GAP-43 Antibody - BSA Free
Novus Biologicals | Catalog # NB300-143

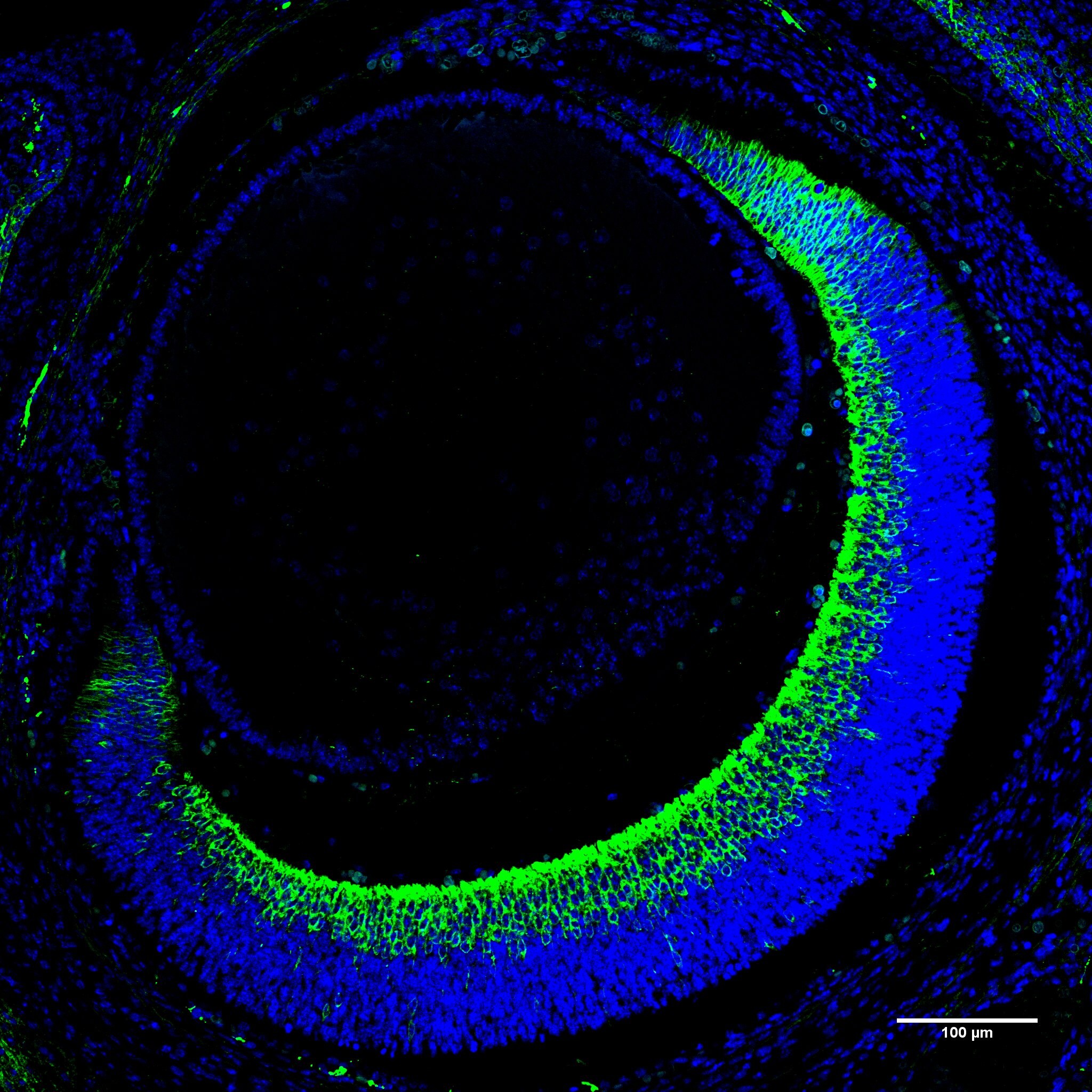
![Immunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143] Immunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunohistochemistry-Paraffin-NB300-143-img0019.jpg)
Key Product Details
Validated by
Biological Validation
Species Reactivity
Validated:
Human, Mouse, Rat, Porcine, Bovine, Canine, Chicken, Drosophila, Equine, Primate
Cited:
Human, Mouse, Rat, Porcine, Avian - Chicken, Canine, Insect - Drosophila, Insect - Drosophila melanogaster, Primate
Applications
Validated:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Flow Cytometry, Immunocytochemistry/ Immunofluorescence
Cited:
Immunohistochemistry, Immunohistochemistry-Paraffin, Immunohistochemistry-Frozen, Western Blot, Immunocytochemistry/ Immunofluorescence, IF/IHC
Label
Unconjugated
Antibody Source
Polyclonal Rabbit
Format
BSA Free
Loading...
