GABA-AR alpha 6 Antibody
Novus Biologicals | Catalog # NB300-150

![Western Blot: GABA-AR alpha 6 AntibodyAzide and BSA Free [NB300-150] Western Blot: GABA-AR alpha 6 AntibodyAzide and BSA Free [NB300-150]](https://resources.rndsystems.com/images/products/GABA-A-R-alpha-6-Antibody-Western-Blot-NB300-150-img0001.jpg)
Loading...
Key Product Details
Species Reactivity
Validated:
Mouse, Rat
Cited:
Mouse
Applications
Validated:
Immunohistochemistry, Western Blot
Cited:
IF/IHC
Label
Unconjugated
Antibody Source
Polyclonal Rabbit IgG
Loading...
Product Specifications
Immunogen
Synthetic peptide corresponding to amino acid residues specific to the alpha 6 subunit conjugated to KLH. Accession # P30191
Specificity
Specific for endogenous levels of the ~57 kDa alpha 6-subunit of the GABAA receptor.
Clonality
Polyclonal
Host
Rabbit
Isotype
IgG
Theoretical MW
57 kDa.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Description
Recommended that the undiluted antibody be aliquoted into smaller working volumes (10-30 uL/vial depending on usage).
Scientific Data Images for GABA-AR alpha 6 Antibody
Western Blot: GABA-AR alpha 6 AntibodyAzide and BSA Free [NB300-150]
Western Blot: GABA-A R alpha 6 Antibody [NB300-150]Applications for GABA-AR alpha 6 Antibody
Application
Recommended Usage
Immunohistochemistry
1:10-1:500
Western Blot
1:1000
Application Notes
In Western blot a band at approximately 57 kDa is seen. Use in Immunohistochemistry reported in scientific literature (PMID 18509076)
Formulation, Preparation, and Storage
Purification
Unpurified
Formulation
Neat whole antisera
Preservative
No Preservative
Concentration
This product is unpurified. The exact concentration of antibody is not quantifiable.
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Store at -20C. Avoid freeze-thaw cycles.
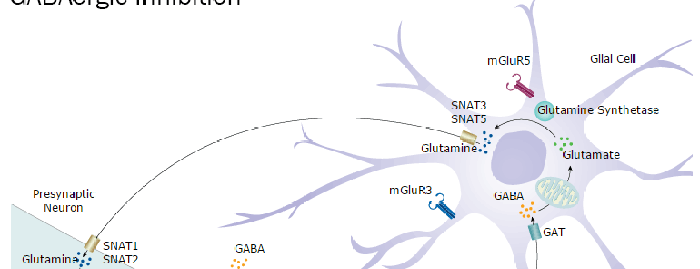
Background: GABA-A R alpha 6
Long Name
gamma-Aminobutyric Acid Type A Receptor, alpha-6 Polypeptide
Alternate Names
GABAAR alpha 6, GABAARa6, GABRA6
Gene Symbol
GABRA6
UniProt
Additional GABA-A R alpha 6 Products
Product Documents for GABA-AR alpha 6 Antibody
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for GABA-AR alpha 6 Antibody
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Related Research Areas
Citations for GABA-AR alpha 6 Antibody
Customer Reviews for GABA-AR alpha 6 Antibody
There are currently no reviews for this product. Be the first to review GABA-AR alpha 6 Antibody and earn rewards!
Have you used GABA-AR alpha 6 Antibody?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Protocols
View specific protocols for GABA-AR alpha 6 Antibody (NB300-150):
Western Blot protocol specific for GABA A Receptor alpha 6 Antibody (NB300-150):
1. Pour lower gels according to recipes layer with about 300 ul ethanol and allow to polymerize at least 45 minutes. 7.5% SDS-PAGE gels work well for Synapsin, NR2A, 2B,and 2C antibodies.
2. Rinse off ethanol with water. Shake and/or use a kimwipe to remove excess water. Pour upper gels (stacks) and insert combs. Let polymerize about 15-20 minutes. Remove combs, making sure you pull them straight out, and rinse with water.
3. Attach gel holders to running electrode apparatus and fill chamber with 1X running buffer.
4. Load gels beginning with 10 ul of the kaleidoscope molecular weight marker.
5. Attach electrodes to power source and run gels at 200 Volts for about 45 minutes or until dye front runs down past gray gasket.
6. Turn off power source and remove gel holders from running apparatus first and then carefully remove plates from holders. Remove one plate and leave gel attached to the other plate. Use a spacer or the green scraper to cut off stacks and discard.
7. Place plate with attached gel in some 1X transfer buffer and let equilibrate while you assemble the transfer genie. Transfer buffer + 20% Methanol is standard for many antibodies. NMDA antibodies seem to look a little better in transfer buffer with 5% Methanol +.05% SDS.
8. Wearing gloves cut PVDF membranes to gel size and wet in Methanol to activate for about 30 seconds. Rinse 2-3 times in water. Be sure to keep membrane wet at all times. Put membrane in some 1X transfer buffer until you are ready to use it.
9. Assemble the genie transfer apparatus per instructions on wall. Fill with 1X transfer buffer.
10. Carefully place gels on filter paper and then place PVDF membranes on top of gels making sure there are no air bubbles. Use the sawed-off pipette to roll over sandwich. Complete the assembly of transfer apparatus making sure there is enough buffer to come to the top of the scotch-brite pads.
11. Clean off electrodes with a Q-tip and attach to battery charger. Plug charger in and set to 6 Volts for the mini-genie and 12 Volts for the large genie. Transfer gel for 1.5-2 hours.
