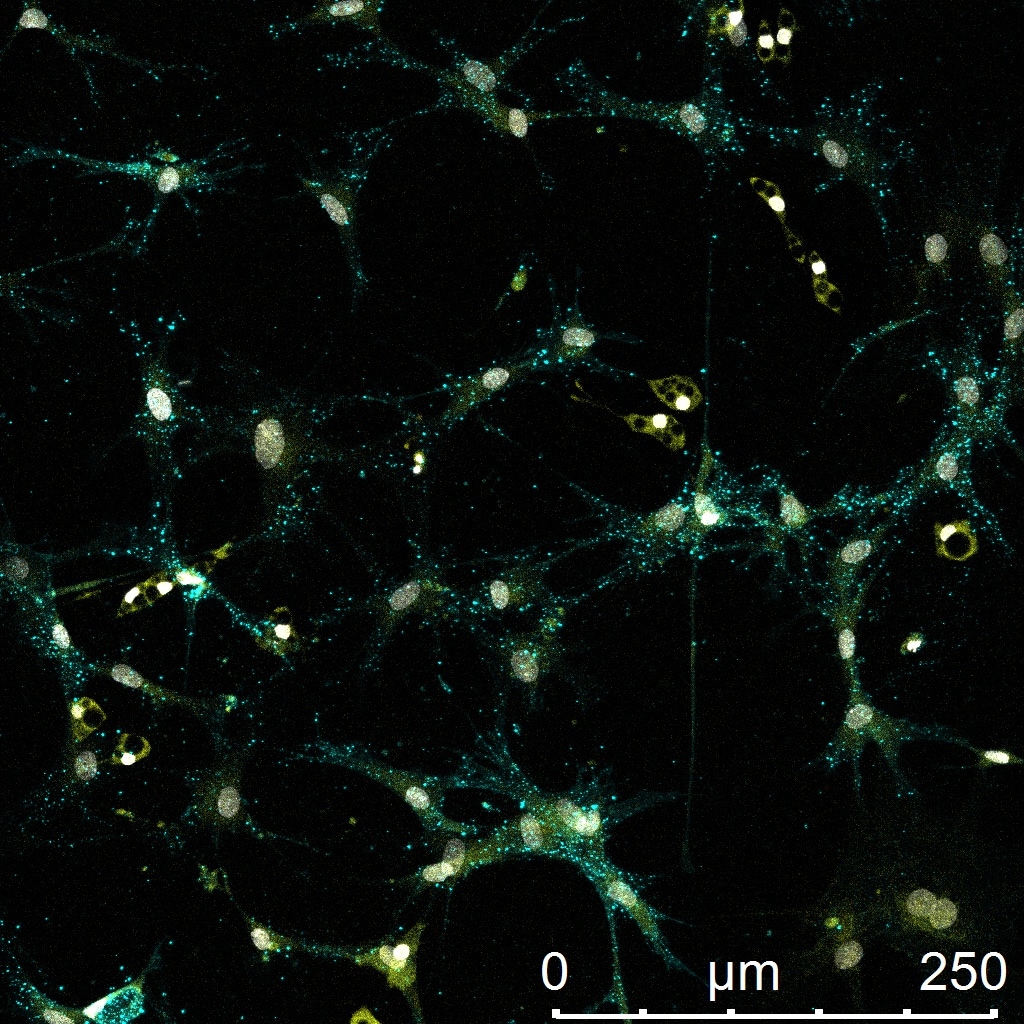
Verified Customer | Posted 03/18/2023Enables a three-dimensional matrix but inhibits proliferation of Schwann cells and neural fibroblasts. Maybe better for other cells. Also sometimes detached from well.
There are no reviews that match your criteria.
FAQs for Cultrex 3-D Culture Matrix Rat Collagen I
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.
-
Q: Can the Organoid Harvesting Solution, Catalog # 3700-100-01, be used to dissociate cells grown on a substrate of Cultrex Rat Collagen 1?
A: The Organoid Harvesting Solution works well with BME (Basement Membrane Extract). For cells grown on Cultrex Rat Collagen 1, the recommended enzyme to use to dissociate cells is Collagenase. Collagenase treatment at 37oC for 30 minutes is recommended.
-
Q: Does the stability of Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) change after opening?
A: We recommend storing the product at 2-8 °C, and provided it has not been neutralized with NaOH or Sodium Bicarbonate, it should remain stable and perform as expected as long as it is used prior to the expiration date.
-
Q: How should cells be cultured prior to setting up the 3-D culture?
A: Cells need to be healthy and actively dividing in 2-D culture. Cells should be passaged two or three times after resuspension from cryopreservation, and they should never surpass 80% confluency during each passage. Cells should also be assessed for viability using trypan blue, and they should exhibit less than 5% staining.
-
Q: Is Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) atelocollagen or telocollagen?
A: Cultrex 3-D Culture Matrix Rat Collagen I (Catalog # 3447-020-01) is telocollagen.
-
Q: What are 3-D cultures?
A: 3-D cultures are in vitro cultures where immortalized cell lines, primary cell lines, stem cells, or tissue explants are placed within hydrogel matrices, such as Cultrex Basement Membrane Extract, that mimic in vivo cell environments and allow cells to proliferate in three dimensions.
-
Q: What are the different types of 3-D culture?
A: The two principal methods for performing 3-D culture are the top assay and embedded assay. For the top assay, cells are seeded on a thick gel of Cultrex Basement Membrane Extract (BME) or Extracellular Matrix Protein. A thin overlay of cell culture medium is then applied to the cells. For the embedded assay, cells are resuspended within a thick gel of Cultrex BME or ECM and the culture media is applied on top. The top assay is easier to setup, to control seeding densities, and to keep cells within one focal plane for analysis.
-
Q: What are the variables associated with 3-D culture?
A: The major variables associated with 3-D culture are cell type, cell seeding density, composition of hydrogel, thickness of hydrogel, stiffness of hydrogel, composition of cell culture medium, and time of culture.
-
Q: What is the advantage of 3-D culture over traditional 2-D culture?
A: While 2-D culture has been used for studying many aspects of cell function and behavior, the tissue-culture treated plastic environment is unlike anything found within living organisms. As a result, cells in 2-D culture exhibit altered morphology, function, proliferation, and gene expression when compared to their emanating tissues. By placing these cells in a 3-D environment, they assume biological and biochemical characteristics similar to what is observed in vivo.
-
Q: What is the difference between the standard Cultrex Rat Collagen I and the Cultrex 3-D Rat Collagen I?
A: Cultrex 3-D Rat Collagen I undergoes the same basic purification and efficacy tests as the standard collagen. However, it undergoes additional 3-D culture validation; it has been tested extensively for the ability to promote growth and differentiation of cell types, visualized by morphology in three dimensions in vitro.
-
Q: What type of analysis is typically applied for organoid or 3-D cell cultures?
A: Within the organoid, spheroid, or 3-D culture, cells may be assessed for morphology, apical/basal polarity, protein localization, and relative proliferation. In addition, cells may be isolated from the 3-D culture and evaluated for levels of RNA and protein expression, as well as modifications to DNA.
-
Q: Will Cultrex 3-D culture Matrix rat Collagen 1, Catalog # 3447-020-01, still work if it has been frozen?
A: This product will likely not form a gel after being frozen.