Product Specifications
Immunogen
C-terminal peptide of rodent GAP43, KEDPEADQEHA coupled to KLH.
Reactivity Notes
Reactivity to Canine, Chicken, and Primate reported in scientific literature (PMID: 30647968, 30819546, and 24249398 respectively). Drosophila reactivity reported in scientific literature (PMID: 30819546).
Localization
Neuronal Marker, developing and regenerating axons and growth cone.
Marker
Neuronal Marker
Clonality
Polyclonal
Host
Rabbit
Theoretical MW
43 kDa.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Disclaimer note: The observed molecular weight of the protein may vary from the listed predicted molecular weight due to post translational modifications, post translation cleavages, relative charges, and other experimental factors.
Scientific Data Images for GAP-43 Antibody - BSA Free
Immunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143]
Immunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143] - Review image from confirmed customer on mouse E15.5 paraffin sections.Western Blot: GAP-43 Antibody [NB300-143]
Western Blot: GAP-43 Antibody [NB300-143] - Different tissue and cell lysates using rabbit pAb to GAP43, dilution 1:20,000 in green: [1] protein standard (red), [2] rat brain, [3] rat spinal cord, [4] mouse brain, [5] mouse spinal cord, [6] SH-SY5Y cells, [7] C6 cells. Single band at 43 kDa mark corresponds to GAP43 protein. The GAP43 protein is detected only in the lysates of neuronal origin. C6 cells are a rat glioma cell line and do not express GAP43 proteinImmunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143]
GAP-43-Antibody-Immunohistochemistry-Paraffin-NB300-143-img0023.jpgImmunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]
Immunocytochemistry/Immunofluorescence: GAP-43 Antibody [NB300-143] - Cortical neuron-glial cell culture from E20 rat stained with rabbit pAb to GAP43, dilution 1:2,000 in green, and costained with mouse mAb to vimentin, dilution 1:2,000, in red. Blue: DAPI staining of nuclear DNA. GAP43 antibody labels protein expressed in the axonal membrane of neuronal cells, while vimentin antibody stains intermediate filaments in fibroblasts and other non-neuronal cells.Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]
Immunocytochemistry/Immunofluorescence: GAP-43 Antibody [NB300-143] - Rat E18 mixed neuron/glia cultures with rabbit GAP43 (red) and 5B10, mouse monoclonal to MAP-tau (green).Western Blot: GAP-43 Antibody [NB300-143]
Western Blot: GAP-43 Antibody [NB300-143] - Western blots of homogenate of cow cerebellum stained with RPCA-GAP-43. A prominent band running at ~43kDa represents the full length GAP-43.Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]
Immunocytochemistry/Immunofluorescence: GAP-43 Antibody [NB300-143] - Immunofluorescence of GAP-43 (green), a molecular marker of neurite outgrowth, demonstrates intense staining in overexpressing wild-type PS-1 (E) PC-12 cells. (Teo, et al, 2005)Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]
Immunocytochemistry/Immunofluorescence: GAP-43 Antibody [NB300-143] - Mixed neuron-glial cultures stained with RPCA-GAP43 (red), blue is DNA staining.Western Blot: GAP-43 Antibody [NB300-143] -
Western Blot: GAP-43 Antibody [NB300-143] - Silencing TFAP4 induces neuroblastoma cell differentiation. a Morphology of neuroblastoma cells after silencing TFAP4. Cells were grown in culture for 8 days, with 1 μg/ml doxycycline (shRNA induced) or without doxycycline. b Neurites # per field, & neurite length (pixels) were quantified with or without doxycycline. Mean ± std dev. *, P < 0.05; **, P < 0.01, ****, P < 0.0001. cGAP43 expression level (qRT-PCR) & protein level (western blot) in neuroblastoma cells. Gene expression was measured by quantitative PCR 4 days after induction of shRNA against TFAP4. Protein samples for western blot were collected from MYCN-amplified cells that were cultured with or without doxycycline (1 μg/ml) for 6 days Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/29880876), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] -
Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] - LAMP1 increases in SDC-null corneas 14 days after trephine wound. Representative 3D images rotated to generate cross-sectional views show the localization of GAP43 (green), beta III tubulin (red), & LAMP1 (blue) in WT (top) & SDC1-null (bottom) corneas. These images show staining within a 0.5-μm cross-sections. Data indicate that axon fragments are being degraded (arrows). Representative INTs (*) & SBNs (#) have been highlighted & enlarged 3-fold. When LAMP1 expression within apical & basal cell layers 14 days after trephine injury is quantified & compared with unwounded controls (Fig. 2B), LAMP1 increases over 3-fold in SDC1-null corneas but remains the same in WT corneas. Scale bar: 10 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28973369), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Western Blot: GAP-43 Antibody [NB300-143] -
Western Blot: GAP-43 Antibody [NB300-143] - Obesity & diabetes led to white adipose tissue neuropathy.The BTBR ob/ob (MUT) model of obesity & diabetes was compared to BTBR +/+ wild-type (WT) for body weight measurements (a), & adiposity (b). Body & tissue weight data were analyzed by two-tailed Student’s t-test. Von Frey tactile allodynia analysis was performed on MUT & WT animals to determine onset of peripheral neuropathy (c). Von Frey data was analyzed by ANOVA with Sidak’s post hoc test. For (a-c), all males; WT N = 8, 12–20 weeks old; MUT N = 6, 12–24 weeks old. Protein levels of PGP9.5 (d), as well as TH (e) in inguinal scWAT of the MUT & WT mice were measured by western blotting. For (e), lane 5 was excluded from analyses due to uneven resolution of housekeeper. For (d-e), all males; WT N = 5, 12–20 weeks old & MUT N = 4, 12–20 weeks old. Protein expression of PGP9.5 (f), TH (g), PSD95 (h), & GAP43 (i) in inguinal scWAT of 12–25 weeks old WT, 12 week old MUT & 24–28 week old MUT was measured by western blotting. All males, N = 3 per group. Western blot data were normalized to either beta -Tubulin or Cyclophilin B, band intensity were quantified in Image J, & analyzed by two-tailed Student’s t-test. Error bars are SEMs. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/31509546), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] -
Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] - Intraepithelial nerve terminals extension into the apical most cell layers increases in SDC1-null corneas after trephine injury. Representative 3D images rotated to generate cross-sectional views show the localization of beta III tubulin (red) & GAP43 (green) at 4, 14, & 28 days after trephine wounding in WT & SDC1-null corneas. Punctate GAP43 is increased within the epithelium in SDC1-null compared with WT corneas at 14 days (*). beta III tubulin+ & GAP43+ INTs in the apical most cell layers were quantified as a function of time after trephine injury & normalized relative to control (presented in Fig. 2A). Asterisks within bars indicate differences that are significant relative to unwounded controls; asterisks between bars indicate significant differences between time points. In WT corneas, the numbers of beta III tubulin+ & GAP43+ INTs extending apically decrease relative to controls at 28 days for beta III tubulin+ INTs & at 4 & 28 days for GAP43+ INTS, in SDC1-null corneas, both beta III tubulin+ & GAP43+ INTs increase at all time points assessed. Scale bar: 25 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28973369), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] -
Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] - Differences in INTs & LAMP1 localization in WT & SDC1-null adult corneas. Representative high-resolution confocal 3D images rotated as indicated in the cartoon to the left were used to generate cross sectional views to show the localization of beta III tubulin (red) & GAP43 (green) in intraepithelial nerve terminals in control WT & SDC1-null corneas. The images shown in (A) project through 135 μm of tissue, images in (B) project through 0.5 μm of tissue. (A) Intraepithelial corneal nerves are shown to be linear, arise from the subbasal nerves (SBNs), & project apically. Quantitation of beta III tubulin & GAP43 within apical & basal layers of the corneal epithelium reveals that WT corneas have significantly more INTs in both basal & apical cell layers compared with SDC1-null corneas. Although SDC1-null corneas have fewer INTs, they are equally capable of extending toward the apical most cell layers. (B) In the 0.5-μm cross-sectional slices through the tissue, INTs appear discontinuous. In addition to beta III tubulin & GAP43, LAMP1 is shown in blue. LAMP1 is a lysosomal marker; co-localization of GAP43 and/or beta III tubulin within corneal epithelial cell lysosomes indicate that axon fragments have been phagocytozed & are being degraded (arrows). The sites indicated by the asterisks have been digitally enlarged 3-fold. LAMP1 was quantified in WT & SDC1-null corneas. Data show that cells within the apical layers express more LAMP1 compared with basal cells in both genotypes of mice. In addition, WT corneas express 2- to 3-fold more LAMP1 in apical & basal layers compared with SDC1-null corneas. Scale bar: 10 μm. Image collected & cropped by CiteAb from the following publication (https://pubmed.ncbi.nlm.nih.gov/28973369), licensed under a CC-BY license. Not internally tested by Novus Biologicals.Applications for GAP-43 Antibody - BSA Free
Application
Recommended Usage
Immunocytochemistry/ Immunofluorescence
1:1000
Immunohistochemistry
1-2 ug/ml
Immunohistochemistry-Frozen
1-2 ug/ml
Immunohistochemistry-Paraffin
1-2 ug/ml
Western Blot
1:10000
Application Notes
This GAP43 antibody is useful for Immunocytochemistry/Immunofluorescence, and Western blot, where it recognizes a band at 43 kDa. Use in IHC-P and IHC-Fr reported in scientific literature (PMID: 29950987 and 27007292 respectively). Use in FLOW reported in scientific literature (PMID: 21614130).
Reviewed Applications
Read 1 review rated 5 using NB300-143 in the following applications:
Flow Cytometry Panel Builder
Bio-Techne Knows Flow Cytometry
Save time and reduce costly mistakes by quickly finding compatible reagents using the Panel Builder Tool.
Advanced Features
- Spectra Viewer - Custom analysis of spectra from multiple fluorochromes
- Spillover Popups - Visualize the spectra of individual fluorochromes
- Antigen Density Selector - Match fluorochrome brightness with antigen density
Formulation, Preparation, and Storage
Purification
Immunogen affinity purified
Formulation
50% PBS, 50% glycerol
Format
BSA Free
Preservative
0.035% Sodium Azide
Concentration
1 mg/ml
Shipping
The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below.
Stability & Storage
Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles.
Background: GAP-43
Long Name
Growth Associated Protein 43
Alternate Names
B-50, Basp2, GAP43, Neuromodulin
Gene Symbol
GAP43
Additional GAP-43 Products
Product Documents for GAP-43 Antibody - BSA Free
Certificate of Analysis
To download a Certificate of Analysis, please enter a lot or batch number in the search box below.
Product Specific Notices for GAP-43 Antibody - BSA Free
This product is for research use only and is not approved for use in humans or in clinical diagnosis. Primary Antibodies are guaranteed for 1 year from date of receipt.