12. Take down transfer apparatus and rinse blot a couple of times in water. Place blot on kimwipe and let air dry about 10-15 minutes to fix proteins. Reactivate membranes by rewetting in Methanol and rinsing in water.
13. Block blots in 5% Non-fat dry milk-TTBS for 30 minutes while shaking at room temperature. Blocking time may be increased to an hour if blots look dirty. It is not necessary to block when working with the Synapsin antibody. Milk works great for the NMDA antibodies, but when working with phospho-site or other antibodies that don't like milk, use 3% BSA-TTBS to block.
14. Incubate blots overnight in cold-room in primary antibody diluted in 1% milk TTBS or 1% BSA-TTBS.
15. Decant unbound primary antibody solution (save in fridge) and wash blot 3 x 10 minutes in TTBS.
16. Incubate blots in secondary antibody at a 1:10,000 1:30,000 dilution in 1% Milk or 1% BSA for 1 hour while shaking at room temperature. Use Goat Anti-Rabbit HRP for polyclonals and Goat Anti-Mouse HRP for monoclonals.
17. Decant secondary antibody solution and wash blots 3 x 15 minutes in TTBS or use TTBS + 0.1% Triton X-100 to reduce excessive background if needed.
18. ECL Detect---- Mix equal volumes of each reagent in the Pierce Super Signal ECL kit using just enough to cover blots (0.125 ml/cm of membrane). I use 1 ml of each for a total of 2 mls per standard size blot. Vortex ECL solution briefly and incubate blots in substrate for 1 minute only. Pour off excess ECL solution and blot with a kimwipe to further remove excess. Place blot on a piece of plastic sheet protector and put into the Alpha Chemi-Imager to visualize bands. Set exposures from roughly 15 seconds to 4 minutes.
1. Pour lower gels according to recipes layer with about 300 ul ethanol and allow to polymerize at least 45 minutes. 7.5% SDS-PAGE gels work well for Synapsin, NR2A, 2B,and 2C antibodies.
2. Rinse off ethanol with water. Shake and/or use a kimwipe to remove excess water. Pour upper gels (stacks) and insert combs. Let polymerize about 15-20 minutes. Remove combs, making sure you pull them straight out, and rinse with water.
3. Attach gel holders to running electrode apparatus and fill chamber with 1X running buffer.
4. Load gels beginning with 10 ul of the kaleidoscope molecular weight marker.
5. Attach electrodes to power source and run gels at 200 Volts for about 45 minutes or until dye front runs down past gray gasket.
6. Turn off power source and remove gel holders from running apparatus first and then carefully remove plates from holders. Remove one plate and leave gel attached to the other plate. Use a spacer or the green scraper to cut off stacks and discard.
7. Place plate with attached gel in some 1X transfer buffer and let equilibrate while you assemble the transfer genie. Transfer buffer + 20% Methanol is standard for many antibodies. NMDA antibodies seem to look a little better in transfer buffer with 5% Methanol +.05% SDS.
8. Wearing gloves cut PVDF membranes to gel size and wet in Methanol to activate for about 30 seconds. Rinse 2-3 times in water. Be sure to keep membrane wet at all times. Put membrane in some 1X transfer buffer until you are ready to use it.
9. Assemble the genie transfer apparatus per instructions on wall. Fill with 1X transfer buffer.
10. Carefully place gels on filter paper and then place PVDF membranes on top of gels making sure there are no air bubbles. Use the sawed-off pipette to roll over sandwich. Complete the assembly of transfer apparatus making sure there is enough buffer to come to the top of the scotch-brite pads.
11. Clean off electrodes with a Q-tip and attach to battery charger. Plug charger in and set to 6 Volts for the mini-genie and 12 Volts for the large genie. Transfer gel for 1.5-2 hours.
12. Take down transfer apparatus and rinse blot a couple of times in water. Place blot on kimwipe and let air dry about 10-15 minutes to fix proteins. Reactivate membranes by rewetting in Methanol and rinsing in water.
13. Block blots in 5% Non-fat dry milk-TTBS for 30 minutes while shaking at room temperature. Blocking time may be increased to an hour if blots look dirty. It is not necessary to block when working with the Synapsin antibody. Milk works great for the NMDA antibodies, but when working with phospho-site or other antibodies that don't like milk, use 3% BSA-TTBS to block.
14. Incubate blots overnight in cold-room in primary antibody diluted in 1% milk TTBS or 1% BSA-TTBS.
15. Decant unbound primary antibody solution (save in fridge) and wash blot 3 x 10 minutes in TTBS.
16. Incubate blots in secondary antibody at a 1:10,000 1:30,000 dilution in 1% Milk or 1% BSA for 1 hour while shaking at room temperature. Use Goat Anti-Rabbit HRP for polyclonals and Goat Anti-Mouse HRP for monoclonals.
17. Decant secondary antibody solution and wash blots 3 x 15 minutes in TTBS or use TTBS + 0.1% Triton X-100 to reduce excessive background if needed.
18. ECL Detect---- Mix equal volumes of each reagent in the Pierce Super Signal ECL kit using just enough to cover blots (0.125 ml/cm of membrane). I use 1 ml of each for a total of 2 mls per standard size blot. Vortex ECL solution briefly and incubate blots in substrate for 1 minute only. Pour off excess ECL solution and blot with a kimwipe to further remove excess. Place blot on a piece of plastic sheet protector and put into the Alpha Chemi-Imager to visualize bands. Set exposures from roughly 15 seconds to 4 minutes.
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...
Associated Pathways