Fetal bovine serum (FBS), also known as fetal calf serum (FCS), is a commonly used cell culture media supplement. It supplies growth factors, hormones, vitamins, and other nutrients that can support the proliferation and viability of cells in culture. The range of these factors provided by FBS enables the culture of a wide variety of cell types that may have uncharacterized culture requirements. Heat inactivation of FBS destroys potentially detrimental complement proteins while retaining the growth-promoting properties of the serum. Specialty grades of FBS are optimized for the culture of particular cell types such as hybridomas, embryonic stem cells, tetracycline-controlled expression systems, and hormone dependent cells for insulin- and estrogen-related research. Manufacturers of FBS control product variability by managing the raw material supply chain, adhering to consistent processing protocols, and rigorous testing to ensure low endotoxin, hemoglobin, and virus load. USDA-approved testing of FBS is important to certify the virus and disease safety of the source before importing animal material into the United States. USDA APHIS certification enables the use of our US origin serum for the manufacture and export of products outside of the USA.
Fetal Bovine Serum - Optima
R&D Systems | Catalog # S12450

Key Product Details
Features
Key Benefits
- US-origin and USDA-Aphis certified
- Suitable for general cell culture applications
- High lot-to-lot consistency
- Excellent cell growth characteristics, low endotoxin, and low hemoglobin levels
- Manufactured in our ISO 9001:2015 certified facility
- Formerly sold under the Atlanta Biologicals brand
Species
Applications
Product Summary for Fetal Bovine Serum - Optima
All lots of Fetal Bovine Serum – Optima are sterility, virus, and performance tested, and include a biochemical profile. Lots are manufactured from USA origin material following stringent manufacturing and quality control procedures. Optima FBS is USDA-APHIS certified and is manufactured in our ISO 9001:2015 certified facility.
Fetal Bovine Serum and other serum products should be stored and handled correctly to assure long-term stability and to preserve growth performance consistency throughout its shelf-life. In addition, heat inactivation of serum is frequently desired to inactivate complement within the serum. Below are protocol links for serum storage/handling and heat inactivation.
Protocol for Fetal Bovine Serum Storage, Thawing, and Freezing.
Protocol for Heat Inactivation of Serum Products.
Virus Testing - Fetal Bovine Serum – Optima undergoes extended testing for adventitious viruses including: Bovine Viral Diarrhea Virus (BVDV), Infectious Bovine Rhinotracheitis Virus (IBRV), Parainfluenza-3 Virus (PI-3V), Bluetongue Virus (BTV), Bovine Respiratory Syncytial Virus (BRSV), Bovine Parvovirus (BPV), Bovine Adenovirus, Type 3 (BAV-3), Bovine Adenovirus, Type 5 (BAV-5), Reo virus, and Rabies virus.
Biochemical Testing - R&D Systems FBS undergo biochemical profiling including total protein, albumin, globulin, IgG, total bilirubin, glucose, iron, cholesterol, triglycerides, sodium and potassium. View the product insert for a complete list of biochemical profiling components, which may vary between grades.
Chemical Analysis - Each lot of FBS is tested for osmolality, pH, hemoglobin, endotoxin (LAL method) and microbiological contamination (using methods recommended by the U.S. Pharmacopeia).