Citations for GAP-43 Antibody - BSA Free
Customer Reviews for GAP-43 Antibody - BSA Free (1)
5 out of 5
1 Customer Rating
Have you used GAP-43 Antibody - BSA Free?
Submit a review and receive an Amazon gift card!
$25/€18/£15/$25CAN/¥2500 Yen for a review with an image
$10/€7/£6/$10CAN/¥1110 Yen for a review without an image
Submit a review
Customer Images
Showing
1
-
1 of
1 review
Showing All
Filter By:
-
Application: Immunohistochemistry-ParaffinSample Tested: E15.5 MouseSpecies: MouseVerified Customer | Posted 11/07/2013
There are no reviews that match your criteria.
Protocols
View specific protocols for GAP-43 Antibody - BSA Free (NB300-143):
IHC-FFPE sections
I. Deparaffinization:
A. Treat slides with Xylene: 3 changes for 5 minutes each. Drain slides for 10 seconds between changes.
B. Treat slides with 100% Reagent Alcohol: 3 changes for 5 minutes each. Drain slides for 10 seconds between changes.
II. Quench Endogenous Peroxidase:
A. Place slides in peroxidase quenching solution: 15-30 minutes. To Prepare 200 ml of Quenching Solution: Add 3 ml of 30% Hydrogen Peroxide to 200 ml of Methanol.
Use within 4 hours of preparation
B. Place slides in distilled water: 2 changes for 2 minutes each.
III. Retrieve Epitopes:
A. Preheat Citrate Buffer. Place 200 ml of Citrate Buffer Working Solution into container, cover and place into steamer. Heat to 90-96 degrees Celsius.
B. Place rack of slides into hot Citrate Buffer for 20 minutes. Cover.
C. Carefully remove container with slides from steamer and cool on bench, uncovered, for 20 minutes.
D. Slowly add distilled water to further cool for 5 minutes.
E. Rinse slides with distilled water. 2 changes for 2 minutes each.
IV. Immunostaining Procedure:
A. Remove each slide from rack and circle tissue section with a hydrophobic barrier pen (e.g. Liquid Blocker-Super Pap Pen).
B. Flood slide with Wash Solution. Do not allow tissue sections to dry for the rest of the procedure.
C. Drain wash solution and apply 4 drops of Blocking Reagent to each slide and incubate for 15 minutes.
D. Drain Blocking Reagent (do not wash off the Blocking Reagent), apply 200 ul of Primary Antibody solution to each slide, and incubate for 1 hour.
E. Wash slides with Wash Solution: 3 changes for 5 minutes each.
F. Drain wash solution, apply 4 drops of Secondary antibody to each slide and incubate for 1 hour.
G. Wash slides with Wash Solution: 3 changes for 5 minutes each.
H. Drain wash solution, apply 4 drops of DAB Substrate to each slide and develop for 5-10 minutes. Check development with microscope.
I. Wash slides with Wash Solution: 3 changes for 5 minutes each.
J. Drain wash solution, apply 4 drops of Hematoxylin to each slide and stain for 1-3 minutes. Increase time if darker counterstaining is desired.
K. Wash slides with Wash Solution: 2-3 changes for 2 minutes each.
L. Drain wash solution and apply 4 drops of Bluing Solution to each slide for 1-2 minutes.
M. Rinse slides in distilled water.
N. Soak slides in 70% reagent alcohol: 3 minutes with intermittent agitation.
O. Soak slides in 95% reagent alcohol: 2 changes for 3 minutes each with intermittent agitation.
P. Soak slides in 100% reagent alcohol: 3 changes for 3 minutes each with intermittent agitation. Drain slides for 10 seconds between each change.
Q. Soak slides in Xylene: 3 changes for 3 minutes each with intermittent agitation. Drain slides for 10 seconds between each change.
R. Apply 2-3 drops of non-aqueous mounting media to each slide and mount coverslip.
S. Lay slides on a flat surface to dry prior to viewing under microscope.
NOTES:
-Use treated slides (e.g. HistoBond) to assure adherence of FFPE sections to slide.
-Prior to deparaffinization, heat slides overnight in a 60 degrees Celsius oven.
-All steps in which Xylene is used should be performed in a fume hood.
-For Epitope Retrieval, a microwave or pressure cooker may be substituted for the steamer method. Adjust times as necessary depending on conditions.
-For the initial IHC run with a new primary antibody, test tissues with and without Epitope Retrieval. In some instances, Epitope Retrieval may not be necessary.
-200 ul is the recommended maximum volume to apply to a slide for full coverage. Using more than 200 ul may allow solutions to wick off the slide and create drying artifacts. For small tissue sections less than 200 ul may be used.
-5 minutes of development with DAB Substrate should be sufficient. Do not develop for more than 10 minutes. If 5 minutes of development causes background staining, further dilution of the primary antibody may be necessary.
-Hematoxylin should produce a light nuclear counterstain so as not to obscure the DAB staining. Counterstain for 1-1.5 minutes for nuclear antigens. Counterstain for 2-3 minutes for cytoplasmic and membranous antigens. If darker counterstaining is desired increase time (up to 10 minutes).
Find general support by application which include: protocols, troubleshooting, illustrated assays, videos and webinars.
- 7-Amino Actinomycin D (7-AAD) Cell Viability Flow Cytometry Protocol
- Antigen Retrieval Protocol (PIER)
- Antigen Retrieval for Frozen Sections Protocol
- Appropriate Fixation of IHC/ICC Samples
- Cellular Response to Hypoxia Protocols
- Chromogenic IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Chromogenic Immunohistochemistry Staining of Frozen Tissue
- ClariTSA™ Fluorophore Kits
- Detection & Visualization of Antibody Binding
- Extracellular Membrane Flow Cytometry Protocol
- Flow Cytometry Protocol for Cell Surface Markers
- Flow Cytometry Protocol for Staining Membrane Associated Proteins
- Flow Cytometry Staining Protocols
- Flow Cytometry Troubleshooting Guide
- Fluorescent IHC Staining of Frozen Tissue Protocol
- Graphic Protocol for Heat-induced Epitope Retrieval
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Graphic Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Graphic Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- ICC Cell Smear Protocol for Suspension Cells
- ICC Immunocytochemistry Protocol Videos
- ICC for Adherent Cells
- IHC Sample Preparation (Frozen sections vs Paraffin)
- Immunocytochemistry (ICC) Protocol
- Immunocytochemistry Troubleshooting
- Immunofluorescence of Organoids Embedded in Cultrex Basement Membrane Extract
- Immunofluorescent IHC Staining of Formalin-Fixed Paraffin-Embedded (FFPE) Tissue Protocol
- Immunohistochemistry (IHC) and Immunocytochemistry (ICC) Protocols
- Immunohistochemistry Frozen Troubleshooting
- Immunohistochemistry Paraffin Troubleshooting
- Intracellular Flow Cytometry Protocol Using Alcohol (Methanol)
- Intracellular Flow Cytometry Protocol Using Detergents
- Intracellular Nuclear Staining Flow Cytometry Protocol Using Detergents
- Intracellular Staining Flow Cytometry Protocol Using Alcohol Permeabilization
- Intracellular Staining Flow Cytometry Protocol Using Detergents to Permeabilize Cells
- Preparing Samples for IHC/ICC Experiments
- Preventing Non-Specific Staining (Non-Specific Binding)
- Primary Antibody Selection & Optimization
- Propidium Iodide Cell Viability Flow Cytometry Protocol
- Protocol for Heat-Induced Epitope Retrieval (HIER)
- Protocol for Liperfluo
- Protocol for Making a 4% Formaldehyde Solution in PBS
- Protocol for VisUCyte™ HRP Polymer Detection Reagent