Product Specifications
Osmolality
Sterility Testing
Cell Culture Testing
Viral Testing
Additional Testing
Endotoxin - ≤ 20.0 EU/mL by Limulus Amebocyte Lysate (LAL) Assay
Hemoglobin - 25 mg/dL
Scientific Data Images for Fetal Bovine Serum - Optima
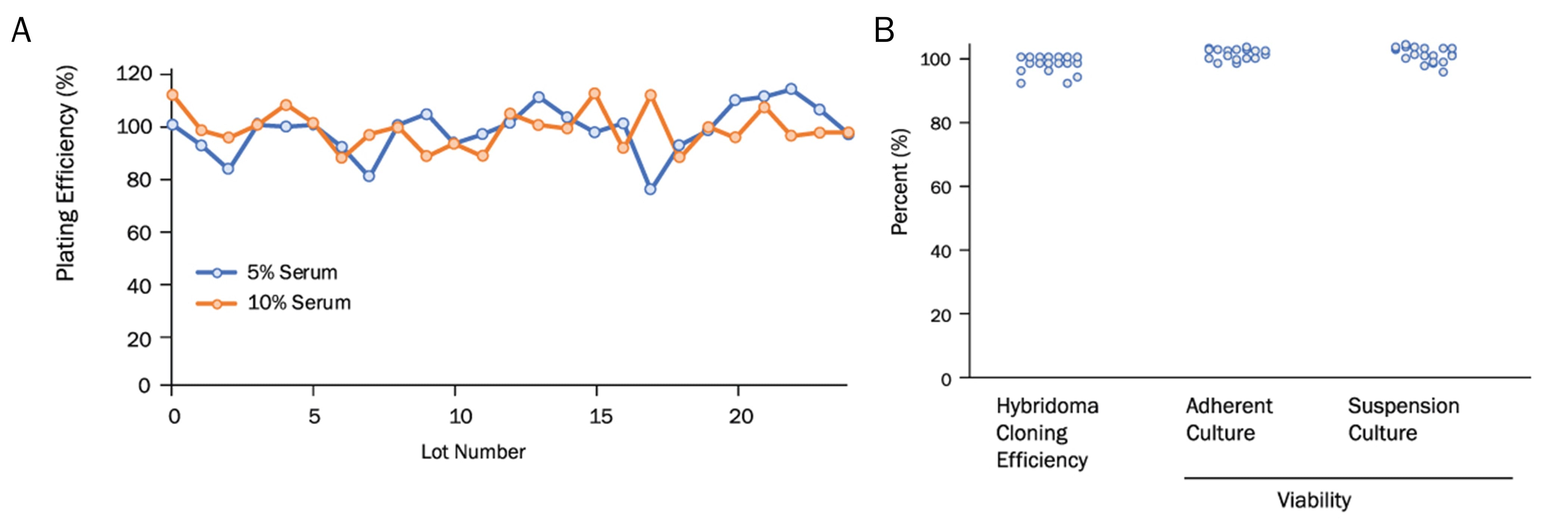
Optima FBS has Consistent Lot-to-Lot Performance.
Quality control data is displayed across multiple lots of Optima FBS. (A) Plating efficiency of cells cultured in media containing 5% or 10% Optima FBS, relative to control lots of FBS. (B) Optima FBS was tested for its ability to support hybridoma cloning as well as the healthy expansion of adherent and suspension cell culture lines. Lots of Optima FBS show consistent and high percentage cloning efficiency and cell viability.Optima FBS Support Expansion and Health Across Cell Lines.
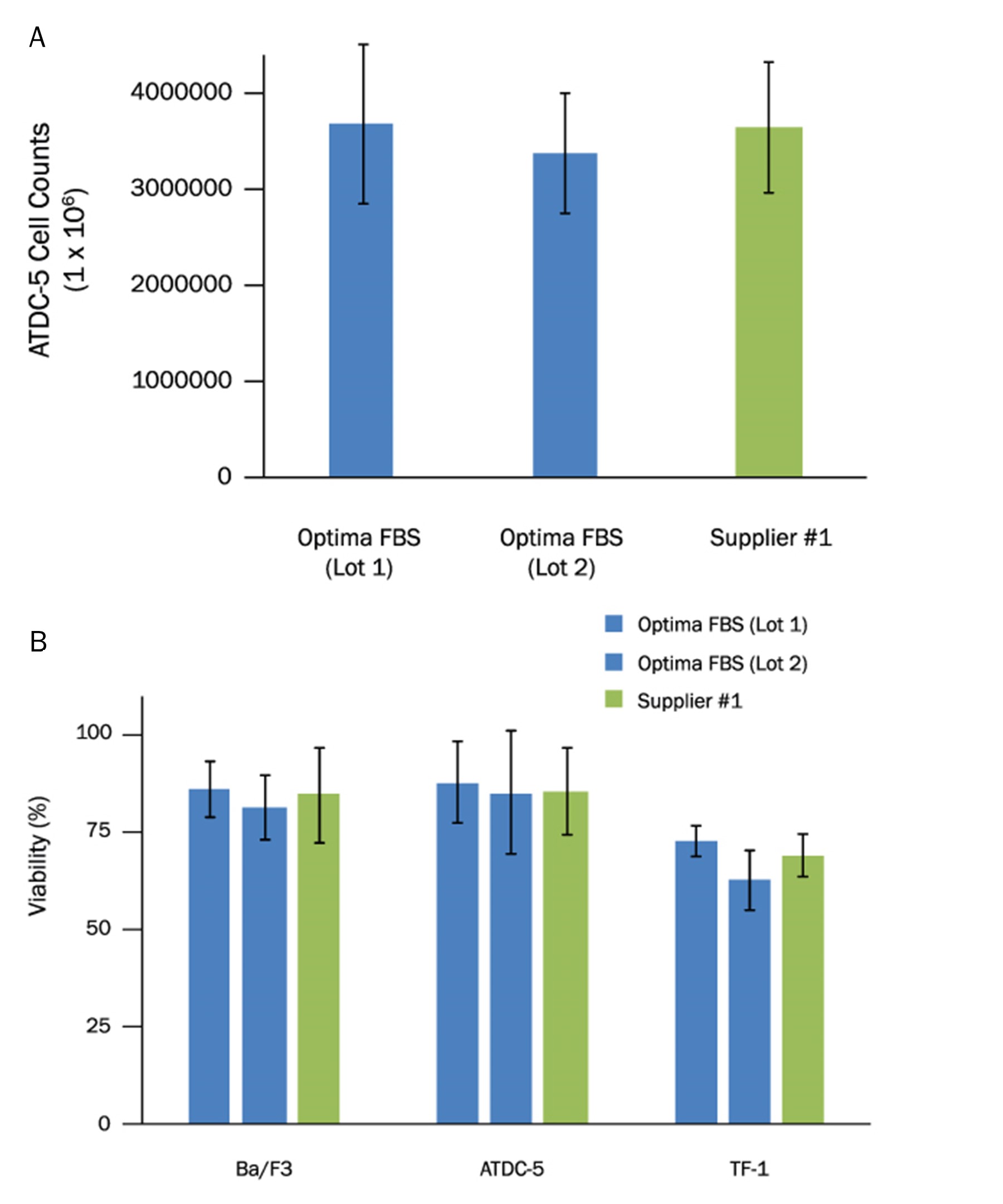
Ba/F3 murine pro-B cell line, ATDC-5 murine teratocarcinoma cell line, and a TF-1 erythroleukemic cell line were evaluated for expansion and health in culture media supplemented with either Optima FBS or FBS from an alternate supplier (Suppler #1). (A) Two lots of Optima FBS showed consistent expansion rates for ATDC-5 cells. The expansion was similar to FBS from another supplier. (B) Ba/F3, ATDC-5, and TF-1 cell lines cultured in Optima FBS show consistent cell viability across multiple lots. The viability was comparable to an alternate supplier of FBS.Formulation, Preparation, and Storage
Purification
Shipping
Storage
Background: Fetal Bovine Serum (FBS)
Product Documents for Fetal Bovine Serum - Optima
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Note: Certificate of Analysis not available for kit components.
Citations for Fetal Bovine Serum - Optima
Customer Reviews for Fetal Bovine Serum - Optima (1)
Have you used Fetal Bovine Serum - Optima?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
-
Sample Tested: fibroblastsSpecies: MouseAssay Type: Ex VivoVerified Customer | Posted 03/29/2026Mouse lung fibroblastMouse lung fibroblast cultured using 10 % FBS
There are no reviews that match your criteria.