- Protocol for the Characterization of Human Th22 Cells
- Protocol for the Characterization of Human Th9 Cells
- Protocol for the Fluorescent ICC Staining of Cell Smears - Graphic
- Protocol for the Fluorescent ICC Staining of Cultured Cells on Coverslips - Graphic
- Protocol for the Preparation & Fixation of Cells on Coverslips
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Frozen Tissue Sections - Graphic
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation and Chromogenic IHC Staining of Paraffin-embedded Tissue Sections - Graphic
- Protocol for the Preparation and Fluorescent ICC Staining of Cells on Coverslips
- Protocol for the Preparation and Fluorescent ICC Staining of Non-adherent Cells
- Protocol for the Preparation and Fluorescent ICC Staining of Stem Cells on Coverslips
- Protocol for the Preparation and Fluorescent IHC Staining of Frozen Tissue Sections
- Protocol for the Preparation and Fluorescent IHC Staining of Paraffin-embedded Tissue Sections
- Protocol for the Preparation of Gelatin-coated Slides for Histological Tissue Sections
- Protocol for the Preparation of a Cell Smear for Non-adherent Cell ICC - Graphic
- Protocol: Annexin V and PI Staining by Flow Cytometry
- Protocol: Annexin V and PI Staining for Apoptosis by Flow Cytometry
- R&D Systems Quality Control Western Blot Protocol
- TUNEL and Active Caspase-3 Detection by IHC/ICC Protocol
- The Importance of IHC/ICC Controls
- Troubleshooting Guide: Fluorokine Flow Cytometry Kits
- Troubleshooting Guide: Immunohistochemistry
- Troubleshooting Guide: Western Blot Figures
- Western Blot Conditions
- Western Blot Protocol
- Western Blot Protocol for Cell Lysates
- Western Blot Troubleshooting
- Western Blot Troubleshooting Guide
- View all Protocols, Troubleshooting, Illustrated assays and Webinars
Loading...

![Western Blot: GAP-43 Antibody [NB300-143] Western Blot: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Western-Blot-NB300-143-img0022.jpg)
![Immunohistochemistry: GAP-43 Antibody [NB300-143] Immunohistochemistry: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunohistochemistry-NB300-143-img0024.jpg)
![Immunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143] Immunohistochemistry-Paraffin: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunohistochemistry-Paraffin-NB300-143-img0023.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunocytochemistry-Immunofluorescence-NB300-143-img0021.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunocytochemistry-Immunofluorescence-NB300-143-img0018.jpg)
![Western Blot: GAP-43 Antibody [NB300-143] Western Blot: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Western-Blot-NB300-143-img0020.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunocytochemistry-Immunofluorescence-NB300-143-img0015.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143]](https://resources.rndsystems.com/images/products/GAP-43-Antibody-Immunocytochemistry-Immunofluorescence-NB300-143-img0017.jpg)
![Western Blot: GAP-43 Antibody [NB300-143] - GAP-43 Antibody](https://resources.rndsystems.com/images/products/nb300-143_rabbit-polyclonal-gap-43-antibody-310202415384199.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] - GAP-43 Antibody](https://resources.rndsystems.com/images/products/nb300-143_rabbit-polyclonal-gap-43-antibody-310202415334931.jpg)
![Western Blot: GAP-43 Antibody [NB300-143] - GAP-43 Antibody](https://resources.rndsystems.com/images/products/nb300-143_rabbit-polyclonal-gap-43-antibody-310202416212375.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] - GAP-43 Antibody](https://resources.rndsystems.com/images/products/nb300-143_rabbit-polyclonal-gap-43-antibody-310202416212312.jpg)
![Immunocytochemistry/ Immunofluorescence: GAP-43 Antibody [NB300-143] - GAP-43 Antibody](https://resources.rndsystems.com/images/products/nb300-143_rabbit-polyclonal-gap-43-antibody-31020241620512.jpg)