Protocols
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- A Guide to the Colony Forming Cell Assay: Methods and Tips
- Cultrex UltiMatrix BME Protocol
- Culture of Mouse Enteric Organoids using Cultrex Basement Membrane Extract
- Culturing BG01V Human Embryonic Stem Cells with Mouse Embryonic Fibroblast (MEF)-Conditioned Media
- Culturing Mouse Cortical Stem Cells: Expansion using the Neurosphere System
- Culturing Rat Cortical Stem Cells: Expansion using the Monolayer System
- Culturing Rat Cortical Stem Cells: Expansion using the Neurosphere System
- Definitive Endoderm Differentiation of BG01V Human Embryonic Stem Cells
- General Support Buffers
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Protocol for CEPT Cocktail Kit
- Protocol for Cell Counting Kit-8
- Protocol for Culturing Embryonic Chick Dorsal Root Ganglion Neurons
- Protocol for Culturing Embryonic Rat Spinal Motor Neurons
- Protocol for Culturing Rat Hippocampal Neurons
- Protocol for Culturing Rat Microglia
- Protocol for MortaLIVE™ Non-Toxic Dye
- Protocol for the Differentiation and Characterization of Human Th1 Cells
- Rat Cortical Stem Cell Culture Expansion Protocol
- The Human Colony Forming Cell (CFC) Assay using Methylcellulose-based Media
- The Mouse Colony Forming Cell (CFC) Assay using Methylcellulose-based Media
- The Mouse Pre-B Colony Forming Cell (CFC) Assay Using Methylcellulose-based Media
- Use of Cultrex Basement Membrane Extract to Improve Take and Growth of Xenografts in Mice
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
FAQs for Fetal Bovine Serum - Optima
-
Q: Should I use heat inactivated fetal bovine serum?
A: Heat inactivation, in general, decreases the growth promoting properties of the serum and causes increased formation of crystalline or flocculent precipitates (salts, proteins, lipids, fibrin, etc.) in the Fetal Bovine Serum (FBS) as well as in other serum products. In fact, for many cell culture applications, heat inactivation may be unnecessary. Warming the serum-containing medium to 37˚C prior to use, as is the practice in many cell culture laboratories, is often sufficient to inactivate heat-labile complement factors. Heat inactivated FBS is frequently used in immunological applications.Since every cell type has different growth requirements, the choice of whether to heat inactivate your serum or not must be made by the researcher. If you have concerns about whether to use heat inactivated serum with your cell line or not, perform a side-by-side comparison of cell growth with heat inactivated versus non-heat inactivated serum.
-
Q: What is the difference between Fetal Bovine Serum (FBS) and Fetal Calf Serum (FCS)?
A: There is no difference. These are different names for the same product. Serum derived from blood of bovine fetuses is referred to as Fetal Bovine Serum or Fetal Calf Serum.
-
Q: Should I use heat inactivated fetal bovine serum?
A: Heat inactivation, in general, decreases the growth promoting properties of the serum and causes increased formation of crystalline or flocculent precipitates (salts, proteins, lipids, fibrin, etc.) in the Fetal Bovine Serum (FBS) as well as in other serum products. In fact, for many cell culture applications, heat inactivation may be unnecessary. Warming the serum-containing medium to 37˚C prior to use, as is the practice in many cell culture laboratories, is often sufficient to inactivate heat-labile complement factors. Heat inactivated FBS is frequently used in immunological applications.Since every cell type has different growth requirements, the choice of whether to heat inactivate your serum or not must be made by the researcher. If you have concerns about whether to use heat inactivated serum with your cell line or not, perform a side-by-side comparison of cell growth with heat inactivated versus non-heat inactivated serum.
-
Q: What is the difference between Fetal Bovine Serum (FBS) and Fetal Calf Serum (FCS)?
A: There is no difference. These are different names for the same product. Serum derived from blood of bovine fetuses is referred to as Fetal Bovine Serum or Fetal Calf Serum.